8-K: Current report
Published on February 10, 2026
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact Name of Registrant as Specified in Its Charter)
(State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
|
|
|
|
||
(Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant’s Telephone Number, Including Area Code: (
N/A
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
|
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
|
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
|
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Item 7.01 Regulation FD Disclosure.
As reported under Item 8.01 of this Current Report on Form 8-K, on February 10, 2026, Evommune, Inc. (the “Company”) issued a press release titled "Evommune Announces Positive Top-line Data from Phase 2a Proof-of-Concept Trial of EVO301 in Moderate-to-Severe Atopic Dermatitis". A copy of the press release is furnished herewith as Exhibit 99.1 and is incorporated herein by reference.
On February 10, 2026, the Company will host a webcast to discuss the results from the Company’s EVO301 Phase 2a trial in adult patients with moderate-to-severe atopic dermatitis (“AD”). The live webcast presentation will also be available on the “News & Events” page of the Company's website at https://ir.evommune.com/news-events/ir-calendar. A copy of the presentation is being furnished as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated by reference herein. The Company's website and any information contained on the Company's website are not incorporated by reference into this Current Report on Form 8-K. The Company undertakes no obligation to update, supplement or amend the materials attached hereto as Exhibit 99.2.
The information in this Item 7.01, including Exhibits 99.1 and 99.2 hereto, is being furnished and shall not be deemed “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
Item 8.01 Other Events.
On February 10, 2026, the Company announced positive top-line results from its randomized, double-blind, placebo-controlled Phase 2a trial evaluating EVO301. This long-acting fusion protein consisting of an interleukin-18 (“IL-18”) binding protein and an anti-serum albumin Fab-associated domain, achieved highly statistically significant outcomes in adult patients with moderate-to-severe AD. The 70-patient trial was designed to evaluate the safety and efficacy of intravenous dosing of 5mg/kg on day 1 and day 28 (n=48 active, n=22 placebo) over 12 weeks.
Key data highlights include:
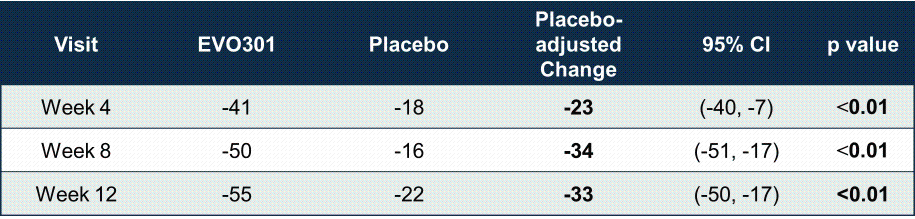
Additionally, 23% of patients treated with EVO301 (vs 0% placebo) achieved vIGA-AD 0/1 (percent of patients achieving a score of 0 or 1 on the validated Investigator’s Global Assessment for Atopic Dermatitis with ≥ 2-point reduction from baseline) at week 12.
1
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
Exhibit Number |
|
Description |
99.1 |
|
|
99.2 |
|
|
104 |
|
Cover Page Interactive Data File (embedded within the Inline XBRL document) |
2
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
Evommune, Inc. |
|
|
|
|
|
Date: February 10, 2026 |
|
By: |
/s/ Luis Peña |
|
|
|
Luis Peña |
|
|
|
President and Chief Executive Officer |
3