EX-99.2
Published on February 10, 2026

EVO301 Top-Line Results from Phase 2a Trial in Moderate-to-Severe Atopic Dermatitis February 10, 2026 © Evommune, Inc.

Forward-Looking Statements © Evommune, Inc. Statements contained in this presentation regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, as amended. Words such as "anticipates," "believes," "expects," "intends," “plans,” “potential,” "projects,” “would” and "future" or similar expressions are intended to identify forward-looking statements. Examples of these forward-looking statements include statements related to data supporting the Company’s ongoing clinical trials, development of EVO301 in inflammatory diseases, EVO301 mechanism of action and EVO301 as a new treatment option for patients with moderate to severe atopic dermatitis; the potential safety and efficacy of the Company’s product candidates; clinical strategy and the design of clinical trials, including the timeline for initiation, target or expected number of patients to be enrolled, dose levels, number of sites and other product development milestones; and the availability and timing of clinical and preclinical data announcements and clinical readouts, including data from the Phase 2a clinical trial for atopic dermatitis with EVO301. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. In particular, the following factors, among others, could cause results to differ materially from those expressed or implied by such forward-looking statements: the Company’s ability to demonstrate sufficient evidence of efficacy and safety in its clinical trials of its product candidates; the accuracy of the Company’s estimates relating to its ability to initiate and/or complete preclinical studies and clinical trials and release data from such studies and clinical trials; the results of preclinical studies and data from clinical trails not being predictive of future results; the Company’s ability to enroll sufficient numbers of patients in its clinical trials; the unpredictability of the regulatory process; regulatory developments in the United States and foreign countries; the costs of clinical trials may exceed expectations; and the Company’s ability to raise additional capital. These and other risks are described more fully in the Company’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, filed with the Securities and Exchange Commission (the “SEC”) on December 11, 2025, as well as other documents that may be filed by the Company from time to time with the SEC. All such statements speak only as of the date made, and the Company undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it cannot guarantee that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and the timing of events and circumstances and actual results could differ materially from those projected in the forward-looking statements. Accordingly, you should not place undue reliance on these forward-looking statements. This presentation concerns products that are under clinical investigation and which have not yet been approved for marketing by the U.S. Food and Drug Administration. Such products are currently limited by Federal law to investigational use, and no representation is made as to its safety or effectiveness for the purposes for which it is being investigated. This presentation also contains estimates made by independent parties relating to industry market size and other data. These estimates involve a number of assumptions and limitations, and you are cautioned not to give undue weight on such estimates. We have not independently verified the accuracy or completeness of such information and we do not take any responsibility with the accuracy or completeness of such information. The trademarks included in this presentation are the property of the owners thereof and are used for reference purposes only.

Luis Peña Founder, President & CEO Mark Jackson, MD SVP, Clinical Development Agenda and Speakers Introduction Luis Peña Founder, President & CEO IL-18 Development Program Mark Jackson, MD SVP, Clinical Development EVO301 Phase 2a Data Mark Jackson, MD SVP, Clinical Development Closing Remarks Luis Peña Founder, President & CEO 3 © Evommune, Inc.
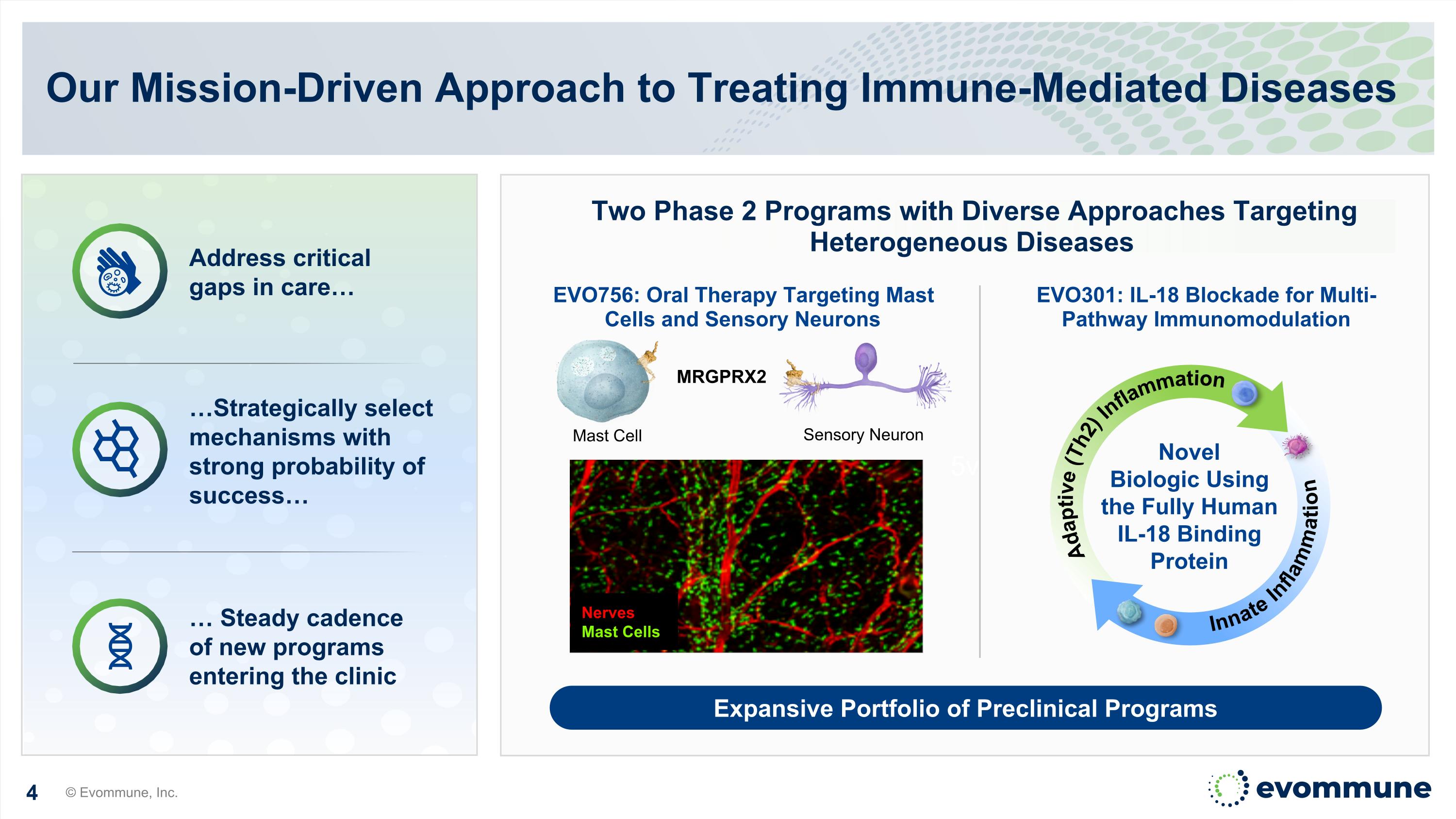
5v Our Mission-Driven Approach to Treating Immune-Mediated Diseases © Evommune, Inc. Two Phase 2 Programs with Diverse Approaches Targeting Heterogeneous Diseases EVO756: Oral Therapy Targeting Mast Cells and Sensory Neurons EVO301: IL-18 Blockade for Multi-Pathway Immunomodulation Address critical gaps in care… Expansive Portfolio of Preclinical Programs Sensory Neuron Mast Cell MRGPRX2 Nerves Mast Cells Novel Biologic Using the Fully Human IL-18 Binding Protein Adaptive (Th2) Inflammation Innate Inflammation …Strategically select mechanisms with strong probability of success… … Steady cadence of new programs entering the clinic
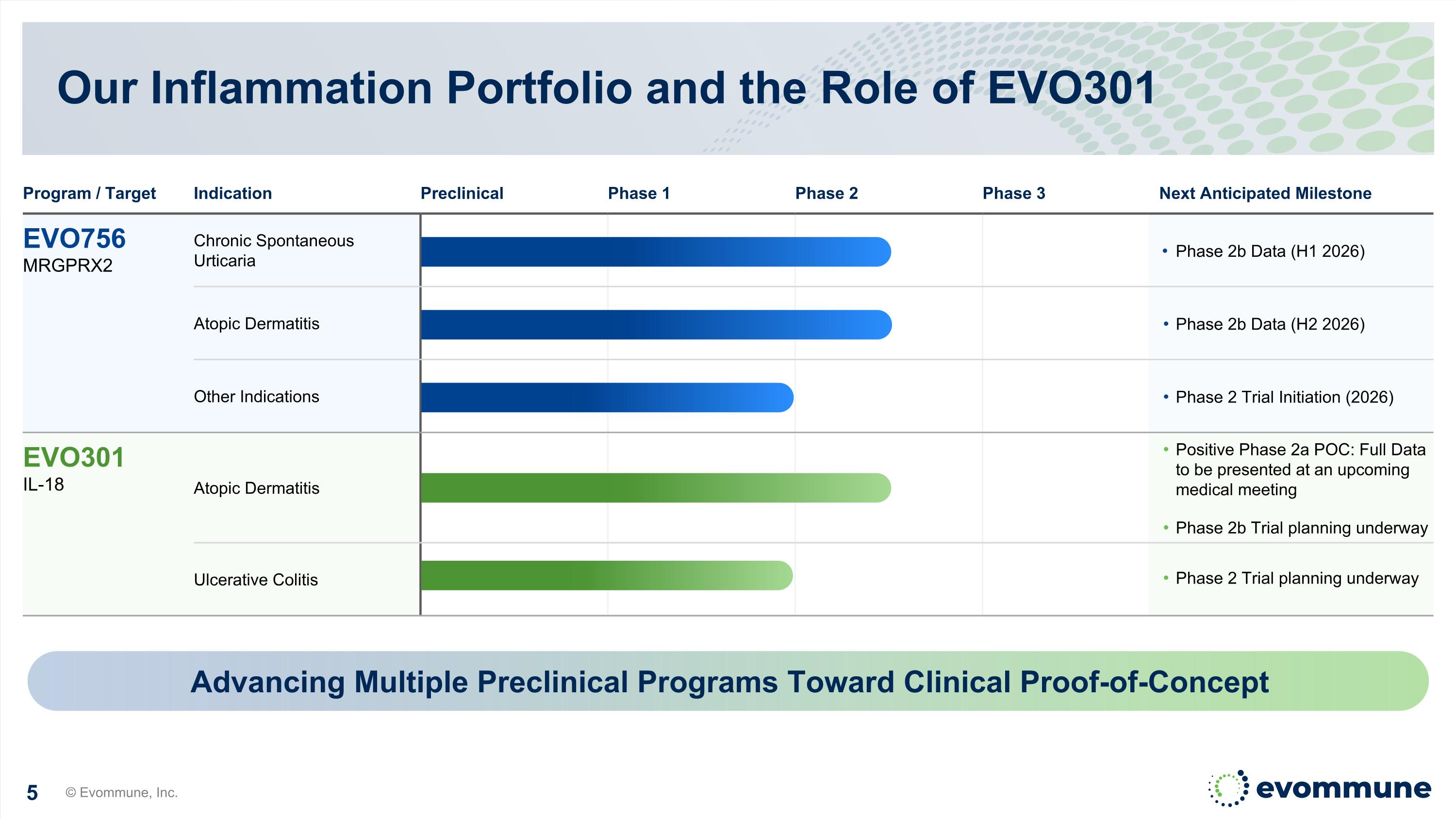
Our Inflammation Portfolio and the Role of EVO301 © Evommune, Inc. Program / Target Indication Preclinical Phase 1 Phase 2 Phase 3 Next Anticipated Milestone EVO756 MRGPRX2 Chronic Spontaneous Urticaria Phase 2b Data (H1 2026) Atopic Dermatitis Phase 2b Data (H2 2026) Other Indications Phase 2 Trial Initiation (2026) EVO301 IL-18 Atopic Dermatitis Positive Phase 2a POC: Full Data to be presented at an upcoming medical meeting Phase 2b Trial planning underway Ulcerative Colitis Phase 2 Trial planning underway Advancing Multiple Preclinical Programs Toward Clinical Proof-of-Concept
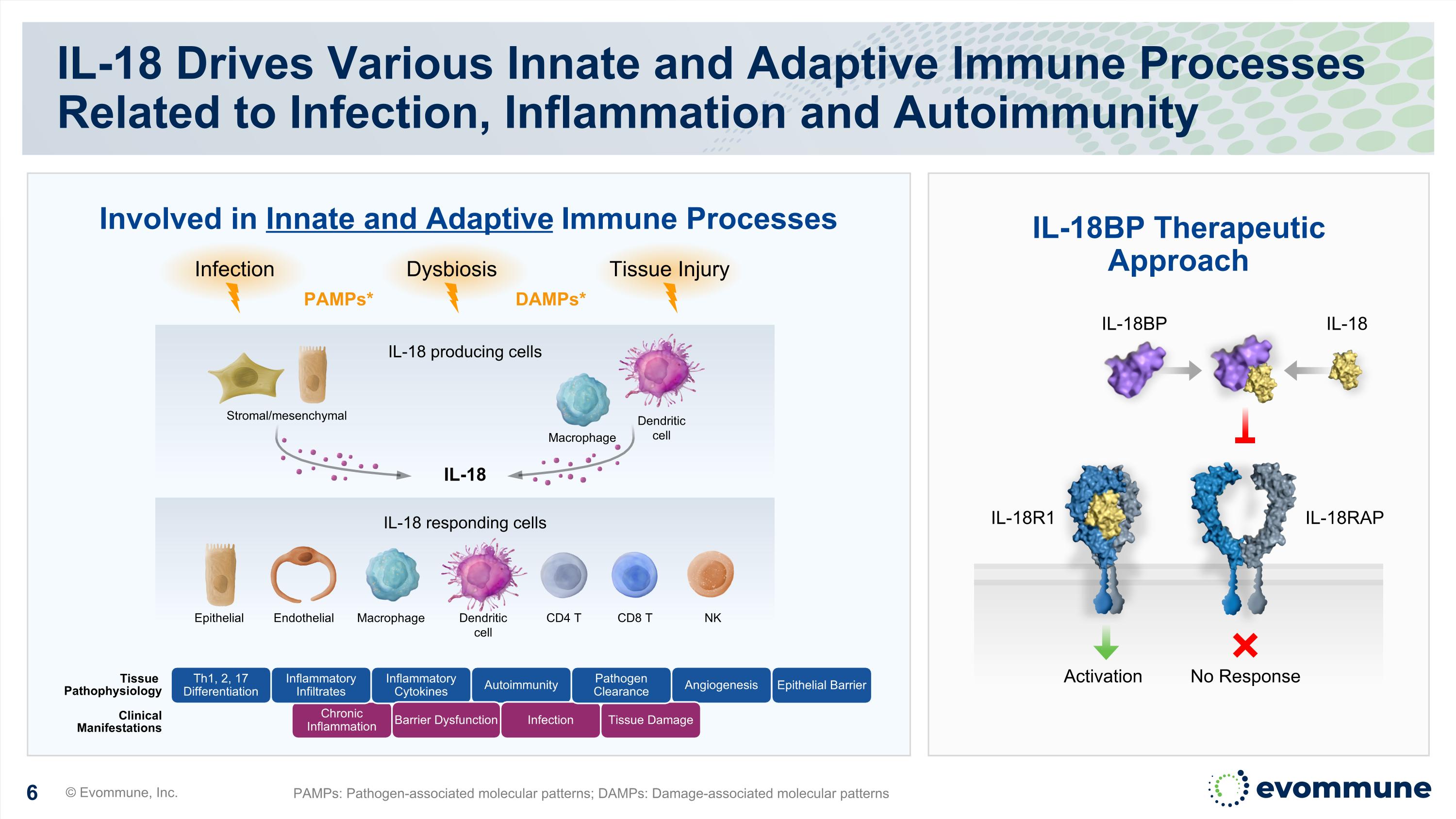
IL-18 Drives Various Innate and Adaptive Immune Processes Related to Infection, Inflammation and Autoimmunity © Evommune, Inc. PAMPs: Pathogen-associated molecular patterns; DAMPs: Damage-associated molecular patterns Chronic Inflammation Tissue Pathophysiology Clinical Manifestations Inflammatory Infiltrates Inflammatory Cytokines Autoimmunity Angiogenesis Th1, 2, 17 Differentiation Barrier Dysfunction Infection Tissue Damage Pathogen Clearance IL-18BP Therapeutic Approach Involved in Innate and Adaptive Immune Processes IL-18 producing cells IL-18 responding cells Stromal/mesenchymal IL-18 Epithelial Endothelial CD4 T NK CD8 T Macrophage Dendritic cell Dendritic cell Macrophage Dysbiosis Tissue Injury Infection DAMPs* PAMPs* IL-18R1 IL-18RAP No Response Activation IL-18 IL-18BP Epithelial Barrier
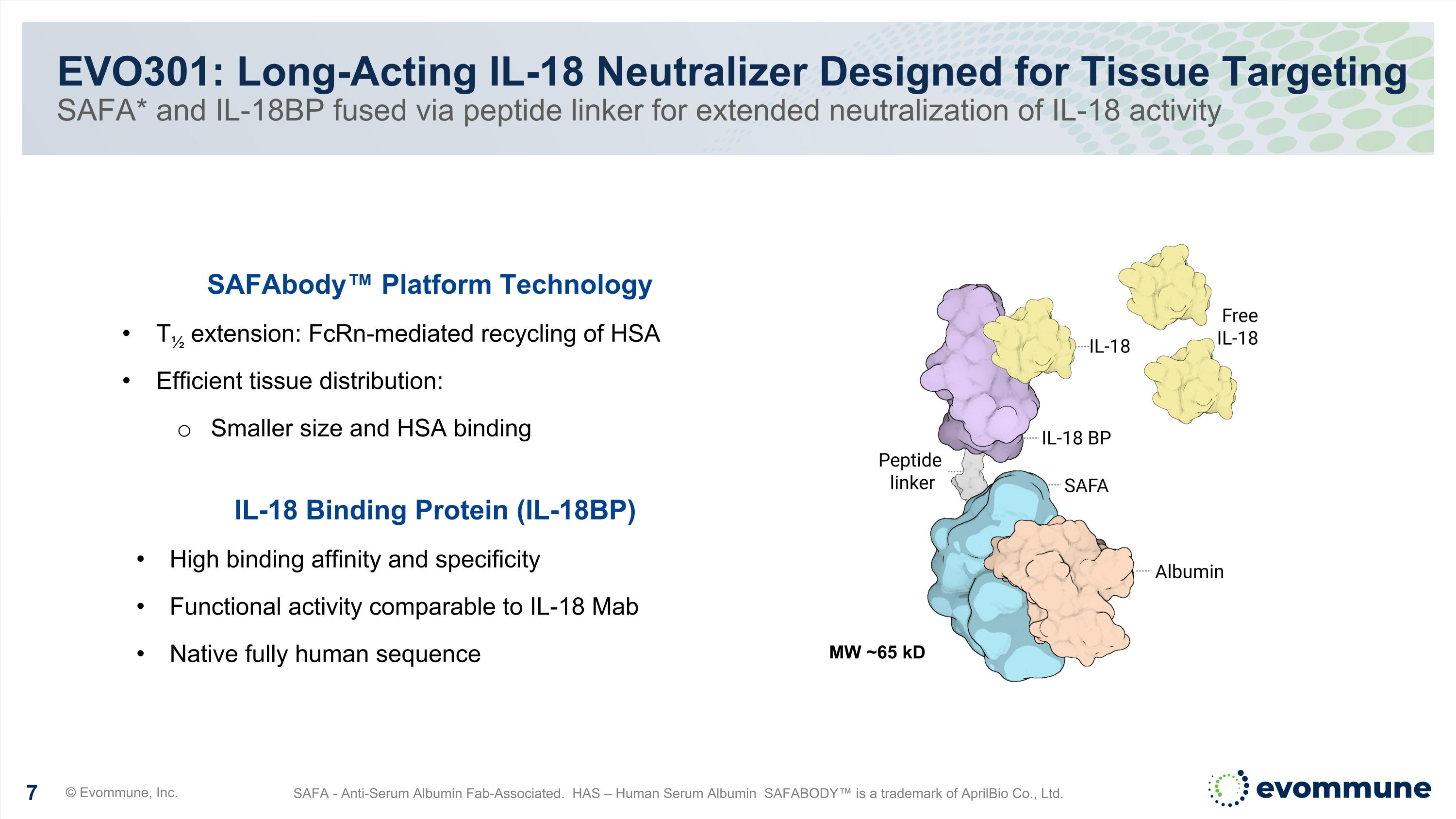
EVO301: Long-Acting IL-18 Neutralizer Designed for Tissue Targeting SAFA* and IL-18BP fused via peptide linker for extended neutralization of IL-18 activity © Evommune, Inc. SAFA - Anti-Serum Albumin Fab-Associated. HAS – Human Serum Albumin SAFABODY™ is a trademark of AprilBio Co., Ltd. SAFAbody™ Platform Technology T½ extension: FcRn-mediated recycling of HSA Efficient tissue distribution: Smaller size and HSA binding IL-18 Binding Protein (IL-18BP) High binding affinity and specificity Functional activity comparable to IL-18 Mab Native fully human sequence MW ~65 kD
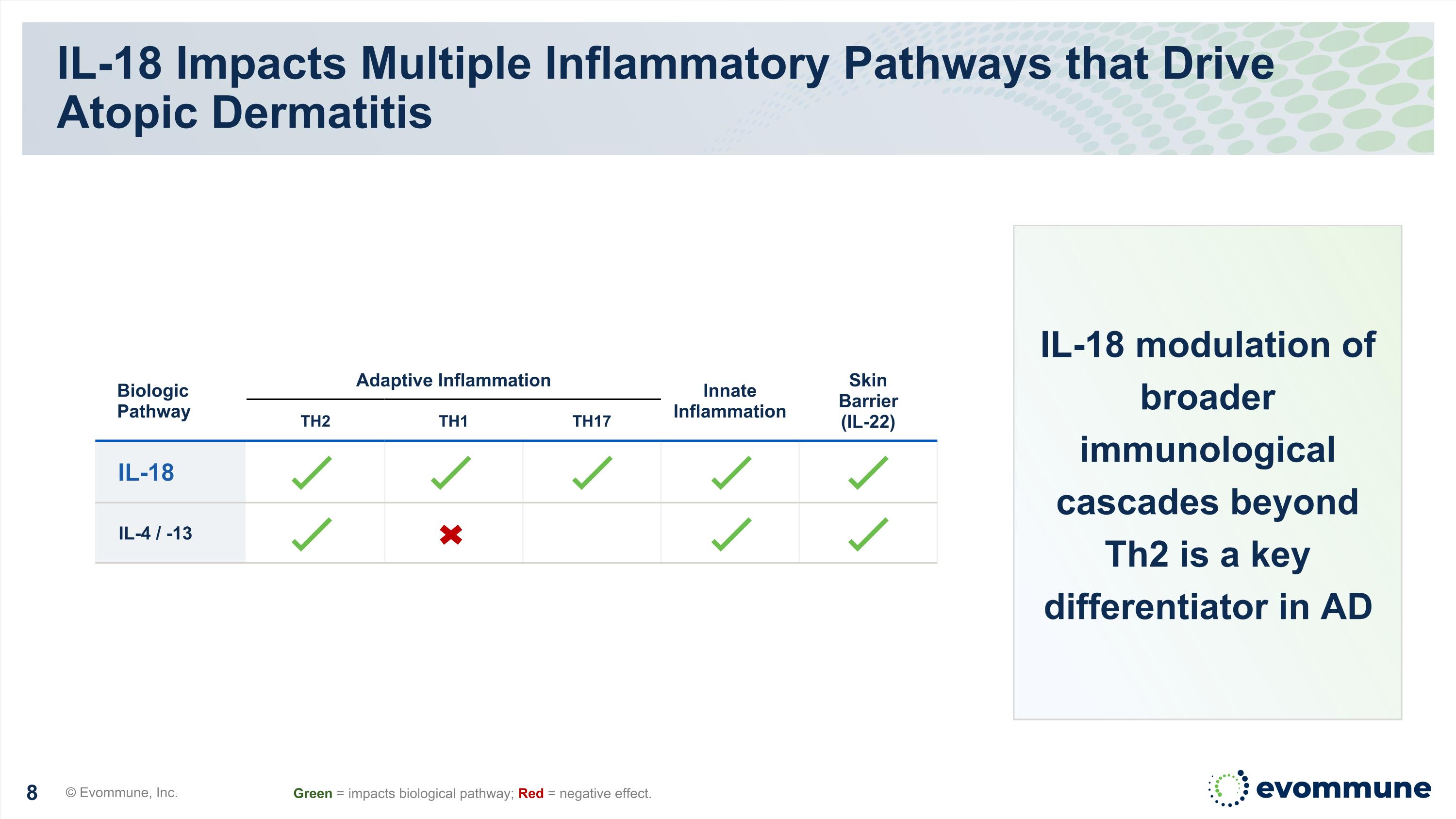
Biologic Pathway Adaptive Inflammation Innate Inflammation Skin Barrier (IL-22) TH2 TH1 TH17 IL-18 IL-4 / -13 IL-18 Impacts Multiple Inflammatory Pathways that Drive Atopic Dermatitis © Evommune, Inc. Green = impacts biological pathway; Red = negative effect. IL-18 modulation of broader immunological cascades beyond Th2 is a key differentiator in AD

EVO301 Achieved the Primary Endpoint Phase 2a Proof-of-Concept Trial in Moderate-to-Severe Atopic Dermatitis Highly statistically significant EASI reductions at weeks 4, 8, and 12 versus placebo 34% and 33% placebo adjusted improvement in EASI at week 8 and 12, respectively 23% of patients achieved IGA 0/1 at week 12 versus 0% placebo Well-tolerated, with no treatment related serious or severe adverse events reported Corresponding reductions in secondary endpoints, as well as key Th2 and non Th2 cytokines Pharmacokinetics (PK) continues to support a Q4 week dosing regimen © Evommune, Inc. Clinical Data Supports Continued Development, with Phase 2b Planning Underway

EVO301 Phase 2a Proof of Concept Trial Design © Evommune, Inc. EASI = Eczema Area and Severity Index; vIGA = Validated Investigator Global Assessment; BSA = Body Surface Area; BL = Baseline; PBO = Placebo Screening Randomization 2 Active : 1 PBO End of Trial BL W12 W4 Adults with Moderate-to-Severe Atopic Dermatitis (N = 70) Randomized, Double-Blind, Parallel Group, Placebo-Controlled Trial AD Population EASI ≥16 vIGA ≥3 BSA ≥10% Primary Endpoint Percent change from EASI at Week 12 (Bayesian) Pharmacokinetics Target Engagement W8 EVO301: 5 mg/kg IV Placebo Dosing day Dosing day W2
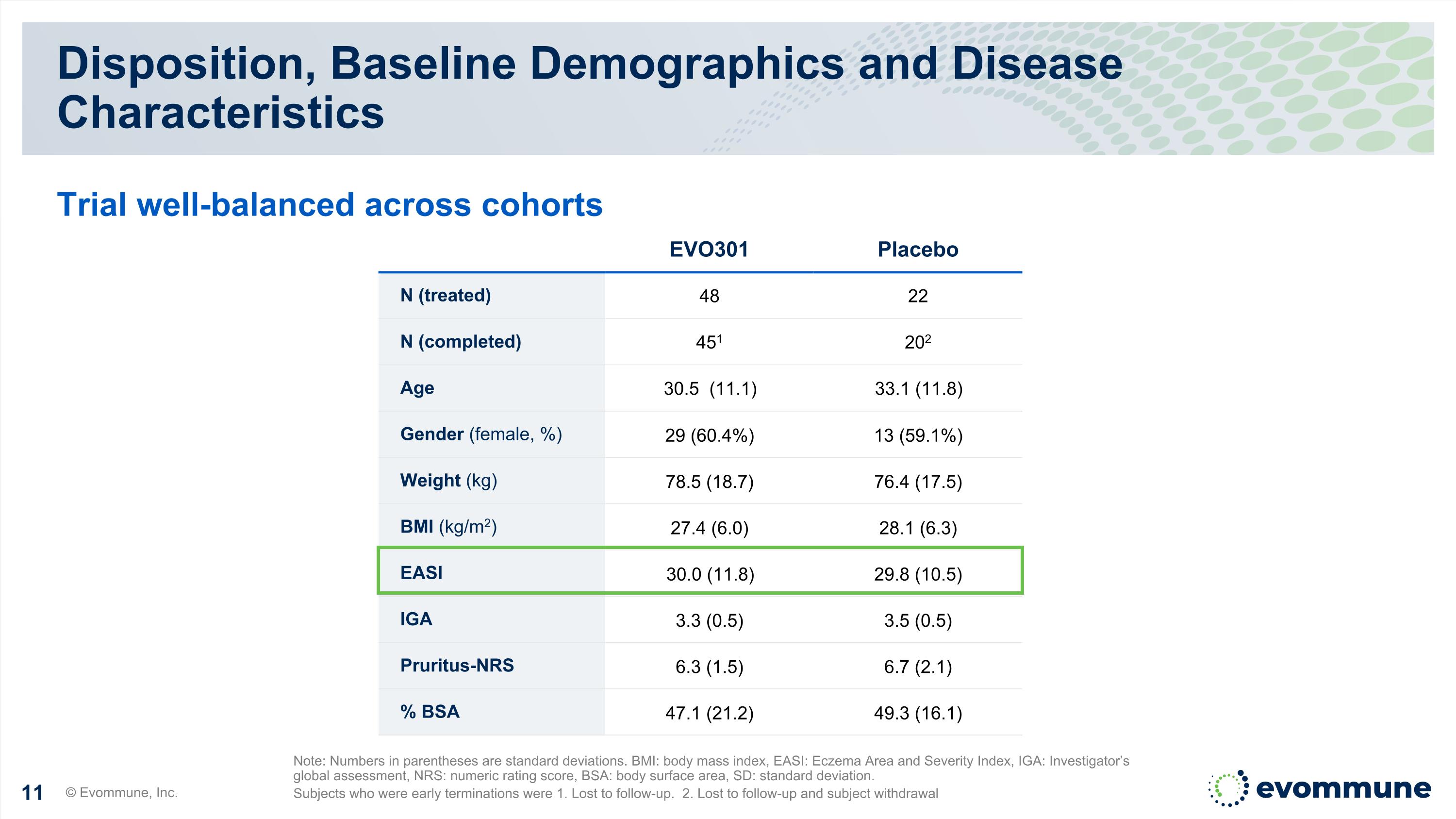
Disposition, Baseline Demographics and Disease Characteristics © Evommune, Inc. Note: Numbers in parentheses are standard deviations. BMI: body mass index, EASI: Eczema Area and Severity Index, IGA: Investigator’s global assessment, NRS: numeric rating score, BSA: body surface area, SD: standard deviation. Subjects who were early terminations were 1. Lost to follow-up. 2. Lost to follow-up and subject withdrawal Trial well-balanced across cohorts EVO301 Placebo N (treated) 48 22 N (completed) 451 202 Age 30.5 (11.1) 33.1 (11.8) Gender (female, %) 29 (60.4%) 13 (59.1%) Weight (kg) 78.5 (18.7) 76.4 (17.5) BMI (kg/m2) 27.4 (6.0) 28.1 (6.3) EASI 30.0 (11.8) 29.8 (10.5) IGA 3.3 (0.5) 3.5 (0.5) Pruritus-NRS 6.3 (1.5) 6.7 (2.1) % BSA 47.1 (21.2) 49.3 (16.1)
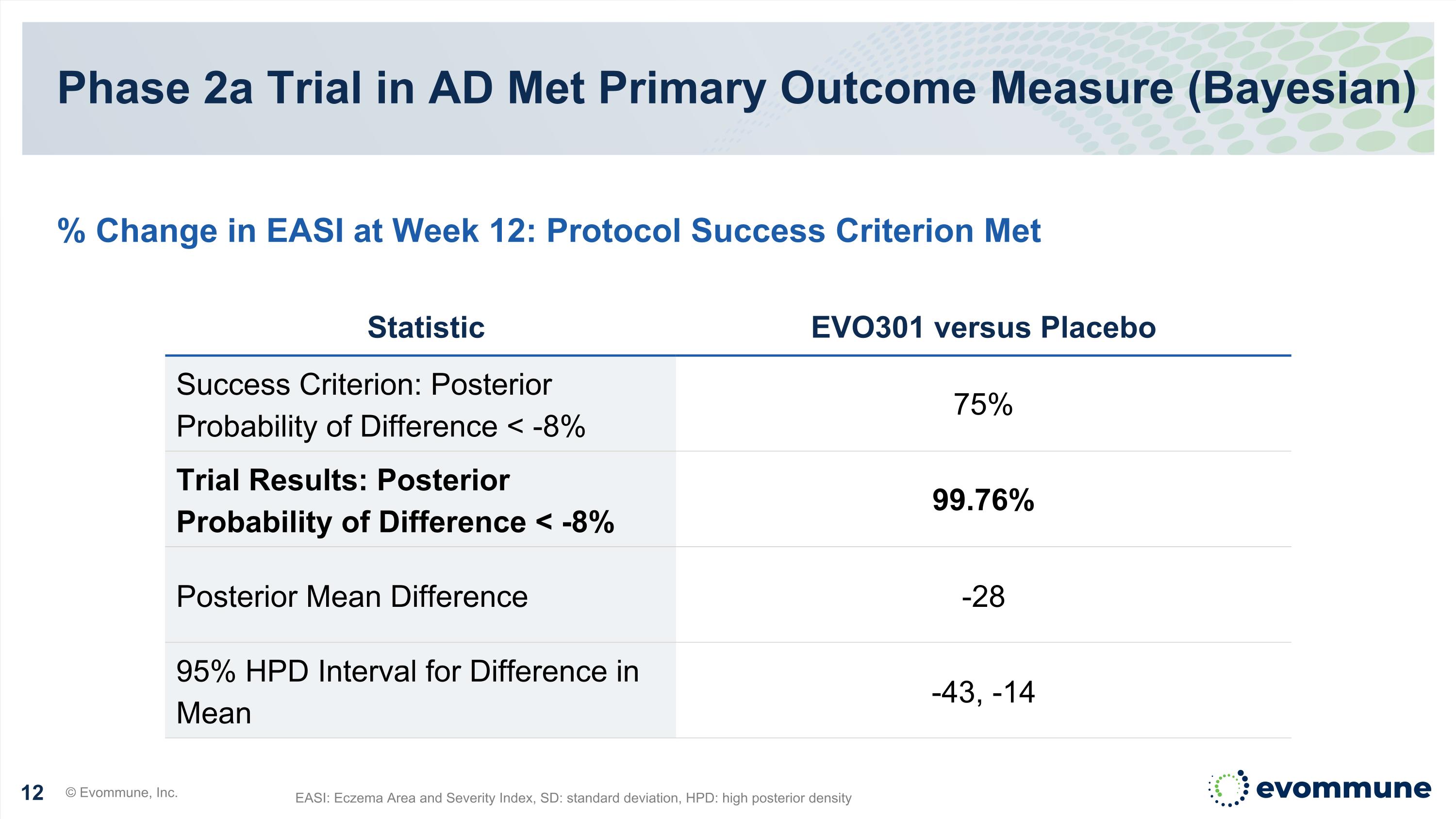
Phase 2a Trial in AD Met Primary Outcome Measure (Bayesian) EASI: Eczema Area and Severity Index, SD: standard deviation, HPD: high posterior density % Change in EASI at Week 12: Protocol Success Criterion Met Statistic EVO301 versus Placebo Success Criterion: Posterior Probability of Difference < -8% 75% Trial Results: Posterior Probability of Difference < -8% 99.76% Posterior Mean Difference -28 95% HPD Interval for Difference in Mean -43, -14 © Evommune, Inc.
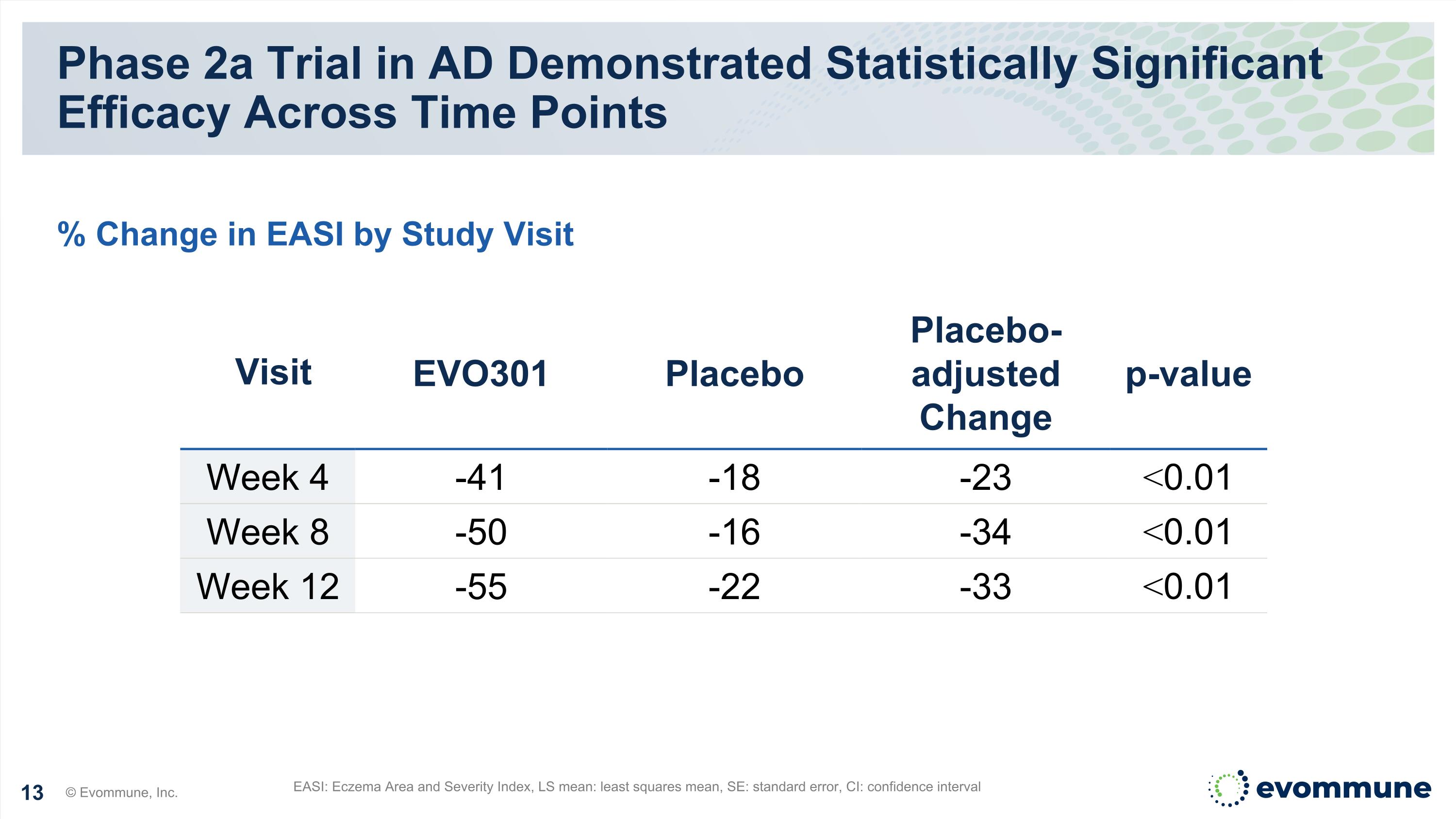
Phase 2a Trial in AD Demonstrated Statistically Significant Efficacy Across Time Points EASI: Eczema Area and Severity Index, LS mean: least squares mean, SE: standard error, CI: confidence interval % Change in EASI by Study Visit Visit EVO301 Placebo Placebo-adjusted Change p-value Week 4 -41 -18 -23 <0.01 Week 8 -50 -16 -34 <0.01 Week 12 -55 -22 -33 <0.01 © Evommune, Inc.
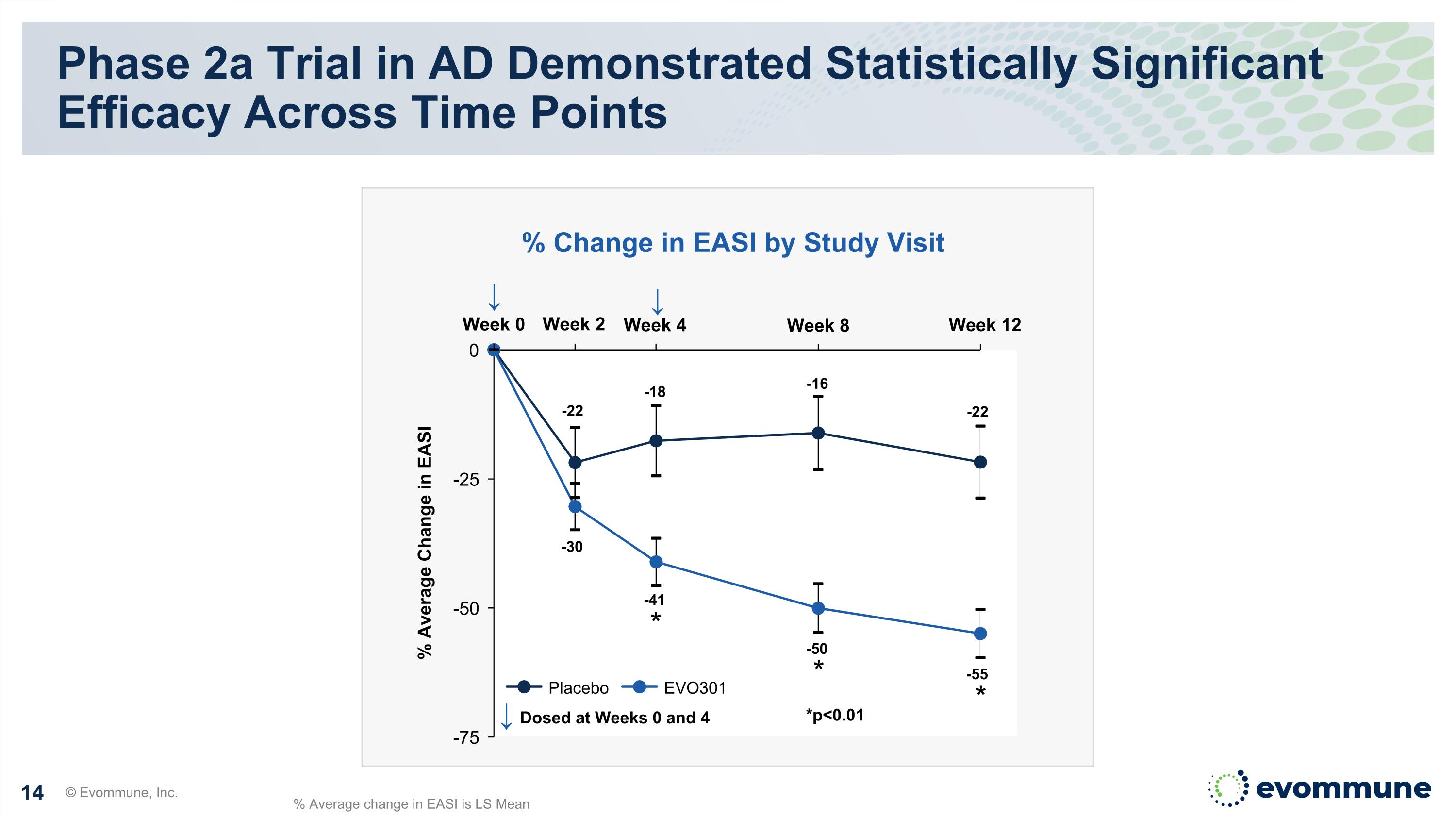
Week 2 Phase 2a Trial in AD Demonstrated Statistically Significant Efficacy Across Time Points © Evommune, Inc. Week 0 Week 4 Week 8 Week 12 % Average Change in EASI ↓ Dosed at Weeks 0 and 4 Placebo EVO301 * * % Change in EASI by Study Visit *p<0.01 ↓ % Average change in EASI is LS Mean -22 -18 -16 -22 -30 -41 -50 -55 ↓ *
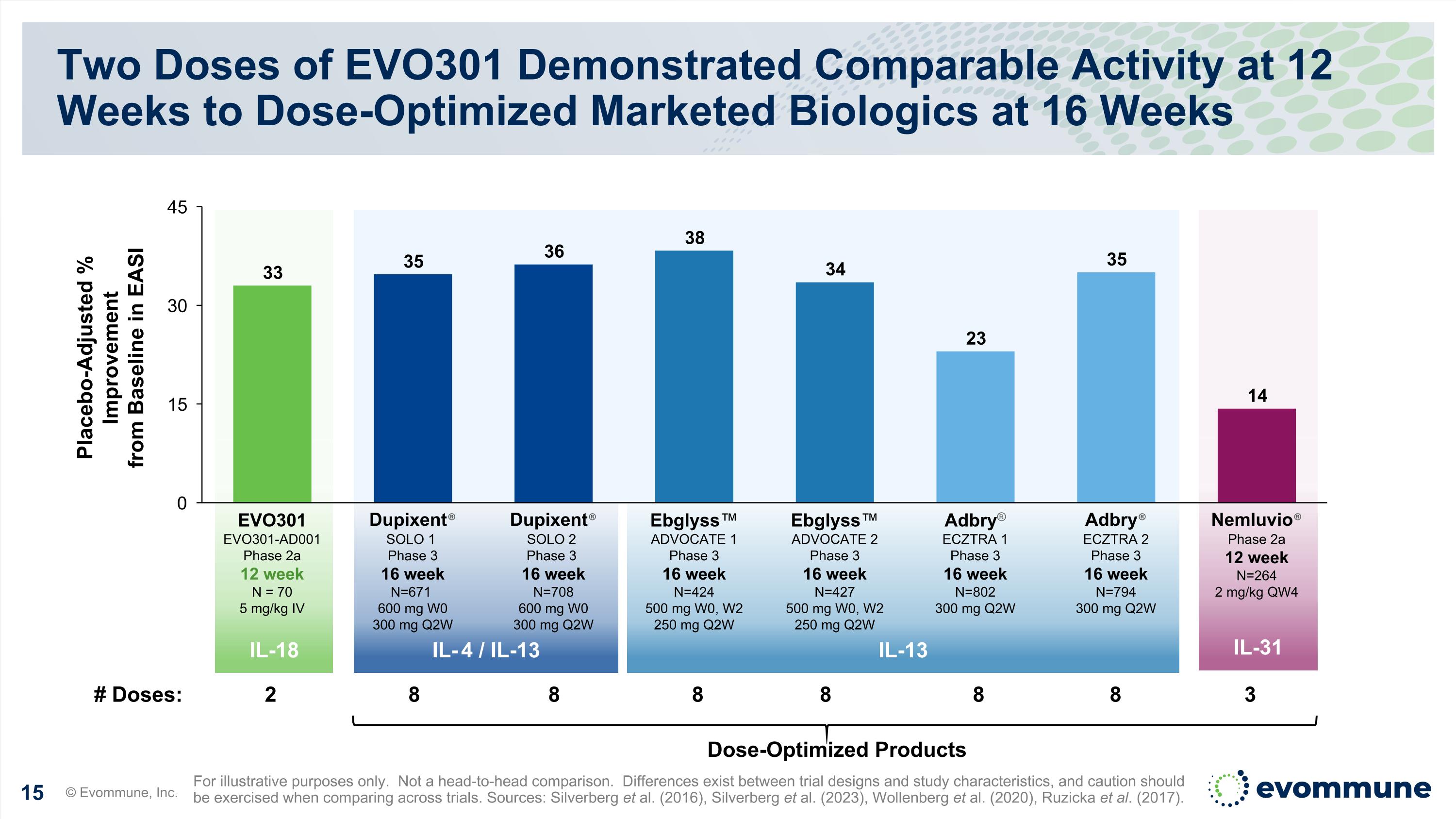
IL-18 IL-31 IL-13 IL-4 / IL-13 Two Doses of EVO301 Demonstrated Comparable Activity at 12 Weeks to Dose-Optimized Marketed Biologics at 16 Weeks © Evommune, Inc. For illustrative purposes only. Not a head-to-head comparison. Differences exist between trial designs and study characteristics, and caution should be exercised when comparing across trials. Sources: Silverberg et al. (2016), Silverberg et al. (2023), Wollenberg et al. (2020), Ruzicka et al. (2017). Placebo-Adjusted % Improvement from Baseline in EASI Dose-Optimized Products # Doses: 2 8 8 8 8 8 8 3 EVO301 EVO301-AD001 Phase 2a 12 week N = 70 5 mg/kg IV Dupixent® SOLO 1 Phase 3 16 week N=671 600 mg W0 300 mg Q2W Dupixent® SOLO 2 Phase 3 16 week N=708 600 mg W0 300 mg Q2W Ebglyss™ ADVOCATE 1 Phase 3 16 week N=424 500 mg W0, W2 250 mg Q2W Ebglyss™ ADVOCATE 2 Phase 3 16 week N=427 500 mg W0, W2 250 mg Q2W Adbry® ECZTRA 1 Phase 3 16 week N=802 300 mg Q2W Adbry® ECZTRA 2 Phase 3 16 week N=794 300 mg Q2W Nemluvio® Phase 2a 12 week N=264 2 mg/kg QW4
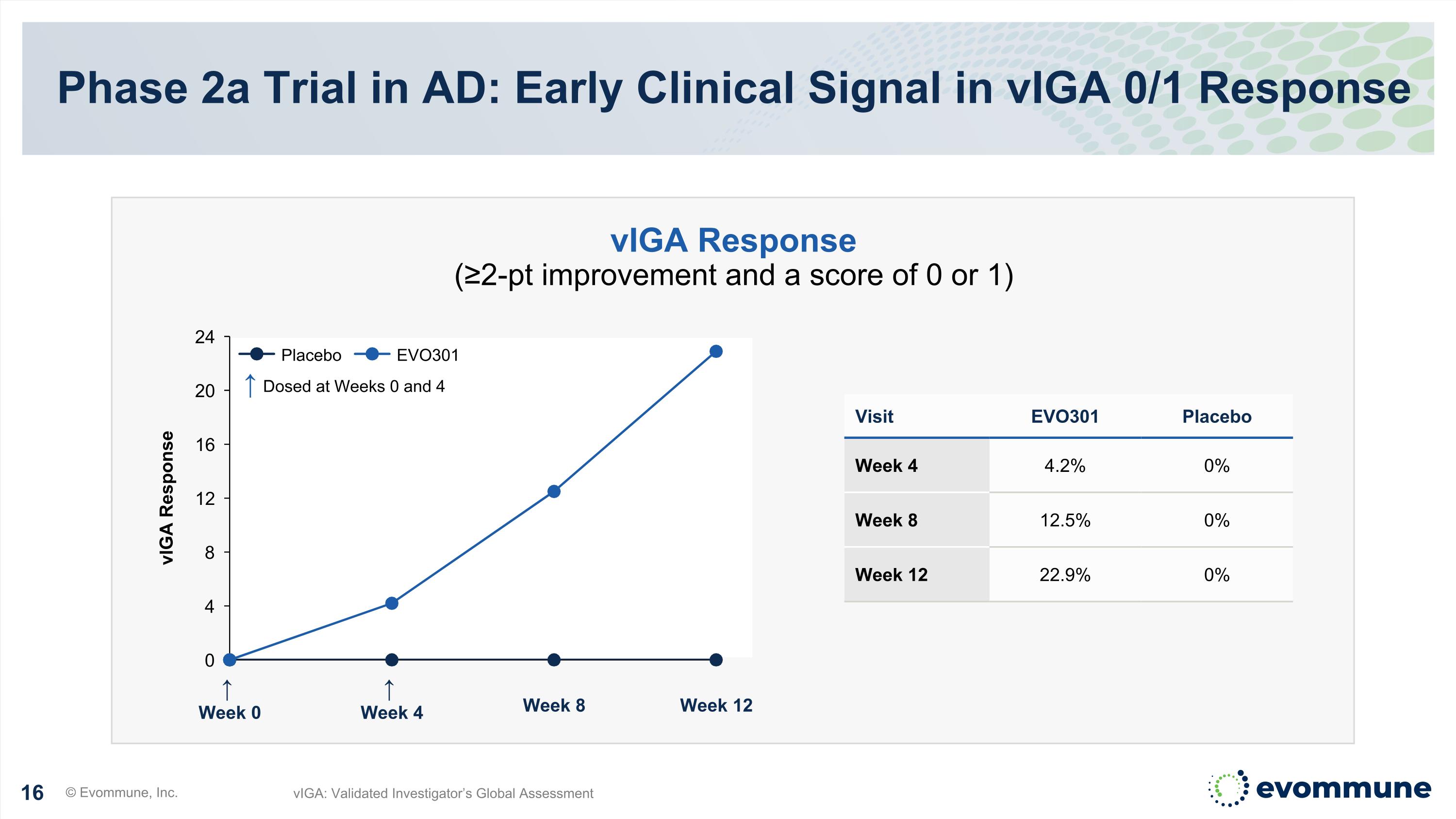
↑ Week 0 ↑ Week 4 Week 8 Week 12 vIGA Response Phase 2a Trial in AD: Early Clinical Signal in vIGA 0/1 Response © Evommune, Inc. vIGA: Validated Investigator’s Global Assessment Visit EVO301 Placebo Week 4 4.2% 0% Week 8 12.5% 0% Week 12 22.9% 0% vIGA Response (≥2-pt improvement and a score of 0 or 1) ↑ Dosed at Weeks 0 and 4 Placebo EVO301

Additional Endpoints and Target Engagement © Evommune, Inc. Full Detailed Results Will Be Presented at a Future Scientific Conference PK and target engagement data continue to support a Q4 week dosing regimen Corresponding reductions in secondary endpoints Corresponding reduction of key Th2 associated biomarkers (CCL-17 (TARC), CCL-22) and non-Th2 associated biomarkers (IL-22)
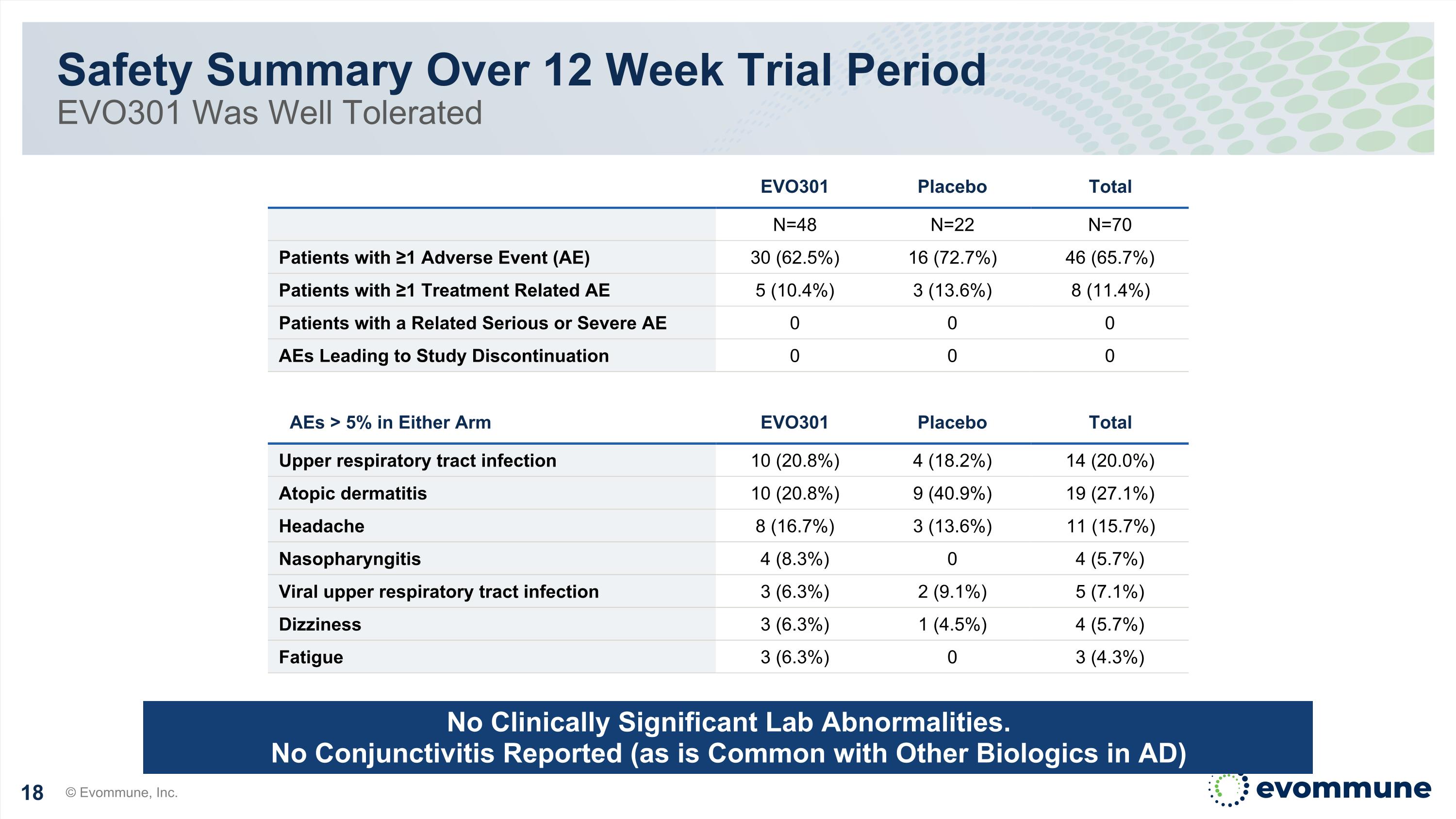
Safety Summary Over 12 Week Trial Period EVO301 Was Well Tolerated © Evommune, Inc. No Clinically Significant Lab Abnormalities. No Conjunctivitis Reported (as is Common with Other Biologics in AD) EVO301 Placebo Total N=48 N=22 N=70 Patients with ≥1 Adverse Event (AE) 30 (62.5%) 16 (72.7%) 46 (65.7%) Patients with ≥1 Treatment Related AE 5 (10.4%) 3 (13.6%) 8 (11.4%) Patients with a Related Serious or Severe AE 0 0 0 AEs Leading to Study Discontinuation 0 0 0 AEs > 5% in Either Arm EVO301 Placebo Total Upper respiratory tract infection 10 (20.8%) 4 (18.2%) 14 (20.0%) Atopic dermatitis 10 (20.8%) 9 (40.9%) 19 (27.1%) Headache 8 (16.7%) 3 (13.6%) 11 (15.7%) Nasopharyngitis 4 (8.3%) 0 4 (5.7%) Viral upper respiratory tract infection 3 (6.3%) 2 (9.1%) 5 (7.1%) Dizziness 3 (6.3%) 1 (4.5%) 4 (5.7%) Fatigue 3 (6.3%) 0 3 (4.3%)
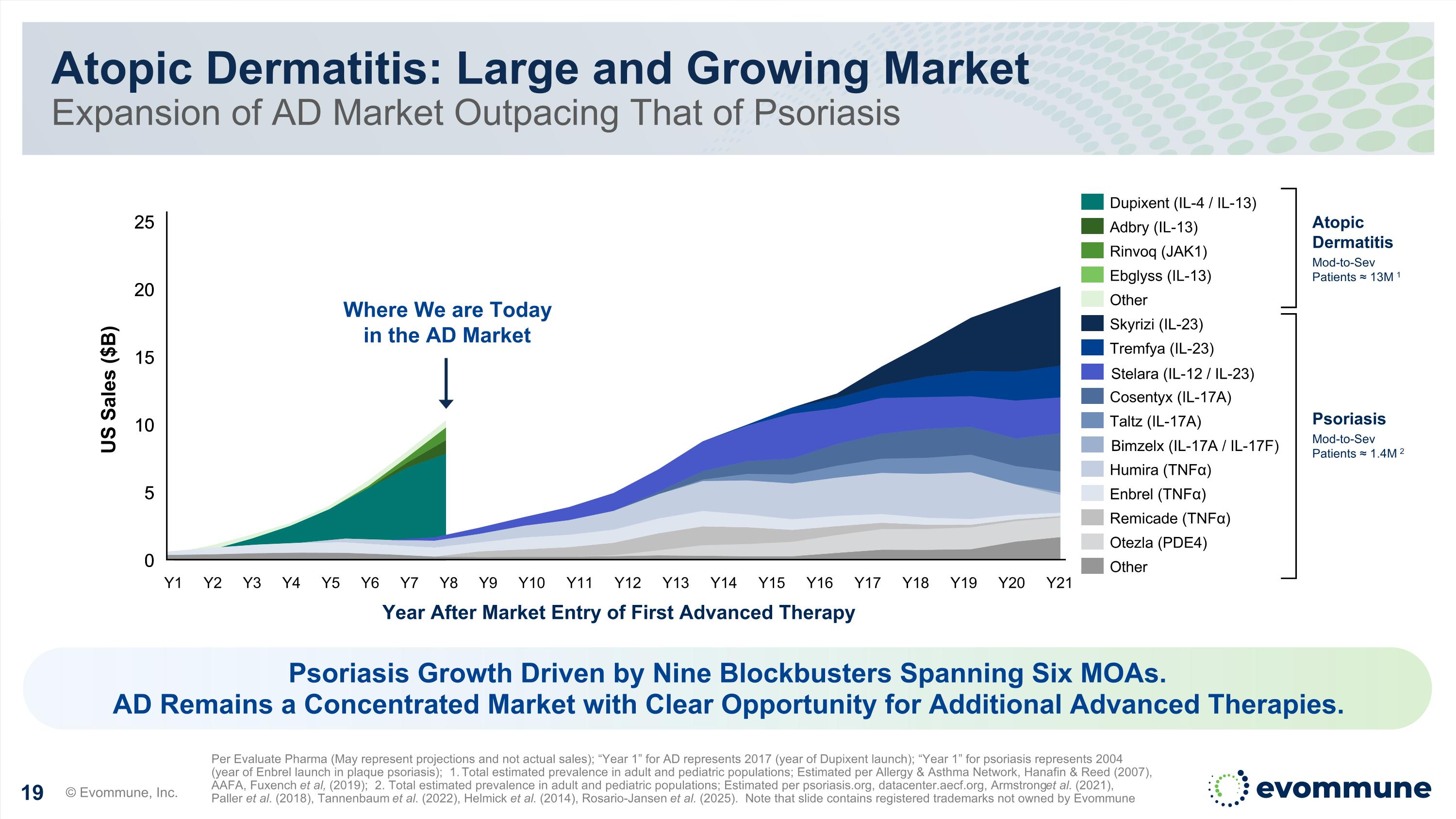
Atopic Dermatitis: Large and Growing Market Expansion of AD Market Outpacing That of Psoriasis © Evommune, Inc. Per Evaluate Pharma (May represent projections and not actual sales); “Year 1” for AD represents 2017 (year of Dupixent launch); “Year 1” for psoriasis represents 2004 (year of Enbrel launch in plaque psoriasis); 1. Total estimated prevalence in adult and pediatric populations; Estimated per Allergy & Asthma Network, Hanafin & Reed (2007), AAFA, Fuxench et al, (2019); 2. Total estimated prevalence in adult and pediatric populations; Estimated per psoriasis.org, datacenter.aecf.org, Armstrong et al. (2021), Paller et al. (2018), Tannenbaum et al. (2022), Helmick et al. (2014), Rosario-Jansen et al. (2025). Note that slide contains registered trademarks not owned by Evommune 0 5 10 15 20 25 0 5 10 15 20 25 Year After Market Entry of First Advanced Therapy US Sales ($B) Y1 Y2 Y3 Y4 Y5 Y6 Y7 Y8 Y9 Y10 Y11 Y12 Y13 Y14 Y15 Y16 Y17 Y18 Y19 Y20 Y21 Dupixent (IL-4 / IL-13) Adbry (IL-13) Rinvoq (JAK1) Ebglyss (IL-13) Other Skyrizi (IL-23) Tremfya (IL-23) Stelara (IL-12/IL-23) Cosentyx (IL-17A) Taltz (IL-17A) Bimzelx (IL-17A/IL-17F) Humira (TNFα) Enbrel (TNFα) Remicade (TNFα) Otezla (PDE4) Other Atopic Dermatitis Mod-to-Sev Patients ≈ 13M 1 Where We are Today in the AD Market Psoriasis Growth Driven by Nine Blockbusters Spanning Six MOAs. AD Remains a Concentrated Market with Clear Opportunity for Additional Advanced Therapies. Stelara (IL-12 / IL-23) Bimzelx (IL-17A / IL-17F) Psoriasis Mod-to-Sev Patients ≈ 1.4M 2

Summary and Path Forward to Phase 2b and Beyond © Evommune, Inc. IL-18 modulation of broader immunological cascades beyond Th2 is a key differentiator in AD EVO301 provides unique approach to IL-18 targeting, as a long-acting fusion protein consisting of a binding protein and an anti-serum albumin Fab EVO301 achieved the primary endpoint in the Ph2a POC trial Highly statistically significant efficacy at weeks 4, 8 and 12, after just two doses of EVO301 23% of EVO301 patients achieved IGA 0/1 at week 12 Full results from Phase 2a trial will be presented at a future scientific conference Clinical data clearly support continued development IL-18 biology extends far beyond AD, with IL-18 upregulated across a number of inflammatory diseases Phase 2b planning underway for AD with SubQ formulation, with POC planning underway for Ulcerative Colitis Multiple Drivers of Value Differentiated Mechanism with Broad Potential Clear Efficacy in AD

With Two Clinically Validated Programs, Evommune Continues to Deliver on our Strategy © Evommune, Inc. Program / Target Indication Preclinical Phase 1 Phase 2 Phase 3 Next Anticipated Milestone EVO756 MRGPRX2 Chronic Spontaneous Urticaria Phase 2b Data (H1 2026) Atopic Dermatitis Phase 2b Data (H2 2026) Other Indications Phase 2 Trial Initiation (2026) EVO301 IL-18 Atopic Dermatitis Positive Phase 2a POC: Full Data to be presented at an upcoming medical meeting Phase 2b Trial planning underway Ulcerative Colitis Phase 2 Trial planning underway

Q&A © Evommune, Inc.

Appendix © Evommune, Inc.
Moderate-to-Severe Atopic Dermatitis: Heterogenous Disease that is Prevalent, Impactful, and Underserved © Evommune, Inc. Source: Habif’s Clinical Dermatology: A Color Guide to Diagnosis and Therapy. National Eczema Association (accessed 2026), Luckhaupt, et al. (2013). Dupixent Prescribing Information. Rinvoq Prescribing Information. Substantial Need for Broad, Safe Treatments ~13M Americans live with moderate-to-severe atopic dermatitis Chronic, relapsing disease characterized by relentless itch and visible inflammation Significant quality-of-life impact ~40% of patients inadequately controlled with current therapies
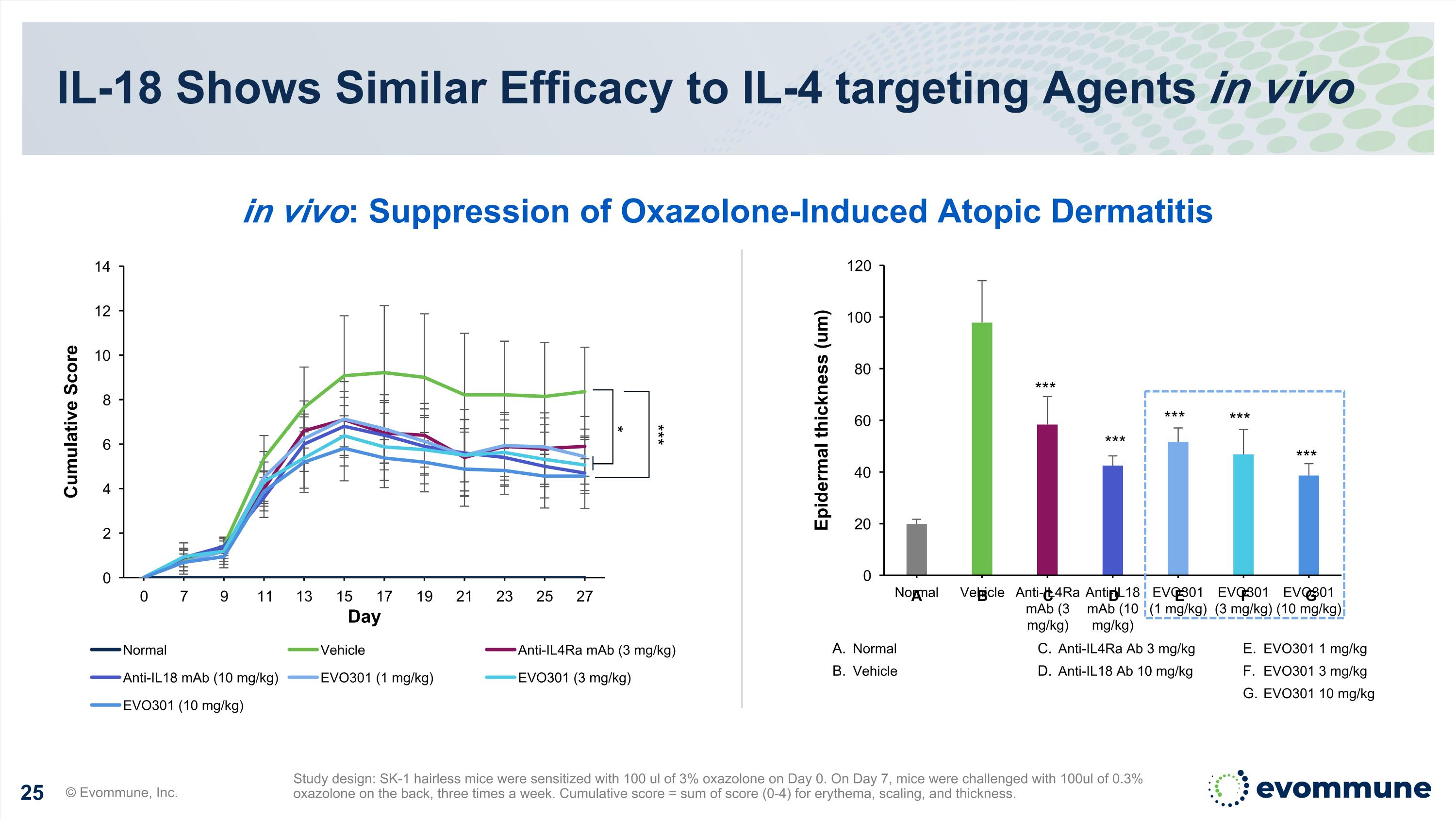
IL-18 Shows Similar Efficacy to IL-4 targeting Agents in vivo Study design: SK-1 hairless mice were sensitized with 100 ul of 3% oxazolone on Day 0. On Day 7, mice were challenged with 100ul of 0.3% oxazolone on the back, three times a week. Cumulative score = sum of score (0-4) for erythema, scaling, and thickness. A B C D E F G *** *** *** *** *** * *** Normal Vehicle Anti-IL4Ra Ab 3 mg/kg Anti-IL18 Ab 10 mg/kg EVO301 1 mg/kg EVO301 3 mg/kg EVO301 10 mg/kg in vivo: Suppression of Oxazolone-Induced Atopic Dermatitis © Evommune, Inc.
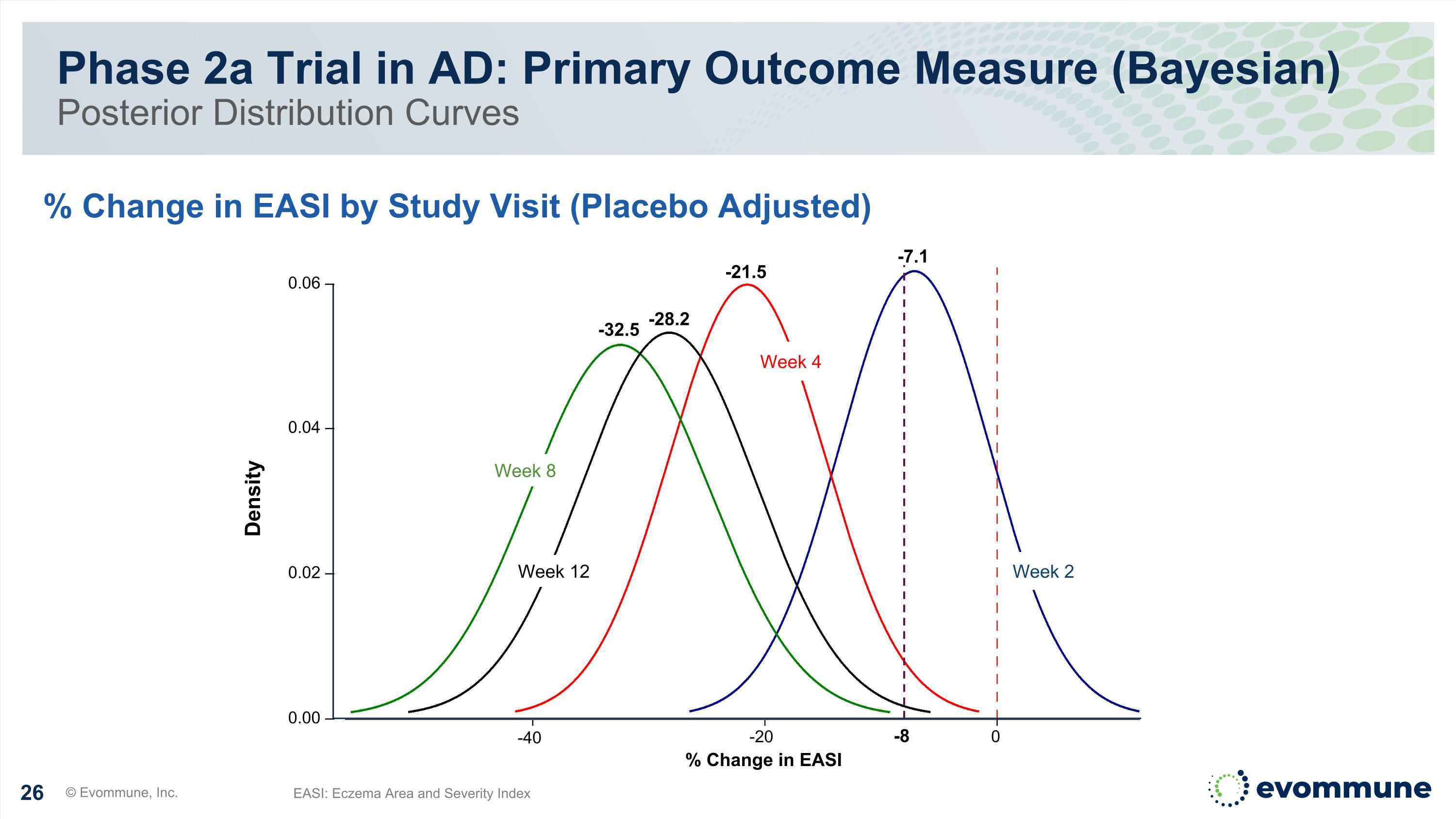
Phase 2a Trial in AD: Primary Outcome Measure (Bayesian) Posterior Distribution Curves © Evommune, Inc. % Change in EASI by Study Visit (Placebo Adjusted) EASI: Eczema Area and Severity Index -8 Week 12 Week 2 Week 4 Week 8 -32.5 -28.2 -21.5 -7.1 % Change in EASI 0 -20 -40 Density 0.06 – 0.04 – 0.02 – 0.00 –
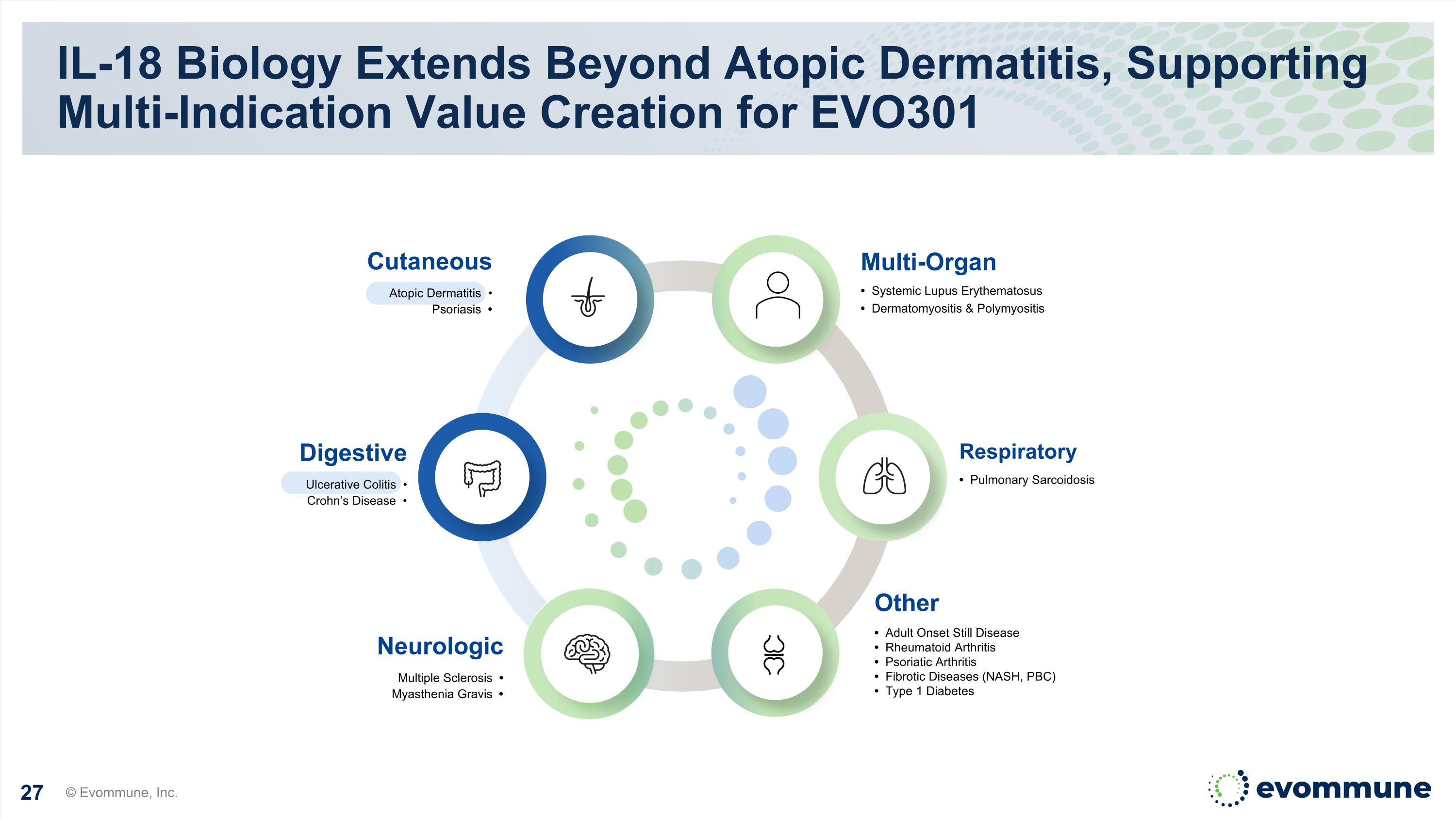
IL-18 Biology Extends Beyond Atopic Dermatitis, Supporting Multi-Indication Value Creation for EVO301 © Evommune, Inc. Neurologic Multiple Sclerosis • Myasthenia Gravis • Multi-Organ • Systemic Lupus Erythematosus • Dermatomyositis & Polymyositis Other • Adult Onset Still Disease • Rheumatoid Arthritis • Psoriatic Arthritis • Fibrotic Diseases (NASH, PBC) • Type 1 Diabetes Respiratory • Pulmonary Sarcoidosis Cutaneous Atopic Dermatitis • Psoriasis • Digestive Ulcerative Colitis • Crohn’s Disease •