S-1: General form for registration of securities under the Securities Act of 1933
Published on April 17, 2026
Table of Contents
As filed with the U.S. Securities and Exchange Commission on April 17, 2026.
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
EVOMMUNE, INC.
(Exact name of registrant as specified in its charter)
| Delaware | 2834 | 85-0742575 | ||
| (State or other jurisdiction of incorporation or organization) |
(Primary Standard Industrial Classification Code Number) |
(I.R.S. Employer Identification Number) |
1891 Page Mill Road
Palo Alto, CA 94304
Telephone: (925) 247-4487
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Luis Peña
President and Chief Executive Officer
Evommune, Inc.
1891 Page Mill Road
Palo Alto, CA 94304
Telephone: (925) 247-4481
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
Eric Blanchard
Minkyu Park
Cooley LLP
500 Boylston Street, 14th Floor
Boston, MA 02116
Tel: (617) 937-2300
Approximate date of commencement of proposed sale to the public: As soon as practicable after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the following box. ☒
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ | |||
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ | |||
| Emerging growth company | ☒ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, as amended, or until the registration statement shall become effective on such date as the Securities and Exchange Commission acting pursuant to said Section 8(a), may determine.
Table of Contents
The information in this preliminary prospectus is not complete and may be changed. Neither we nor the selling stockholders may sell these securities until the registration statement filed with the Securities and Exchange Commission is declared effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting offers to buy these securities in any state or other jurisdiction where the offer or sale is not permitted.
PROSPECTUS (Subject to Completion, dated April 17, 2026)

Up to 4,494,279 Shares of Common Stock
This prospectus relates to the resale from time to time of up to 4,494,279 shares of common stock, par value $0.0001 per share (the “Shares”), of Evommune, Inc. by the selling stockholders listed on page 37 (the “Selling Stockholders”), including their pledgees, assignees, donees, transferees or their respective successors-in-interest.
We will not receive any proceeds from the sale of the Shares offered by this prospectus.
Sales of the Shares by the Selling Stockholders may occur at fixed prices, at market prices prevailing at the time of sale, at prices related to prevailing market prices or at negotiated prices. The Selling Stockholders may sell the Shares to or through underwriters, broker-dealers or agents, who may receive compensation in the form of discounts, concessions or commissions from the Selling Stockholders, the purchasers of the Shares, or both.
We are paying the cost of registering the Shares as well as various related expenses. The Selling Stockholders are responsible for all selling commissions, transfer taxes and other costs related to the offer and sale of their Shares. See “Plan of Distribution” for more information about how the Selling Stockholders may sell or dispose of their Shares.
Our common stock is traded on the New York Stock Exchange (“NYSE”) under the symbol “EVMN.” On April 16, 2026, the last reported sale price of our common stock was $26.77 per share.
We are an “emerging growth company” and a “smaller reporting company” as defined under the federal securities laws and, as such, will be subject to certain reduced public company reporting requirements for this prospectus. See “Prospectus Summary—Implications of Being an Emerging Growth Company and a Smaller Reporting Company.”
Investing in our common stock involves a high degree of risk. You should review carefully the risks and uncertainties described in the section titled “Risk Factors” beginning on page 5 of this prospectus, and under similar headings in any amendments or supplements to this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission or other regulatory body has approved or disapproved of these securities or passed upon the accuracy or adequacy of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2026.
Table of Contents
| ii | ||||
| iii | ||||
| 1 | ||||
| 4 | ||||
| 5 | ||||
| 6 | ||||
| 7 | ||||
| 8 | ||||
| 18 | ||||
| 25 | ||||
| 29 | ||||
| 32 | ||||
| 37 | ||||
| MATERIAL U.S. FEDERAL INCOME TAX CONSIDERATIONS TO NON-U.S. HOLDERS |
41 | |||
| 46 | ||||
| 48 | ||||
| 48 | ||||
| 48 | ||||
| 49 |
You should rely only on the information contained in this prospectus, as well as the information incorporated by reference to exhibits to the registration statement of which this prospectus forms a part and any applicable prospectus supplement or amendment. Neither we nor the Selling Stockholders have authorized anyone to provide you with different information. Neither we nor the Selling Stockholders are making an offer of these securities in any jurisdiction where the offer is not permitted. You should not assume that the information in this prospectus or any applicable prospectus supplement is accurate as of any date other than the date of the applicable document. Since the date of this prospectus and the documents filed as exhibits to the registration statement of which this prospectus forms a part, our business, financial condition, results of operations and prospects may have changed.
i
Table of Contents
This prospectus is part of a registration statement on Form S-1 that we filed with the Securities and Exchange Commission (the “SEC”) using the “shelf” registration process. Under this shelf registration process, the Selling Stockholders may, from time to time, sell the Shares offered by them described in this prospectus.
Neither we nor the Selling Stockholders have authorized anyone to provide you with any information or to make any representations other than those contained or incorporated by reference in this prospectus or any applicable prospectus supplement or any free writing prospectuses prepared by or on behalf of us or to which we have referred you. Neither we nor the Selling Stockholders take responsibility for, or provide any assurance as to the reliability of, any other information that others may give you. Neither we nor the Selling Stockholders will make an offer to sell these securities in any jurisdiction where the offer or sale is not permitted.
We may also provide a prospectus supplement or free writing prospectus to add information to, or update or change information contained or incorporated by reference in, this prospectus. You should read this prospectus, any applicable prospectus supplement or free writing prospectus, including the documents we incorporate herein and therein by reference, together with the additional information to which we refer you in the section of this prospectus entitled “Where You Can Find Additional Information.”
In this prospectus, references to “Evommune,” the “Company,” the “registrant,” “we,” “us,” and “our” refer to Evommune, Inc. The phrase “this prospectus” refers to this prospectus and any applicable prospectus supplement, unless the context requires otherwise.
This prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and you may obtain copies of those documents as described below under “Where You Can Find Additional Information.”
ii
Table of Contents
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus, including the sections entitled “Prospectus Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business,” and any applicable prospectus supplement or free writing prospectus, including the documents that we incorporate by reference herein and therein, contain forward-looking statements concerning our business, operations and financial performance and condition, as well as our plans, objectives and expectations for our business operations and financial performance and condition that are based on our management’s belief and assumptions and on information currently available to our management. Although we believe that the expectations reflected in these forward-looking statements are reasonable, these statements relate to our strategy, future operations, future financial position, future revenue, projected costs, prospects, plans, objectives of management and expected market growth and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. Forward-looking statements in this prospectus include, but are not limited to, statements about:
| • | the success, cost and timing of our product development activities and clinical trials of our most-advanced product candidates, EVO756 and EVO301, and any future product candidates; |
| • | the characteristics and potential advantages of our product candidates; |
| • | our need to raise additional funding before we can expect to generate any revenues from product sales; |
| • | our ability to obtain regulatory approval for our current or future product candidates that we may identify or develop; |
| • | our ability to ensure adequate supply of our current or future product candidates; |
| • | our ability to maintain third-party relationships necessary to conduct our business; |
| • | our heavy dependence upon the success of our research to generate and advance additional product candidates; |
| • | our ability to establish an adequate safety or efficacy profile for our current or future product candidates; |
| • | the implementation of our strategic plans for our business, our current or future product candidates we may develop and our technology; |
| • | our intellectual property position, including the scope of protection and contractual rights that we are able to establish and maintain for intellectual property rights covering our product candidates and technology; |
| • | estimates of the number of patients with certain diseases, market sizes for certain diseases, conditions we intend to treat and the number of subjects that we intend to enroll in our clinical trials; |
| • | our plans relating to the further development and manufacturing of our product candidates, including additional indications for which we may pursue; |
| • | the rate and degree of clinical utility for our current or future product candidates and their acceptance by physicians, patients, third-party payors and others in the medical community; |
| • | our estimates about the size of market opportunities relating to our product candidates; |
| • | our expectations related to our capital requirements and our needs for additional financing; |
| • | our ability to maintain and establish collaborations, licensing or other arrangements, including our ability to comply with our obligations pursuant to the terms of such agreements; |
| • | our financial performance and liquidity; |
| • | our ability to effectively manage our potential growth; |
iii
Table of Contents
| • | developments relating to our competitors and our industry, including the impact of government regulation and policy; |
| • | our ability to retain the continued service of our key professionals and consultants and to identify, hire and retain additional qualified professionals; and |
| • | our ability to maintain adequate internal controls over financial reporting and to manage our business in accordance with applicable laws and the highly regulated industry in which we participate. |
In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “expects,” “intends,” “plans,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue” or the negative of these terms or other comparable terminology. These statements are only predictions. You should not place undue reliance on forward-looking statements because they involve known and unknown risks, uncertainties and other factors, which are, in some cases, beyond our control and which could materially affect results. Factors that may cause actual results to differ materially from current expectations include, among other things, those listed in the section entitled “Risk Factors” and contained or incorporated by reference in this prospectus. If one or more of these risks or uncertainties occur, or if our underlying assumptions prove to be incorrect, actual events or results may vary significantly from those implied or projected by the forward-looking statements. No forward-looking statement is a guarantee of future performance. You should read this prospectus, any applicable prospectus supplement, together with the documents that we reference in this prospectus and have filed with the SEC that are incorporated by reference and any free writing prospectus we have authorized for use, completely and with the understanding that our actual future results may be materially different from any future results expressed or implied by these forward-looking statements.
The forward-looking statements in this prospectus represent our views as of the date of this prospectus. We anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements at some point in the future, we assume no obligation to update or revise any forward-looking statements except to the extent required by applicable law. You should, therefore, not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this prospectus.
In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this prospectus and, while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and you are cautioned not to rely unduly upon these statements.
iv
Table of Contents
This summary highlights information contained elsewhere in this prospectus and in the documents we incorporate herein by reference, and does not contain all of the information that you should consider in making your investment decision. Before investing in our securities, you should carefully read this entire prospectus, along with our financial statements and the related notes thereto and the information set forth in the sections titled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” included in or incorporated herein by reference. Unless the context otherwise requires, we use the terms “Evommune,” “Company,” “we,” “us” and “our” in this prospectus to refer to Evommune, Inc.
Overview
Evommune is a clinical-stage biotechnology company developing innovative therapies that target key drivers of chronic inflammatory diseases, with initial clinical development programs focusing on chronic spontaneous urticaria (“CSU”), atopic dermatitis (“AD”) and ulcerative colitis (“UC”). Chronic inflammation is a significant healthcare problem in the world, substantially impacting patients’ quality of life and leading to life-threatening conditions. These conditions, if not prevented, ultimately lead to fatal diseases, such as cardiovascular diseases, diabetes and cancer, which contribute to three out of every five deaths worldwide and result in an estimated $90 billion of annual cost to the healthcare system in the United States.
Our mission is to improve patients’ daily lives and prevent the long-term effects of uncontrolled inflammation that are a consequence of the limitations of existing therapies. To achieve this, we are advancing a portfolio of differentiated product candidates that target key drivers of chronic inflammation.
Our management team’s proven drug development expertise and experience in the field of immunology and inflammation, combined with advanced scientific tools, enable us to identify and advance potent, highly selective molecules with distinctive mechanisms of action. By identifying treatment gaps of chronic inflammatory diseases, we strive to transform the treatment landscape, developing therapies that have the potential to offer rapid symptom relief and provide safe, durable resolution of the underlying disease. Among our portfolio of programs, we currently have two product candidates, EVO756 and EVO301, in Phase 2 development. We are initially developing EVO756 for the treatment of CSU, AD and migraine, and EVO301 for the treatment of AD and UC. We see broad expansion potential for both programs across additional chronic inflammatory diseases. We also intend to advance additional preclinical programs into clinical development.
Our most advanced clinical-stage product candidate, EVO756, is a potent and highly selective oral small molecule antagonist of MRGPRX2, a receptor predominantly found on mast cells and peripheral sensory neurons. Dysregulated MRGPRX2 activity can play a key role as both a catalyst and perpetuator of disease pathogenesis across a multitude of systemic chronic inflammatory diseases. By targeting MRGPRX2, we believe EVO756 is the only dual mechanism clinical approach that modulates both mast cells and peripheral sensory neurons, representing a new potential therapeutic option to reduce inflammation and provide rapid relief of itch (pruritus). Mast cells are critical regulators of immune response and can be found in most vascularized tissues including skin, lung and the digestive tract. These cells tend to be distributed in close proximity to peripheral sensory neurons, where the activation of the neurons can trigger the inflammatory cascade and mast cells are implicated in further perpetuating neuroinflammation and its related symptoms. We believe MRGPRX2 is the only clinical approach aimed at inhibiting this neuroimmune interaction.
1
Table of Contents
Our current pipeline is summarized below:
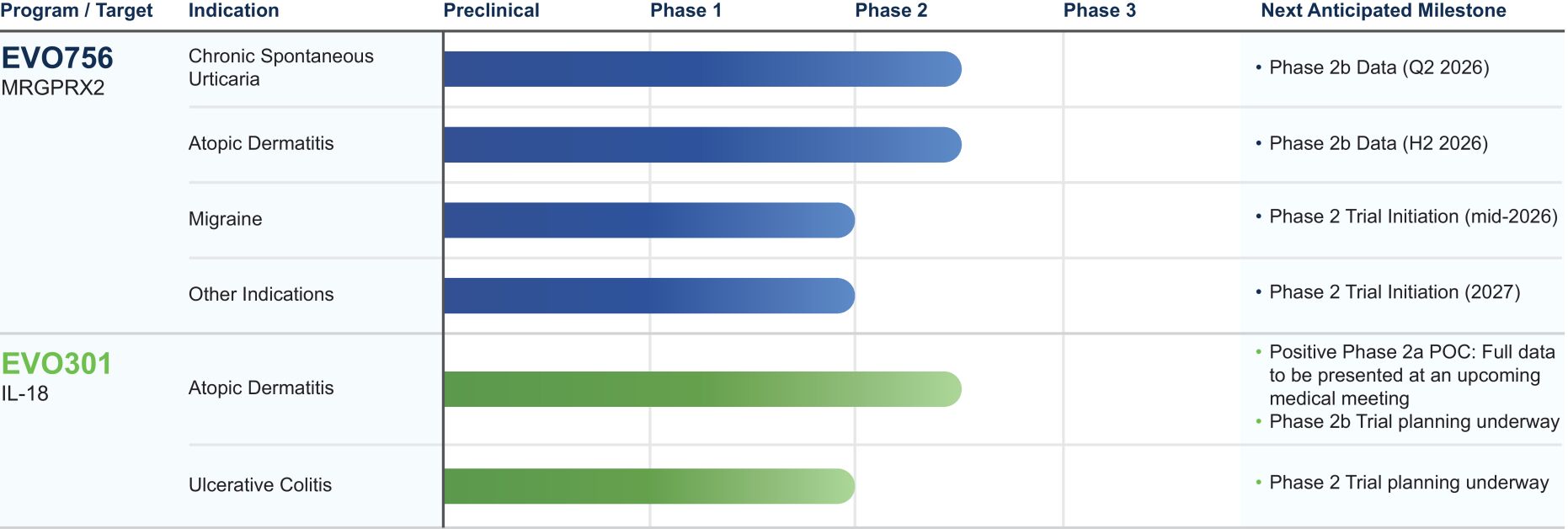
Notes: (1) Other potential indications for EVO756 include asthma, interstitial cystitis, irritable bowel syndrome and pruritus. To date, based on our data from the successful completion of our Phase 1 proof-of-concept trial of EVO756 in healthy volunteers, we believe there is a path to proceed to Phase 2 clinical development for these other indications, similar to our initiation of our Phase 2b trial in AD, subject to standard regulatory requirements. (2) We are currently prioritizing development in other indications ahead of CIndU and may conduct additional CIndU trials in the future.
Private Placement
On February 12, 2026, we entered into a Securities Purchase Agreement (the “Purchase Agreement”) with the Selling Stockholders pursuant to which we agreed to issue and sell to the Selling Stockholders an aggregate of 4,494,279 Shares (the “Private Placement”). Each Share was offered and sold at a purchase price of $27.88 before deducting placement agent fees.
In connection with the Private Placement, we entered into a Registration Rights Agreement, dated February 12, 2026 (the “Registration Rights Agreement”) with the Selling Stockholders, pursuant to which we are obligated to prepare and file with the SEC a registration statement to register for resale of the Shares within 60 days of the closing date of the Private Placement and to use our reasonable best efforts to have the registration statement declared effective as soon as possible, but no later than 90 days after the initial filing date of the registration statement, subject to extension under the terms of the Registration Rights Agreement. We also agreed to use reasonable best efforts to keep such registration statement effective until the earlier of the date the Shares covered by such registration statement have been sold or may be resold pursuant to Rule 144 without restriction.
Corporate Information and Trademarks
Evommune, Inc. was incorporated under the laws of the State of Delaware in April 2020. Our principal executive office is located at 1891 Page Mill Road, Palo Alto, CA 94304. Our telephone number is (925) 247-4481. Our website address is https://www.evommune.com. Information contained in, or accessible through, our website does not constitute a part of, and is not incorporated into, this prospectus.
The Evommune logo, the name Evommune, and other trademarks of Evommune, Inc. appearing in this prospectus are the property of Evommune, Inc. Solely for convenience, trade names, trademarks, and service marks contained in this prospectus may appear without the “®” or “™” symbols. Such references are not intended to indicate, in any way, that the respective owners will not assert, to the fullest extent possible under applicable law, their rights to those trade names, trademarks, and service marks.
2
Table of Contents
Implications of Being an Emerging Growth Company and a Smaller Reporting Company
We are an “emerging growth company” as defined in the Jumpstart Our Business Startups Act (“JOBS Act”) and we may remain an emerging growth company for up to five years following our initial public offering (“IPO”). For so long as we remain an emerging growth company, we are permitted and intend to rely on certain exemptions from various public company reporting requirements, including not being required to have our internal control over financial reporting audited by our independent registered public accounting firm pursuant to Section 404(b) of the Sarbanes-Oxley Act of 2002, as amended (“Sarbanes-Oxley Act”), reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and any golden parachute payments not previously approved.
In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This provision allows an emerging growth company to delay the adoption of some accounting standards until those standards would otherwise apply to private companies. We have elected to take advantage of the benefits of this extended transition period and, therefore, we are not subject to the same requirements to adopt new or revised accounting standards as other public companies that are not emerging growth companies; however, we may adopt certain new or revised accounting standards early. We would cease to be an emerging growth company upon the earliest to occur of: (i) the last day of the fiscal year in which we have $1.235 billion or more in annual revenue; (ii) the date on which we first qualify as a large accelerated filer under the rules of the SEC; (iii) the date on which we have, in any three-year period, issued more than $1.0 billion in non-convertible debt securities; and (iv) the last day of the fiscal year ending after the fifth anniversary of our initial public offering.
We are also a “smaller reporting company” as defined in the Securities Exchange Act of 1934, as amended (the “Exchange Act”). We may continue to be a smaller reporting company even after we are no longer an emerging growth company. We may take advantage of certain of the scaled disclosures available to smaller reporting companies and will be able to take advantage of these scaled disclosures for so long as the market value of our voting and non-voting common stock held by non-affiliates is less than $250.0 million measured on the last business day of our second fiscal quarter, or our annual revenue is less than $100.0 million during the most recently completed fiscal year, and the market value of our voting and non-voting common stock held by non-affiliates is less than $700.0 million measured on the last business day of our second fiscal quarter.
As a result, the information in this prospectus and that we provide to our investors in the future may be different than what you might receive from other public reporting companies.
3
Table of Contents
| Common stock offered by the Selling Stockholders |
Up to 4,494,279 shares |
| Use of proceeds |
We will not receive any proceeds from the sale of the Shares covered by this prospectus. |
| Risk factors |
Before investing in our securities, you should carefully read and consider the information set forth in “Risk Factors” beginning on page 5. |
| NYSE trading symbol |
“EVMN” |
For additional information concerning the offering, see “Plan of Distribution” beginning on page 46.
4
Table of Contents
Investing in our common stock involves a high degree of risk. You should carefully consider the risks and uncertainties described under the heading “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2025, which is incorporated by reference into this prospectus in its entirety, as well as any amendment or updates to our risk factors reflected in subsequent filings with the SEC, including any prospectus supplement hereto or any related free writing prospectus, before deciding to invest in our common stock. If any of the events or developments described therein were to occur, our business, prospects, operating results and financial condition could suffer materially, the trading price of our common stock could decline, and you could lose all or part of your investment. The risks and uncertainties described therein are not the only ones we face. Additional risks and uncertainties not presently known to us or that we currently believe to be immaterial may also adversely affect our business.
5
Table of Contents
We are filing the registration statement of which this prospectus forms a part to permit the Selling Stockholders to resell the Shares, as described in the section below titled “Selling Stockholder.”
The Selling Stockholders will receive all of the net proceeds from any sales of the Shares sold pursuant to this prospectus. We will not receive any proceeds from the resale of any Shares offered by this prospectus by the Selling Stockholders.
We, and not the Selling Stockholders, will pay the costs, expenses and fees in connection with the registration of the Shares covered by this prospectus, but the Selling Stockholders will pay all discounts, commissions or brokers’ fees or fees of similar securities industry professionals and transfer taxes, if any, attributable to sales of the Shares.
6
Table of Contents
We have never declared or paid any cash dividends on our capital stock. We do not anticipate paying any dividends on our capital stock in the foreseeable future. We currently intend to retain all available funds and any future earnings to fund the development and growth of our business. Any future determination to declare dividends will be subject to the discretion of our board of directors and will depend on various factors, including applicable laws, our results of operations, financial condition, future prospects and any other factors deemed relevant by our board of directors. Investors should not purchase our common stock with the expectation of receiving cash dividends.
In addition, our ability to pay cash dividends on our capital stock in the future may be limited by the terms of any future debt or preferred securities we issue or any credit facilities we enter into.
7
Table of Contents
Executive Officers and Directors
The following table sets forth certain information about our executive officers and directors, including their ages, as of March 31, 2026.
| Name |
Age | Position(s) | ||||
| Executive Officers: |
||||||
| Luis Peña(3) | 63 | President & Chief Executive Officer and Director | ||||
| Kyle Carver, M.B.A., C.P.A. | 38 | Chief Financial Officer | ||||
| Eugene A. Bauer, M.D.(4) | 83 | Chief Medical Officer and Director | ||||
| Gregory S. Moss, Esq. | 42 | Chief Business and Legal Officer, Corporate Secretary and Chief Compliance Officer | ||||
| Jeegar Patel, Ph.D. | 48 | Chief Scientific Officer | ||||
| Janice Drew, M.P.H. | 63 | Executive Vice President, Operations | ||||
| Non-Employee Directors: | ||||||
| Benjamin F. McGraw, III, Pharma. D.(1)(2)(3)(4) |
76 | Director, Chairman of the Board | ||||
| David E. Cohen, M.D., M.P.H.(2)(3)(4) | 60 | Director | ||||
| Derek DiRocco, Ph.D.(4) | 45 | Director | ||||
| Rob Hopfner, R.Ph., Ph.D.(1)(4) | 52 | Director | ||||
| Felice Verduyn-van Weegen(3)(4) | 38 | Director | ||||
| Arthur Kirsch(1)(2) | 73 | Director | ||||
| (1) | Member of the Audit Committee (as defined below) |
| (2) | Member of the Compensation Committee (as defined below) |
| (3) | Member of the Nominating and Corporate Governance Committee (as defined below) |
| (4) | Member of the Science and Technology Committee (as defined below) |
Executive Officers
Luis Peña has served as our President and Chief Executive Officer and as a member of our board of directors since inception in April 2020. Prior to joining us, Mr. Peña co-founded Dermira and served as its Chief Development Officer, where he headed the R&D group from inception through to Dermira’s 2020 sale to Eli Lilly & Company (“Lilly”) for $1.1 billion. Before Dermira, Mr. Peña served as Vice President, Head of Global Prescription Development, at Stiefel, a GSK company, and was Senior Vice President, Portfolio Planning and Management, at Stiefel Laboratories Inc. prior to its acquisition by GSK plc. He also held leadership roles in portfolio management and product development at Connetics and Theravance, as well as various leadership positions in manufacturing and research and development at Genentech. Mr. Peña holds a B.S. in biochemistry from San Francisco State University. We believe Mr. Peña is qualified to serve on our board of directors because of his extensive experience in executive leadership and drug development roles in the biopharmaceutical industry.
Kyle Carver, M.B.A., C.P.A. has served as our Chief Financial Officer since March 2022. Prior to joining us, Mr. Carver served as Chief Accounting Officer at Kadmon Holdings, Inc., a Sanofi Company (“Kadmon”), where he oversaw all aspects of financial and accounting operations, including Kadmon’s transition to a public company in its 2016 initial public offering through the approval and commercialization of Rezurock (belumosudil) in 2021. Mr. Carver drove significant improvement in accounting operations and internal controls, helped raise more than
8
Table of Contents
$500 million through follow-on and convertible note offerings and contributed to deal-making activities, including the acquisition of Kadmon by Sanofi for $1.9 billion in November 2021. Prior to Kadmon, Mr. Carver held a senior role at KPMG, serving a broad base of life sciences clients. Mr. Carver earned an M.B.A. from the Stern School of Business at New York University and a B.S. in accounting from Villanova University. Mr. Carver is a Certified Public Accountant.
Eugene A. Bauer, M.D. has served as our Chief Medical Officer and as a member of our board of directors since inception in April 2020. Prior to joining us, Dr. Bauer was Founder, Chief Medical Officer and a member of the board of directors of Dermira from 2010 to 2020. Prior to Dermira, Dr. Bauer was President and Chief Medical Officer of Peplin, a position he held from 2008 through 2009 when Peplin was acquired by LEO Pharma. Peplin focused on an innovative product (Picato) that was approved by the FDA for use in the treatment of actinic keratosis. Prior to Peplin, he was CEO of Neosil, a development stage dermatology pharmaceutical company acquired by Peplin in 2008. Dr. Bauer is Professor Emeritus in the School of Medicine at Stanford University and past Vice President for the Medical Affairs (1997-2001) and Dean of the School of Medicine (1995-2001) of Stanford University. Dr. Bauer received his M.D. from Northwestern University. We believe Dr. Bauer is qualified to serve on our board of directors because of his extensive experience managing biopharmaceutical and life science companies and his vast medical background.
Gregory S. Moss, Esq. has served as our Chief Business and Legal Officer, Corporate Secretary and Chief Compliance Officer since June 2022. Prior to joining us, Mr. Moss served as Executive Vice President, General Counsel and Corporate Secretary, Chief Compliance Officer at Kadmon, where he led legal, compliance and business development operations, culminating in Kadmon’s successful $1.9 billion acquisition in 2021. Prior to joining Kadmon in 2012, Mr. Moss served as a solicitor in the corporate risk department of a large Australian law firm and as an associate at a boutique law firm and hedge fund in New York, where he focused on complex litigation and event-driven outcomes. Mr. Moss has been serving on the board of Anteris Technologies Global Corp. since June 2025, and currently serves on the board of Vitls, Inc. Mr. Moss earned a B.A. and an L.L.B. from Macquarie University, Australia, and is a member of the Bar Associations of New York, and New South Wales, Australia, with admissions before the Supreme Court of the United States of America; Southern District of New York; Supreme Court of New South Wales, Australia; and High Court of Australia.
Jeegar Patel, Ph.D. has served as our Chief Scientific Officer since October 2022. Prior to joining us, Dr. Patel served as Senior Vice President, Research and Nonclinical Development at Kadmon, where he led the research and development organization with a therapeutic focus in fibrosis, immunology and immuno-oncology. He has directly contributed to multiple IND and NDA filings across therapeutic areas, including the development and approval of Rezurock. The FDA approval of Rezurock in July 2021 led to Sanofi’s $1.9 billion acquisition of Kadmon in late 2021, with Dr. Patel leading Kadmon’s R&D activity integration into Sanofi. Prior to joining Kadmon in 2014, Dr. Patel was a Nonclinical Development Project Leader at ImClone Systems (acquired by Lilly in 2008) and led nonclinical development activities resulting in the approval of Cyramza (aVEGFR2 Mab), Portrazza (aEGFR Mab) and Lartruvo (aPDGFb Mab). Dr. Patel began his career as a Nonclinical Development Project Toxicologist at Abbott Laboratories and Hoechst Marion Roussel. Dr. Patel received his B.S. in pharmacology and toxicology from the University of the Sciences in Philadelphia and his Ph.D. in pharmacology and physiology from Drexel University College of Medicine.
Janice Drew, M.P.H. has served as our Executive Vice President, Operations since July 2020. Prior to joining us, Ms. Drew served as Senior Vice President, Portfolio Planning and Management at Dermira where she supported clinical strategy, clinical program development and protocol development for topical and biologic products targeting acne, rosacea, psoriasis, hyperhidrosis and atopic dermatitis. While at Dermira, Ms. Drew supported Phase 3 development and the BLA for Cimziane for plaque psoriasis, the NDA for Qbrexza for axillary hyperhidrosis and the global Phase 3 program for lebrikizumab for atopic dermatitis. Ms. Drew has had direct responsibility for five NDAs/BLAs, multiple INDs and end-of-Phase 2 and pre-NDA/BLA meetings, including oversight for four global clinical development programs. Prior to joining Dermira in 2011, Ms. Drew was at Peplin, where she led the Phase 3 program, NDA and global regulatory submissions for Picato, a topical
9
Table of Contents
treatment for actinic keratosis. Prior to Peplin, Ms. Drew worked at Allergan, building the Phase 3 development team for Botox® for the treatment of neurogenic and idiopathic overactive bladder. Ms. Drew also spent eight years at Roche in Palo Alto, California and Basel, Switzerland, leading the global clinical operations team working on CellCept® and Zenapax®, both indicated for solid organ transplantation. Ms. Drew received her B.S. from the University of California, Irvine, and a M.P.H. from California State University, Fullerton.
Non-Employee Directors
Benjamin F. McGraw, III, Pharma. D. has served as Chairman of our board of directors since October 2020. Dr. McGraw is the Executive Chairman of Perfuse Therapeutics, a clinical-stage biopharmaceutical company committed to reducing the incidence of blindness. He is also a director at TheraVida, a private pharmaceutical company focused on therapies for dermatologic diseases. Dr. McGraw has served as Executive Chairman of Auration Biotech, a private biotechnology company focused on a regenerative protein and other therapies for ear, nose and throat diseases since 2014. Dr. McGraw is the Lead Independent Director for the board of directors of Aerie Pharmaceuticals, a public pharmaceutical company focused on new treatments for glaucoma and other diseases of the front and back of the eye. He is also Executive Chairman of Trefoil Therapeutics, a private biopharmaceutical company focused on developing an engineered protein as a regenerative treatment for corneal endothelial dystrophies and epithelial disorders. Previously, Dr. McGraw served as the Managing Member of Long Shadows Asset Management, LLC, an investment advisory company; Chairman, President and CEO of Valentis, a public gene therapy company; Vice President of Corporate Development at Allergan, a public pharmaceutical company; and Vice President of Development at Marion Laboratories and Marion Merrell Dow, both public pharmaceutical companies. Dr. McGraw received his B.S. and his Pharm.D. from the University of Tennessee Health Science Center, where he also completed a clinical practice residency. We believe Dr. McGraw is qualified to serve on our board of directors because of his distinguished scientific background, experience in leadership roles in the biopharmaceutical industry as well as his service on the boards of directors of numerous companies.
David E. Cohen, M.D., M.P.H. has served as a member of our board of directors since May 2020. Dr. Cohen has held various positions at the New York University School of Medicine since 1994, including Charles C. and Dorothea E. Harris Professor of Dermatology, Vice Chairman of Clinical Affairs, Chief of Allergy and Contact Dermatitis and Director of Occupational and Environmental Dermatology. He has served as a Lecturer of Environmental Sciences at the Columbia University School of Public Health. He has been an Attending Physician at the Ronald O. Perelman Department of Dermatology at the Tisch Hospital at New York University Medical Center and Bellevue Hospital Center. Dr. Cohen has served as a clinical consultant to numerous companies and on the boards and committees of many professional organizations, including President of the American Dermatological Association, American Contact Dermatitis Society, Dermatology Section of the New York Academy of Medicine. He is also a founding board member of the American Acne and Rosacea Society and served on several committees of the American Academy of Dermatology and the American College of Allergy, Asthma and Immunology. He is also a member of the editorial board of the Journal of Drugs in Dermatology and the editorial advisory boards of Dermatitis and Skin and Allergy News. He also served on the boards of directors of Timber, Connetics, Vyteris, Kadmon and Dermira. Dr. Cohen earned a B.S. in biomedical science from the City University of New York, an M.D. from Stony Brook School of Medicine at SUNY and an M.P.H. in environmental science from Columbia University School of Public Health. We believe Dr. Cohen is qualified to serve on our board of directors because of his education and experience in the life sciences and dermatology fields, as well as his service on the boards of directors of numerous companies.
Derek DiRocco, Ph.D. has served as a member of our board of directors since October 2024. Dr. DiRocco has been a partner at RA Capital Management since December 2020. Dr. DiRocco works on both public and private investments and serves as a board director for 89Bio, Mineralys, Werewolf Therapeutics, Acrivon Therapeutics, Rivus Pharmaceuticals, Forward Therapeutics and Sera Medicines. Dr. DiRocco joined RA Capital Management in July 2013 and initially covered solid tumor oncology landscapes, later broadening coverage to multiple therapeutic areas. Dr. DiRocco holds a B.A. in Biology from Holy Cross College and a Ph.D. in
10
Table of Contents
Pharmacology from the University of Washington. He conducted his postdoctoral research at Brigham and Women’s Hospital/Harvard Medical School, where he researched signaling pathways responsible for chronic and acute kidney disease, as well as cellular mechanisms underlying fibrotic disease. We believe Dr. DiRocco is qualified to serve on our board of directors because of his education and experience in the life sciences industry, venture capital experience, as well as his service on the boards of directors of numerous companies.
Rob Hopfner, R.Ph., Ph.D. has served as a member of our board of directors since July 2020. Dr. Hopfner has been a Managing Partner at Pivotal bioVenture Partners since October 2017, where he focuses on drug discovery and development investments. Dr. Hopfner has a long track record of working successfully with entrepreneurs to progress novel, important medicines through development and onto the market. Prior to joining Pivotal in 2017, Dr. Hopfner was previously a Managing Director at Bay City Capital and prior to that worked in business development and investment roles at DuPont Pharmaceuticals and at Ag-West Biotech. Dr. Hopfner holds a Ph.D. in pharmacology from the University of Saskatchewan and an M.B.A. from the University of Chicago Booth School of Business. Dr. Hopfner completed his postdoctoral work at Harvard Medical School and started his career as a pharmacist. We believe Dr. Hopfner is qualified to serve on our board of directors because of his education and experience in drug development and venture capital experience.
Felice Verduyn-van Weegen has served as a member of our board of directors since September 2021. Ms. Weegen currently serves as a Partner at EQT. Ms. Weegen worked for LSP from 2015 until March 2022, when LSP joined forces with EQT and became EQT Life Sciences. Prior to joining LSP, Ms. Weegen worked as a Consultant at McKinsey & Company in Amsterdam. Prior to working at McKinsey, Ms. Weegen was a Neuroscientist and Statistical Geneticist, working with the prestigious Complex-Traits Genetics Group at the Broad Institute and Harvard Medical School in Cambridge, Massachusetts. Ms. Weegen holds an M.S. (cum laude) in neuroscience from Vrije Universiteit Amsterdam and an M.B.A. (with distinction) from Columbia Business School in New York. We believe Ms. Weegen is qualified to serve on our board of directors because of her education and experience in the life sciences industry and venture capital experience.
Arthur Kirsch has served as a member of our board of directors since November 2025. Mr. Kirsch currently serves as a board member of Liquidia Corporation. Mr. Kirsch has served as a director on the board and as a member of the audit and compensation committees for Atea Pharmaceuticals, Inc. (Nasdaq: AVIR) since February 2025. Mr. Kirsch previously served as a director of Anavasi Diagnostics, a private company, from August 2022 through June 2023, as a director of POZEN Inc. (Nasdaq: POZN) from May 2004 until May 2015, as a director of Aralez Pharmaceuticals, Inc. (Nasdaq: ARLZ) from February 2016 until May 2019, as a director of Kadmon Corporation (NYSE: KDMN) from May 2019 to November 2021 and as a director of Immunomedics, Inc. (Nasdaq: IMMU) from August 2015 until October 2016. Mr. Kirsch has served as a consultant at Alvarez & Marsal since 2019. From June 2005 until June 2019, Mr. Kirsch served as a managing director and senior advisor for GCA Global, LLC, a global investment banking firm. From May 1994 to May 2004, he served as executive vice president, head of research at Vector Securities, LLC, a brokerage firm. From February 1990 to May 1993, Mr. Kirsch served as president of Natwest Securities Limited, a brokerage firm. From June 1979 to February 1990, Mr. Kirsch worked at Drexel Burnham Lambert, Inc., an investment banking firm, where he held the position of executive vice president, head of equity division. Mr. Kirsch graduated from the University of Rhode Island with a Bachelor of Science and also holds a Master of Business Administration from Baruch College. We believe Mr. Kirsch is qualified to serve on our board of directors because of his business and financial expertise and his experience serving on the boards of directors of several public pharmaceutical and life sciences companies.
Family Relationships
There are no family relationships among any of our executive officers or directors.
11
Table of Contents
Composition of Our Board of Directors
The authorized number of our board of directors is set at eight and currently consists of eight members with no vacancy. Our nominating and corporate governance committee and our board of directors may consider a broad range of factors relating to the qualifications and background of nominees. Our nominating and corporate governance committee’s and our board of directors’ priority in selecting board members is to identify persons who will further the interests of our stockholders through his or her established record of professional accomplishments, the ability to contribute positively to the collaborative culture among board members, knowledge of our business, understanding of the competitive landscape, diversity of background and perspective and professional and personal experiences and expertise relevant to our growth strategy. Our directors hold office until their successors have been elected and qualified or until the earlier of their resignation or removal. Our amended and restated certificate of incorporation and amended and restated bylaws also provide that our directors may be removed only for cause by the affirmative vote of the holders of at least a majority of the votes that all our stockholders would be entitled to cast in an annual election of directors and that any vacancy on our board of directors, including a vacancy resulting from an enlargement of our board of directors, may be filled only by vote of a majority of our directors then in office.
Director Independence
Our common stock is traded on the NYSE. Applicable NYSE rules require a majority of a listed company’s board of directors consist of independent directors within one year of listing. In addition, the NYSE rules require that (i) on the date of the initial listing, at least one member of each of a listed company’s audit, compensation and nominating and corporate governance committees be independent, (ii) within 90 days of the date of the initial listing, a majority of the members of such committees be independent and (iii) within one year of the date of the initial listing, all the members of such committees be independent. Audit committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Exchange Act. Under applicable NYSE rules, a director will only qualify as an “independent director” if, in the opinion of the listed company’s board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director.
In order to be considered independent for purposes of Rule 10A-3, a member of an audit committee of a listed company may not, other than in his or her capacity as a member of the audit committee, the board of directors or any other board committee, accept, directly or indirectly, any consulting, advisory or other compensatory fee from the listed company or any of its subsidiaries or otherwise be an affiliated person of the listed company or any of its subsidiaries.
Our board of directors has determined that a majority of the members of the board of directors are independent directors, including for purposes of the rules of the NYSE and the SEC. In making such independence determination, our board of directors considered the relationships that each non-employee director has with us and all other facts and circumstances that our board of directors deemed relevant in determining his or her independence, including the beneficial ownership of our capital stock by each non-employee director. In considering the independence of the directors listed above, our board of directors considered the association of our directors with the holders of more than 5% of our common stock. The composition and function of our board of directors and each of our committees comply with all applicable requirements of the NYSE and the rules and regulations of the SEC.
Staggered Board
In accordance with the terms of our amended and restated certificate of incorporation and amended and restated bylaws, our board of directors will be divided into three staggered classes of directors and each will be assigned to one of the three classes. At each annual meeting of the stockholders, a class of directors will be elected for a three-year term to succeed the directors of the same class whose terms are then expiring. The terms
12
Table of Contents
of the directors will expire upon the election and qualification of successor directors at the annual meeting of stockholders to be held during the years 2026 for Class I directors, 2027 for Class II directors and 2028 for Class III directors.
| • | Our Class I directors are Luis Peña and Eugene A. Bauer, M.D.; |
| • | Our Class II directors are Rob Hopfner, R.Ph., Ph.D., Derek DiRocco, Ph.D. and Felice Verduyn-van Weegen; and |
| • | Our Class III directors are Benjamin F. McGraw, III, Pharma. D., David E. Cohen, M.D., M.P.H. and Arthur Kirsch |
Our amended and restated certificate of incorporation and amended and restated bylaws provide that the number of directors shall be fixed from time to time by a resolution of the majority of our board of directors.
The division of our board of directors into three classes with staggered three-year terms may delay or prevent stockholder efforts to effect a change of our board of directors or a change in control.
Board Leadership Structure and Board’s Role in Risk Oversight
The roles of lead director or chairman and the chief executive officer (“CEO”) are separated. Benjamin F. McGraw, III, Pharma. D. is our current chairman of the board of directors and Luis Peña is our current CEO. We believe that separating these positions allows our CEO to focus on setting our overall strategic direction, expanding the organization to deliver on our strategy and overseeing our day-to-day business, while allowing a lead director of the board to lead the board of directors in its fundamental role of providing strategic advice. Our board of directors recognizes the time, effort and energy that the CEO is required to devote to his position in the current business environment, as well as the commitment required to serve as our chairman, particularly as the board of directors’ oversight responsibilities continue to grow. While our amended and restated bylaws and corporate governance guidelines do not require that our lead director and CEO positions be separate, our board of directors believes that having separate positions is the appropriate leadership structure for us at this time and demonstrates our commitment to good corporate governance.
Risk is inherent with every business, and how well a business manages risk can ultimately determine its success. We face a number of risks, including risks relating to our financial condition, development and commercialization activities, operations, strategic direction and intellectual property as more fully discussed in the section entitled “Risk Factors” appearing elsewhere in this prospectus. Management is responsible for the day-to-day management of risks we face, while our board of directors, as a whole and through its committees, has responsibility for the oversight of risk management. In its risk oversight role, our board of directors has the responsibility to satisfy itself that the risk management processes designed and implemented by management are adequate and functioning as designed.
The role of the board of directors in overseeing the management of our risks is conducted through our board of directors, including through its committees, as disclosed in the descriptions of each of the committees below and in the charters of each of the committees. The full board of directors (or the appropriate board committee in the case of risks that are under the purview of a particular committee) discusses with management our major risk exposures, their potential impact on us and the steps we take to manage them. When a board committee is responsible for evaluating and overseeing the management of a particular risk or risks, the chairperson of the relevant committee reports on the discussion to the full board of directors during the committee reports portion of the next board meeting. This enables the board of directors and its committees to coordinate the risk oversight role, particularly with respect to risk interrelationships.
Committees of Our Board of Directors
Our board of directors has established an audit committee, a compensation committee and a nominating and corporate governance committee, each of which will operate pursuant to a charter adopted by our board of
13
Table of Contents
directors. The composition and functioning of all of our committees comply with all applicable requirements of the Sarbanes-Oxley Act and with NYSE and SEC rules and regulations. Each committee’s charter is posted on the investor relations section of our website, which is located at www.evommune.com. The inclusion of our website address in this prospectus does not incorporate by reference the information on or accessible through our website into this prospectus.
Audit Committee
Dr. McGraw, Dr. Hopfner and Mr. Kirsch currently serve on the audit committee, which is chaired by Mr. Kirsch. Our board of directors has determined that each of Dr. McGraw, Dr. Hopfner and Mr. Kirsch are “independent” for audit committee purposes as that term is defined in the rules of the SEC and the applicable NYSE rules and each has sufficient knowledge in financial and auditing matters to serve on the audit committee. Our board of directors has designated each of Dr. McGraw and Mr. Kirsch as an “audit committee financial expert,” as defined under the applicable NYSE rules. The audit committee’s responsibilities include:
| • | appointing, approving the compensation of and assessing the independence of our independent registered public accounting firm; |
| • | pre-approving auditing and permissible non-audit services and the terms of such services, to be provided by our independent registered public accounting firm; |
| • | reviewing and discussing with management and our independent registered public accounting firm our annual and quarterly financial statements and related disclosures as well as critical accounting policies and practices used by us; |
| • | coordinating the oversight and reviewing the adequacy of our internal control over financial reporting; |
| • | establishing policies and procedures for the receipt and retention of accounting-related complaints and concerns; |
| • | recommending based upon the audit committee’s review and discussions with management and our independent registered public accounting firm whether our audited financial statements shall be included in our Annual Report on Form 10-K; |
| • | monitoring the integrity of our financial statements and our compliance with legal and regulatory requirements as they relate to our financial statements and accounting matters; |
| • | preparing the audit committee report required by SEC rules to be included in our annual proxy statement; |
| • | reviewing material related person transactions for potential conflict of interest situations and approving such transactions; and |
| • | at least annually, reviewing and reassessing the adequacy of the audit committee charter and recommending to the board of directors any amendments or modifications to the charter that the audit committee deems appropriate. |
Compensation Committee
Dr. Cohen, Dr. McGraw and Mr. Kirsch currently serve on the compensation committee, which is chaired by Dr. McGraw. Our board of directors has determined that each member of the compensation committee is “independent” as defined in the applicable NYSE rules. Each of the members of this committee is also a “nonemployee director” as that term is defined under Rule 16b-3 of the Exchange Act and an “outside director” as that term is defined in Treasury Regulation Section 1.162-27(3). The compensation committee’s responsibilities include:
| • | reviewing on a periodic basis the operation of our executive compensation programs to determine whether they remain supportive of our business objectives; |
14
Table of Contents
| • | reviewing the performance of our CEO and approving the compensation of our CEO; |
| • | reviewing the performance of our other executive officers and approving or recommending to the board of directors the compensation of our other executive officers; |
| • | overseeing and administering our compensation and similar plans; |
| • | reviewing and approving structures and guidelines for various incentive compensation and benefit plans; |
| • | approving and recommending to the board of directors the compensation of our outside directors; |
| • | preparing the compensation committee report required by SEC rules, if and when required, to be included in our annual proxy statement; |
| • | reviewing and approving the retention, termination or compensation of any consulting firm or outside advisor to assist in the evaluation of compensation matters; and |
| • | at least annually, reviewing and reassessing the adequacy of the compensation committee charter and recommending to the board of directors any amendments or modifications to the charter that the compensation committee deems appropriate. |
Nominating and Corporate Governance Committee
Dr. McGraw, Dr. Cohen, Ms. Weegen and Mr. Peña currently serve on the nominating and corporate governance committee, which is chaired by Dr. Cohen. Our board of directors has determined that each member of the nominating and corporate governance committee, other than Mr. Peña, is “independent” as defined in the applicable NYSE rules and all applicable laws. Mr. Peña, by virtue of his position as our Chief Executive Officer, is not independent under applicable rules and regulations of the SEC and the NYSE listing rules. We are relying on the phase-in exemption provided under the NYSE listing rules with respect to the composition of our nominating and corporate governance committee. Mr. Peña will serve on our nominating and corporate governance committee under the phase-in exemption referenced above. The nominating and corporate governance committee’s responsibilities include:
| • | establishing procedures for identifying and evaluating board of director candidates, including nominees recommended by stockholders; |
| • | reviewing the composition of the board of directors to ensure that it is composed of members containing the appropriate skills and expertise to advise us; |
| • | identifying individuals qualified to become members of the board of directors; |
| • | recommending to the board of directors the persons to be nominated for election as directors and to each of the board’s committees; |
| • | developing and recommending to the board of directors a code of business conduct and ethics and a set of corporate governance guidelines; |
| • | overseeing the evaluation of our board of directors; and |
| • | at least annually, reviewing and reassessing the adequacy of the nominating and corporate governance committee charter and recommending to the board of directors any amendments or modifications to the charter that the nominating and corporate governance committee deems appropriate. |
Science and Technology Committee
We also maintain a science and technology committee to primarily assist our board of directors in its oversight of our research and development strategies, programs, priorities and opportunities. Dr. Bauer,
15
Table of Contents
Dr. McGraw, Dr. Cohen, Dr. DiRocco, Dr. Hopfner and Ms. Weegen serve on the science and technology committee, which is chaired by Dr. Cohen. The science and technology committee’s responsibilities include:
| • | reviewing, articulating, translating and contextualizing our scientific data for the general board discussion; |
| • | reviewing, evaluating and advising our board of directors and management regarding the long-term strategic goals and objectives and the quality and direction of our research and development programs; |
| • | considering trends in research and development, and recommending to our board of directors and management emerging technologies for building our technological strength; |
| • | advising our board of directors and management on the scientific aspects of potential business development transactions and market opportunities; |
| • | as needed, acting as a liaison between the medical community and our board of directors and/or management; and |
| • | periodically reviewing our overall intellectual property strategies. |
Our board of directors may from time to time establish other committees.
Compensation Committee Interlocks and Insider Participation
None of the members of our compensation committee has been one of our officers or employees at any time during the prior three years. None of our executive officers currently serves, or in the past fiscal year has served, as a member of the board of directors or compensation committee of any entity that has one or more executive officers serving on our board of directors or Compensation Committee.
Corporate Governance
We have adopted a written code of business conduct and ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller or persons performing similar functions. A current copy of the code of business conduct and ethics is posted on the investor relations section of our website, which is located at www.evommune.com. The inclusion of our website address in this prospectus does not incorporate by reference the information on or accessible through our website into this prospectus. If we make any substantive amendments to, or grant any waivers from, the code of business conduct and ethics for any officer or director, we will disclose the nature of such amendment or waiver on our website or in a current report on Form 8-K.
Limitation on Liability and Indemnification Matters
Our amended and restated certificate of incorporation contain provisions that limit the liability of our directors for monetary damages to the fullest extent permitted by Delaware law. Consequently, our directors will not be personally liable to us or our stockholders for monetary damages for any breach of fiduciary duties as directors, except liability for:
| • | any breach of the director’s duty of loyalty to us or our stockholders; |
| • | any act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; |
| • | unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the DGCL; or |
| • | any transaction from which the director derived an improper personal benefit. |
16
Table of Contents
Our amended and restated bylaws provide that we are required to indemnify our directors and officers, in each case to the fullest extent permitted by Delaware law. Our amended and restated bylaws also obligate us to advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding and permit us to secure insurance on behalf of any officer, director, employee or other agent for any liability arising out of his or her actions in that capacity regardless of whether we would otherwise be permitted to indemnify him or her under Delaware law. We have entered into agreements to indemnify our directors, executive officers and other officers as determined by our board of directors or our compensation committee. With specified exceptions, these agreements provide for indemnification for related expenses including, among other things, attorneys’ fees, judgments, fines and settlement amounts incurred by any of these individuals in certain actions or proceedings.
We believe that these certificate of incorporation and bylaw provisions and indemnification agreements are necessary to attract and retain qualified persons as directors and officers. We also maintain directors’ and officers’ liability insurance.
The limitation of liability and indemnification provisions in our amended and restated certificate of incorporation and amended and restated bylaws may discourage stockholders from bringing a lawsuit against our directors and officers for breach of their fiduciary duty. They may also reduce the likelihood of derivative litigation against our directors and officers, even though an action, if successful, might benefit us and our stockholders. Further, a stockholder’s investment may be adversely affected to the extent that we pay the costs of settlement and damage.
17
Table of Contents
Our named executive officers for fiscal year 2025, consisting of our principal executive officer and the next two most highly compensated executive officers, were:
| • | Luis Peña, Chief Executive Officer; |
| • | Gregory S. Moss, our Chief Business and Legal Officer; and |
| • | Kyle Carver, our Chief Financial Officer. |
Summary Compensation Table
The following table presents all of the compensation awarded to or earned by or paid to our named executive officers for fiscal years 2025 and 2024.
| Name and Principal Position |
Year | Salary ($) | Bonus ($)(1) | Stock awards ($)(2) |
Option awards ($)(2) |
All other compensation ($)(3) |
Total ($) | |||||||||||||||||||||
| Luis Peña | 2025 | 588,462 | 740,675 | — | 9,693,533 | — | 11,022,670 | |||||||||||||||||||||
| Chief Executive Officer |
2024 | 546,083 | 382,200 | 565,119 | 5,950,489 | 714 | 7,444,605 | |||||||||||||||||||||
| Gregory S. Moss | 2025 | 515,042 | 508,433 | — | 3,570,591 | 6,900 | 4,600,966 | |||||||||||||||||||||
| Chief Business & Legal Officer |
2024 | 480,500 | 269,080 | 222,632 | 382,548 | 714 | 1,355,474 | |||||||||||||||||||||
| Kyle Carver | 2025 | 501,875 | 496,658 | — | 3,399,210 | 6,900 | 4,404,643 | |||||||||||||||||||||
| Chief Financial Officer |
2024 | 470,000 | 263,200 | 217,149 | 286,008 | 714 | 1,237,071 | |||||||||||||||||||||
| (1) | Amounts reported for 2025 represent discretionary bonuses paid to our named executive officers with respect to 2025. |
| (2) | Amounts reported represent the aggregate grant date fair value of stock and option and stock appreciation right awards granted to our named executive officers in each respective fiscal year as determined in accordance with ASC Topic 718. Assumptions used in the calculation of these amounts are included in Note 9 to our financial statements included in our Annual Report on Form 10-K for the year ended December 31, 2025. These amounts do not necessarily correspond to the actual value recognized or that may be recognized by the named executive officers. |
| (3) | Amounts reported for 2025 include 401(k) match contributions paid by the Company for the benefit of Messrs. Moss and Carver, respectively. |
Narrative Disclosure to Summary Compensation Table
Base Salaries
Each of our current named executive officer’s annual base salary is a fixed component of annual compensation for performing specific duties and functions, and is established by our Board taking into account each individual’s role, responsibilities, skills, and expertise. Base salaries are reviewed annually, typically in connection with our annual performance review process, approved by our Board, and adjusted from time to time to realign salaries with market levels and internal benchmarking, after taking into account individual responsibilities, performance and experience. The 2026 annual base salaries for our named executive officers were as follows: $686,600 for Mr. Peña, $595,000 for Mr. Moss and $565,000 for Mr. Carver. Please see the “Salary” column in the Summary Compensation Table above for the actual base salary amount received by each current named executive officer for fiscal year 2025 and fiscal year 2024.
Bonus
In addition to base salaries, each of our named executive officers is eligible to receive a discretionary annual bonus of a percentage of the executive’s gross base salary based on individual performance, Company performance metrics such as certain research, development and clinical program execution, or as otherwise determined appropriate by our board of directors. For the year ended December 31, 2025, cash bonus targets, as a percentage of base salary, were 60% for Mr. Peña, and 40% for Messrs. Moss and Carver. The amount of any annual discretionary annual bonus is determined at the end of the year for which the bonus relates. On October 8, 2025, the compensation committee of our board of directors approved a post-IPO cash bonus target for Mr. Peña of 60%,
18
Table of Contents
increased from 50%. At the end of 2025, based on individual and Company performance, our compensation committee determined to pay discretionary annual bonuses at 60% of Mr. Peña’s base salary, 40% of Mr. Moss’s base salary, and 40% of Mr. Carver’s base salary. The actual 2025 bonus amounts for each of our named executive officers are reported in the “Bonus” column in the Summary Compensation Table above.
Equity-Based Incentive Awards
Our equity-based incentive awards are designed to align our named executive officers’ interests with those of our stockholders and to retain and incentivize our named executive officers over the long-term. Our compensation committee is responsible for approving equity grants. Vesting of equity awards is generally tied to continuous service with us and serves as an additional retention measure. Additional grants may occur periodically in order to specifically incentivize our named executive officers with respect to achieving certain corporate goals or to reward our named executive officers for exceptional performance.
We currently maintain the 2025 Equity Incentive Plan (“2025 Plan”), which our board of directors and stockholders approved in connection with our IPO for purposes of granting equity-based incentive awards to our employees and consultants, including our named executive officers. Prior to the effectiveness of our IPO, we granted equity incentive awards under the 2020 Stock Plan (“2020 Plan”), which was terminated in connection with our IPO.
In November 2025, prior to our IPO, our board of directors granted of Mr. Peña, Mr. Moss and Mr. Carver an option to purchase 796,841, 293,496 and 279,408 shares of our common stock, respectively. Each of these stock options has an exercise price of $16.00 per share. The terms of these awards are described under “—Outstanding Equity Awards at Fiscal Year End” below.
Outstanding Equity Awards as of December 31, 2025
The following table sets forth certain information about outstanding equity awards granted to our named executive officers that remain outstanding as of December 31, 2025.
| Option Awards (1) | Stock Awards (1) | |||||||||||||||||||||||||||||||
| Name |
Grant Date | Vesting Commencement Date |
Number of securities underlying unexercised options (#) exercisable |
Number of securities underlying unexercised options (#) unexercisable |
Option exercise price per share ($)(2) |
Option expiration date |
Number of shares or units of stock that have not vested (#) |
Market value of shares of units of stock that have not vested ($)(2) |
||||||||||||||||||||||||
| Luis Peña |
5/11/2022 | (3) | 5/11/2022 | 353,701 | 41,129 | 3.07 | 5/10/2032 | — | — | |||||||||||||||||||||||
| 1/13/2023 | (3) | 1/13/2023 | 74,879 | 27,813 | 3.58 | 1/12/2033 | — | — | ||||||||||||||||||||||||
| 5/12/2023 | (3) | 5/12/2023 | 124,073 | 68,041 | 3.84 | 5/11/2033 | — | — | ||||||||||||||||||||||||
| 1/6/2024 | (3) | 1/6/2024 | 11,402 | 12,395 | 1.71 | 1/5/2034 | — | — | ||||||||||||||||||||||||
| 12/9/2024 | (3) | 12/9/2024 | 72,693 | 218,081 | 2.99 | 12/8/2034 | — | — | ||||||||||||||||||||||||
| 12/9/2024 | (4) | 12/9/2024 | — | — | — | — | 141,753 | 2,426,811 | ||||||||||||||||||||||||
| 12/9/2024 | (5) | 12/9/2024 | — | 444,992 | 2.99 | 12/8/2034 | — | — | ||||||||||||||||||||||||
| 11/5/2025 | (3) | 11/5/2025 | — | 796,841 | 16.00 | 11/4/2035 | — | — | ||||||||||||||||||||||||
| Gregory S. Moss, Esq. |
7/15/2022 | (3) | 6/27/2022 | 117,104 | 16,730 | 3.07 | 7/14/2032 | — |
|
— |
| |||||||||||||||||||||
| 1/13/2023 | (3) | 1/13/2023 | 25,756 | 9,567 | 3.58 | 1/12/2033 | — | — | ||||||||||||||||||||||||
| 5/12/2023 | (3) | 5/12/2023 | 42,184 | 23,134 | 3.84 | 5/11/2033 | — | — | ||||||||||||||||||||||||
| 1/6/2024 | (3) | 1/6/2024 | 3,876 | 4,214 | 1.71 | 1/5/2034 | — | — | ||||||||||||||||||||||||
| 3/22/2024 | (3) | 3/22/2024 | 27,154 | 34,914 | 1.71 | 3/21/2034 | — | — | ||||||||||||||||||||||||
| 12/9/2024 | (3) | 12/9/2024 | 28,638 | 85,914 | 2.99 | 12/8/2034 | — | — | ||||||||||||||||||||||||
| 12/9/2024 | (4) | 12/9/2024 | — | — | — | — | 55,845 | 956,066 | ||||||||||||||||||||||||
| 11/5/2025 | (3) | 11/5/2025 | — | 293,496 | 16.00 | 11/4/2035 | — | — | ||||||||||||||||||||||||
19
Table of Contents
| Option Awards (1) | Stock Awards (1) | |||||||||||||||||||||||||||||||
| Name |
Grant Date | Vesting Commencement Date |
Number of securities underlying unexercised options (#) exercisable |
Number of securities underlying unexercised options (#) unexercisable |
Option exercise price per share ($)(2) |
Option expiration date |
Number of shares or units of stock that have not vested (#) |
Market value of shares of units of stock that have not vested ($)(2) |
||||||||||||||||||||||||
| Kyle Carver, |
5/11/2022 | (3) | 3/2/2022 | 111,045 | 7,404 | 3.07 | 5/10/2032 | — | — | |||||||||||||||||||||||
| M.B.A., C.P.A. |
1/13/2023 | (3) | 1/13/2023 | 22,463 | 8,344 | 3.58 | 1/12/2033 | — | — | |||||||||||||||||||||||
| 5/12/2023 | (3) | 5/12/2023 | 37,221 | 20,413 | 3.84 | 5/11/2033 | — | — | ||||||||||||||||||||||||
| 1/6/2024 | (3) | 1/6/2024 | 3,569 | 3,570 | 1.71 | 1/5/2034 | — | — | ||||||||||||||||||||||||
| 12/9/2024 | (3) | 12/9/2024 | 27,932 | 83,799 | 2.99 | 12/8/2034 | — | — | ||||||||||||||||||||||||
| 12/9/2024 | (4) | 12/9/2024 | — | — | — | — | 54,469 | 932,509 | ||||||||||||||||||||||||
| 11/5/2025 | (3) | 11/5/2025 | — | 279,408 | 16.00 | 11/4/2035 | — | — | ||||||||||||||||||||||||
| (1) | All of the equity awards were granted under our 2020 Plan or 2025 Plan, except for the stock appreciation rights (“SARs”) awarded to Mr. Peña on December 9, 2024, which were granted outside of our 2020 Plan. All of the option awards were granted with a per share exercise price equal to the fair value of one share of our common stock on the date of grant, as determined in good faith by our Board. |
| (2) | Amounts are calculated by multiplying the number of shares shown in the table by $17.12, the closing market price of our common stock on the NYSE as of December 31, 2025. |
| (3) | This stock option vests over a period of four years, with 25% of the shares underlying the option vesting on the one-year anniversary of the vesting commencement date, and 1/48 of the shares underlying the option vesting on a monthly basis thereafter, subject to continued service through each vesting date. |
| (4) | The restricted stock units (“RSUs”) vest based on the satisfaction of all of the following: (i) a liquidity event requirement; (ii) a Series C Preferred Stock requirement that was met in June 2025; and (iii) a service-based requirement, which is satisfied in equal annual installments on the first four anniversaries of the grant date. |
| (5) | The SARs vest upon the satisfaction all of the following: (i) a liquidity event requirement, which will be satisfied upon the first to occur of: (a) a Change in Control (as defined in the applicable SAR award agreement) or (b) at such time as the 30-day volume-weighted average price of our common stock is greater than $40.75 and (ii) a continued service requirement, which will be satisfied as to 25% of the shares subject to the SAR award on each of the first four anniversaries of the vesting commencement date, subject to Mr. Peña’s continued service with us through each such date. In addition to both the liquidity event and continued service requirements, 251,517 shares underlying such SAR award are subject to a Series C Preferred Stock requirement, which was met in June 2025. All unvested SARs are forfeited upon termination or resignation for any reason. |
Employment Arrangements with Our Named Executive Officers
We have entered into employment agreements with each of our named executive officers setting forth the terms and conditions of such executive’s employment with us. The employment agreements generally provide for at-will employment and set forth the executive officer’s initial base salary. Each of our named executive officers has executed our standard confidential information, inventions assignment and arbitration agreement.
Luis Peña
Effective November 1, 2024, we entered into an employment agreement with Mr. Peña (the “Peña Employment Agreement”). Under the Peña Employment Agreement, Mr. Peña’s employment with us can be terminated at any time and for any reason by him or us. The Peña Employment Agreement provides that Mr. Peña is entitled to an annualized base salary of $546,500, subject to adjustment in accordance with normal business practices and at our sole discretion, during his employment with us and that he is eligible, at our sole discretion, to earn an annual bonus targeted at 50% of his base salary. In 2025, we paid Mr. Peña an annualized base salary of $588,461, and we awarded him a bonus of $740,675, which bonus amount was determined by the board of directors in its discretion based on the achievement of predetermined Company performance metrics. Mr. Peña is also entitled to certain severance benefits, the terms of which are described below under “—Potential Payments Upon Termination or Change in Control.”
Gregory S. Moss
Effective November 1, 2024, we entered into an employment agreement with Mr. Moss (the “Moss Employment Agreement”). Under the Moss Employment Agreement, Mr. Moss’s employment with us can be terminated at any time and for any reason by him or us. The Moss Employment Agreement provides that
20
Table of Contents
Mr. Moss is entitled to an annualized base salary of $480,500, subject to adjustment in accordance with normal business practices and at our sole discretion, during his employment with us and that he is eligible, at our sole discretion, to earn an annual bonus targeted at 40% of his base salary. In 2025, we paid Mr. Moss an annualized base salary of $515,041, and we awarded him a bonus of $508,433, which bonus amount was determined by the board of directors in its discretion based on the achievement of predetermined Company performance metrics. Mr. Moss is also entitled to certain severance benefits, the terms of which are described below under “—Potential Payments Upon Termination or Change in Control.”
Kyle Carver
Effective November 1, 2024, we entered into an employment agreement with Mr. Carver (the “Carver Employment Agreement”). Under the Carver Employment Agreement, Mr. Carver’s employment with us can be terminated at any time and for any reason by him or us. The Carver Employment Agreement provides that Mr. Carver is entitled to an annualized base salary of $470,000, subject to adjustment in accordance with normal business practices and at our sole discretion, during his employment with us and that he is eligible, at our sole discretion, to earn an annual bonus targeted at 40% of his base salary. In 2025, we paid Mr. Carver an annualized base salary of $501,875, and we awarded him a bonus of $496,658, which bonus amount was determined by the board of directors in its discretion based on the achievement of predetermined Company performance metrics. Mr. Carver is also entitled to certain severance benefits, the terms of which are described below under “—Potential Payments Upon Termination or Change in Control.”
Potential Payments Upon Termination or Change in Control
Regardless of the manner in which a named executive officer’s service terminates, each of our named executive officers is entitled to receive amounts earned during his term of service, including unpaid salary and any vested benefits. In the event of certain termination scenarios, each named executive officer will be entitled to receive the additional benefits summarized below pursuant to his employment agreement, subject to the timely execution and non-revocation of a separation agreement and general release of claims.
Termination for Cause or Resignation Without Good Reason
Pursuant to the terms of their respective employment agreements, if we terminate any of our named executive officer’s employment for cause or the named executive officer resigns without good reason (each of “cause” and “good reason” as defined in the applicable employment agreement), then the named executive officer will be entitled to receive (i) base salary through the date of termination and (ii) any vested benefits under our employee benefit plan through the date of termination.
Termination Without Cause or for Good Reason not in Connection with a Change in Control
Pursuant to the terms of their respective employment agreements, if we terminate any of our named executive officer’s employment without cause or the named executive officer resigns for good reason not in connection with a change in control (as defined in the applicable employment agreement), then the named executive officer will be entitled to receive (i) base salary continuation for 12 months following termination, (ii) an amount equal to the greater of (A) the full amount of his target bonus for the calendar year in which the termination of employment occurs and (B) the average annual bonus paid to him over the three calendar years ending prior to the year in which such termination of employment occurs and (iii) monthly payments of the portion of the premiums equal to the amounts that the Company would have paid to provide health insurance to the named executive officer had he remained employed with the Company until the earliest of (A) 12 months following termination or (B) the named executive officer becoming eligible for group medical plan benefits under any other employer’s group health plan. Each equity incentive award granted by the Company to the named executive officer that is then outstanding or any other then outstanding equity incentive award granted to the named executive officer under any successor plan or otherwise (each an “Equity Award” and, collectively the
21
Table of Contents
“Equity Awards”), including Equity Awards that would otherwise vest only upon satisfaction of performance criteria, shall accelerate and vest in full as of the date of such termination and, if applicable, become exercisable (with any such Equity Awards subject to vesting based on satisfaction of performance criteria vesting at target).
Termination Without Cause or for Good Reason in Connection with a Change in Control
Pursuant to the terms of their respective employment agreements, if we terminate a named executive officer’s employment without cause or the named executive officer resigns for good reason within three months prior to or twelve months following a change in control, the named executive officer will be entitled to receive (i) a lump sum cash payment equal to two times, in the case of Mr. Peña, or one-and-a-half times, in the case of Messrs. Moss and Carver, the sum of (x) the then-current base salary plus (y) the greater of (A) the full amount of his target bonus for the calendar year in which the termination of employment occurs and (B) the average annual bonus paid to him over the three calendar years ending prior to the year in which such termination of employment occurs and (ii) monthly payments of the portion of the premiums equal to the amounts that the Company would have paid to provide health insurance to the named executive officer had he remained employed with the Company until the earliest of (x) 24 months following termination or (y) the named executive officer becoming eligible for group medical plan benefits under any other employer’s group health plan. Each Equity Award that is then outstanding, including Equity Awards that would otherwise vest only upon satisfaction of performance criteria, shall accelerate and vest in full as of the date of such termination and, if applicable, become exercisable (with any such Equity Awards subject to vesting based on satisfaction of performance criteria vesting at target). In the case of the consummation of a change in control in which an outstanding Equity Award is not continued, assumed or substituted for by the Company or the acquiror or any of its affiliates as part of such change in control transaction, then such Equity Award will become fully vested and exercisable on or immediately prior to the closing date of the change in control, whether the named executive officer terminates employment or not.
Health and Welfare Benefits; Perquisites
Each of our named executive officers is eligible to participate in our employee benefit plans, including our medical, dental, vision, life and long-term disability plans, in each case on the same basis as all of our other employees. We pay a majority of the premiums for the medical, dental, vision and life insurance for all of our employees, including our named executive officers. We generally do not provide perquisites or personal benefits to our named executive officers. In addition, we provide the opportunity to participate in a 401(k) plan to our employees, including each of our named executive officers, as discussed in the section below entitled “401(k) Plan.”
401(k) Plan
Our named executive officers are eligible to participate in a defined contribution retirement plan that provides eligible U.S. employees with an opportunity to save for retirement on a tax-advantaged basis. Eligible employees may defer eligible compensation on a pre-tax or after-tax (Roth) basis, up to the statutorily prescribed annual limits on contributions under the Code. Individual contributions are allocated to each participant’s individual account and are then invested in selected investment alternatives according to the participants’ directions. The 401(k) plan is intended to be qualified under Section 401(a) of the Code with the 401(k) plan’s related trust intended to be tax exempt under Section 501(a) of the Code. As a tax-qualified retirement plan, contributions to the 401(k) plan (except for Roth contributions) and earnings on those contributions are not taxable to the employees until distributed from the 401(k) plan. We may elect, at our discretion, to make matching employee contributions.
Compensation Recovery (“Clawback”) Policy
As a public company, if we are required to restate our financial results due to our material noncompliance with any financial reporting requirements under the federal securities laws as a result of misconduct, the Chief
22
Table of Contents
Executive Officer and Chief Financial Officer may be legally required to reimburse our company for any bonus or other incentive-based or equity-based compensation they receive in accordance with the provisions of Section 304 of the Sarbanes-Oxley Act of 2002, as amended. Additionally, we adopted an incentive compensation clawback policy at the time of our IPO that complies with the new SEC rules under the Dodd-Frank Wall Street Reform and Consumer Protection Act and related stock exchange listing rules.
Emerging Growth Company Status
We became a public company in November 2025 and we are an “emerging growth company” under applicable federal securities laws and therefore permitted to take advantage of certain reduced public company reporting requirements. As an emerging growth company, we provide in this prospectus the scaled disclosure permitted under the JOBS Act, including certain executive compensation disclosures required of a “smaller reporting company,” as that term is defined in Rule 12b-2 promulgated under the Exchange Act. In addition, as an emerging growth company, we are not required to conduct votes seeking approval, on an advisory basis, of the compensation of our named executive officers or the frequency with which such votes must be conducted. We will remain an emerging growth company until the earliest of (i) December 31, 2030, (ii) the last day of the first fiscal year in which our annual gross revenue is $1.235 billion or more, (iii) the date on which we have, during the previous rolling three-year period, issued more than $1.235 billion in non-convertible debt securities, or (iv) the date on which we are deemed to be a “large accelerated filer” as defined in the Exchange Act.
Non-Employee Director Compensation
Director Compensation Table
The following table sets forth information regarding the compensation of our non-employee directors earned for service on our Board during the year ended December 31, 2025.
| Name |
Fees Earned or Paid in Cash ($) |
Option Awards ($)(1) |
Total ($) | |||||||||
| Benjamin F. McGraw, III, Pharm.D. |
50,833 | 439,994 | 490,827 | |||||||||
| David E. Cohen, M.D., M.P.H. |
44,583 | 439,994 | 484,577 | |||||||||
| Derek DiRocco, Ph.D. |
— | — | — | |||||||||
| Rob Hopfner, R.Ph., Ph.D. |
— | — | — | |||||||||
| Felice Verduyn-van Weegen |
— | — | — | |||||||||
| Arthur Kirsch |
11,250 | 439,994 | 451,244 | |||||||||
| François Beaubien, Ph.D., C.F.A. (2) |
— | — | — | |||||||||
| Yuexin Yu (2) |
— | — | — | |||||||||
| (1) | The amounts reported in this column represent the aggregate grant date fair value of the stock options granted to our non-employee directors, computed in accordance with FASB ASC Topic 718. The assumptions used in calculating the grant date fair value of the stock options reported in this column are set forth in Note 9 to our audited financial statements included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2025. The amounts do not reflect dollar amounts actually received by the non-employee director or the economic value that may be received by the non-employee director upon stock option exercise or any sale of the underlying shares of common stock. |
23
Table of Contents
The following table provides information regarding the number of shares of common stock underlying options held by our non-employee directors that were outstanding as of December 31, 2025:
| Shares underlying Outstanding Options as of December 31, 2025 |
||||
| Benjamin F. McGraw, III, Pharm.D. |
137,467 | |||
| David E. Cohen, M.D., M.P.H. |
122,793 | |||
| Derek DiRocco, Ph.D. |
— | |||
| Rob Hopfner, R.Ph., Ph.D. |
— | |||
| Felice Verduyn-van Weegen |
— | |||
| Arthur Kirsch |
37,960 | |||
| François Beaubien, Ph.D., C.F.A. |
— | |||
| Yuexin Yu |
— | |||
| (2) | Dr. Beaubien and Mr. Yu served as members of our Board until their respective resignations in connection with our IPO. |
No compensation was paid or earned by any other directors. Under our director compensation program, we pay our non-employee directors a cash retainer for service on the board of directors and for service on each committee on which the director is a member. The chairman of the board of directors and of each committee receive higher retainers for such service. These fees will be payable in arrears in four equal quarterly installments on the last day of each quarter, provided that the amount of such payment will be prorated for any portion of such quarter that the director is not serving on our board of directors. The fees paid to non-employee directors for service on the board of directors and for service on each committee of the board of directors on which the director is a member are as follows:
| Member Annual Fee ($) | Chairman Annual Fee ($) | |||||||
| Board of Directors |
40,000 | 35,000 | ||||||
| Audit Committee |
10,000 | 20,000 | ||||||
| Compensation Committee |
7,500 | 15,000 | ||||||
| Nominating and Corporate Governance Committee |
5,000 | 10,000 | ||||||
We also reimburse our non-employee directors for reasonable travel and other expenses incurred in connection with attending meetings of our board of directors and any committee of our board of directors on which he or she serves.
In addition, under our director compensation program, each non-employee director will receive, upon his or her initial election or appointment to our board of directors, an option to purchase the largest whole number of shares of our common stock that results in such option having an aggregate Black-Scholes value not exceeding $440,000. One-third of each of these options will vest on the one-year anniversary of the date of grant, with the remainder vesting in twenty-four equal monthly installments thereafter until the third anniversary of the grant date, subject to the non-employee director’s continued service as a director.
Further, on the date of the first board meeting held after each annual meeting of stockholders, each non-employee director will receive an option to purchase the largest whole number of shares of our common stock that results in such option having an aggregate Black-Scholes value not exceeding $220,000. Each of these options will vest the earlier of the first anniversary of the date of grant or the next annual stockholder meeting, subject to the non-employee director’s continued service as a director. All options issued to our non-employee directors under our director compensation program will be issued at exercise prices equal to the fair market value of our common stock on the date of grant and will become exercisable in full upon specified change in control events.
24
Table of Contents
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Other than the compensation arrangements, including employment, termination of employment and change in control arrangements, with our directors and executive officers, including those discussed in the sections entitled “Management” and “Executive and Director Compensation” and the registration rights described in the section entitled “Description of Capital Stock—Registration Rights,” the following is a description of each transaction since January 1, 2024 and each currently proposed transaction in which:
| • | we have been or are to be a participant; |
| • | the amounts involved exceeded or will exceed the lesser of $120,000 or 1% of the average of our total assets at year-end for the last two completed fiscal years; and |
| • | any of our directors, executive officers or beneficial owners of more than 5% of our capital stock or any member of the immediate family of the foregoing persons, had or will have a direct or indirect material interest. |
Private Placements of Securities
Convertible Preferred Stock Financings
In October 2024, we sold 31,493,523 shares of our Series C Preferred Stock at a purchase price of $1.59453 per share for gross proceeds of approximately $50.2 million. In June 2025, we sold an additional 40,941,587 shares of our Series C Preferred Stock at a purchase price of $1.59453 per share for gross proceeds of approximately $65.2 million. The following table summarizes purchases of shares of our Series C Preferred Stock by related persons:
| Stockholder |
Shares of Series C Preferred Stock |
Total Purchase Price |
||||||
| Entities affiliated with RA Capital(1) |
18,814,319 | $ | 29,999,997 | |||||
| LSP 7 Coöperatief UA(2) |
6,585,011 | $ | 10,499,998 | |||||
| Entities affiliated with Pivotal bioVenture(3) |
3,135,717 | $ | 4,999,995 | |||||
| BioDiscovery 6 FPCI(4) |
1,881,431 | $ | 2,999,999 | |||||
| (1) | Beneficial owner of greater than 5% of our outstanding capital stock. Additionally, Derek DiRocco, a member of our board of directors, is affiliated with RA Capital. Additional details regarding this stockholder and its equity holdings are included elsewhere in this prospectus under the caption “Principal Stockholders.” |
| (2) | Beneficial owner of greater than 5% of our outstanding capital stock. Additionally, Felice Verduyn-van Weegen, a member of our board of directors, is affiliated with LSP Coöperatief UA. Additional details regarding this stockholder and its equity holdings are included elsewhere in this prospectus under the caption “Principal Stockholders.” |
| (3) | Beneficial owner of greater than 5% of our outstanding capital stock. Consists of shares purchased by Pivotal bioVenture Partners Fund I, L.P., Pivotal bioVenture Partners Fund II, L.P. and NFLS Delta III Limited. Additionally, Robert Hopfner, a member of our board of directors, is affiliated with Pivotal bioVenture. Additional details regarding this stockholder and its equity holdings are included elsewhere in this prospectus under the caption “Principal Stockholders.” |
| (4) | Beneficial owner of greater than 5% of our outstanding capital stock. Additionally, Yuexin Yu, a former member of our board of directors, is affiliated with BioDiscovery 6 FPCI. For additional details regarding this stockholder and its equity holdings, see the section titled “Principal Stockholders.” |
25
Table of Contents
Initial Public Offering – Directed Share Program
In November 2025, we closed the IPO, pursuant to which we issued and sold 10,781,250 shares of our common stock, including full exercise of the underwriters’ option to purchase 1,406,250 additional shares, at a public offering price of $16.00 per share. The following table sets forth the aggregate cash purchase price paid by our directors, executive officers, and 5% stockholders and their affiliates and the number of shares of our common stock issued in consideration of such amounts. Such purchases were made through the underwriters at the initial public offering price of $16.00 per share.
| Shares of Common Stock (#) |
Total Purchase Price ($) |
|||||||
| LSP 7 Coöperatief UA(1) |
1,562,500 | 25,000,000 | ||||||
| Entities Affiliated with RA Capital(2) |
1,250,000 | 20,000,000 | ||||||
| BioDiscovery 6 FPCI(3) |
93,750 | 1,500,000 | ||||||
| Peter Bisgaard(4) |
10,000 | 160,000 | ||||||
| Harvey Carver(5) |
3,000 | 48,000 | ||||||
| Martin Forster(6) |
550 | 8,800 | ||||||
|
|
|
|
|
|||||
| Total |
2,919,800 | 46,716,800 | ||||||
|
|
|
|
|
|||||
| (1) | Beneficial owner of greater than 5% of our outstanding capital stock. Derek DiRocco, a member of our board of directors, is affiliated with RA Capital. |
| (2) | Beneficial owner of greater than 5% of our outstanding capital stock. Felice Verduyn-van Weegen, a member of our board of directors, is affiliated with LSP Coöperatief UA. |
| (3) | Beneficial owner of greater than 5% of our outstanding capital stock. Yuexin Yu, a former member of our board of directors, is affiliated with BioDiscovery 6 FPCI. |
| (4) | A Managing Director at Pivotal bioVenture. Entities affiliated with Pivotal bioVenture beneficially own greater than 5% of our outstanding capital stock. |
| (5) | Harvey Carver is the father of Kyle Carver, our Chief Financial Officer. |
| (6) | The father-in-law of Luis Peña, our Chief Executive Officer. |
2026 Private Placement
On February 12, 2026, we entered into the Purchase Agreement with the Selling Stockholders, pursuant to which we issued and sold to the Selling Stockholders an aggregate of 4,494,279 Shares. Each Share was offered and sold at a purchase price of $27.88. The Private Placement closed on February 17, 2026.
Immediately prior to the closing of the Private Placement, each of LSP 7 Coöperatief UA and RA Capital was the beneficial owner of more than 5% of our capital stock and participated in the Private Placement purchasing an aggregate of 358,680 shares and 35,868 shares of our common stock, respectively. For more information regarding the investors’ beneficial ownership, see section titled “Principal Stockholders” above.
Investor Agreements
In connection with our convertible preferred stock financings, we entered into investors’ rights, right of first refusal, and co-sale and voting agreements, which contain, among other things, registration rights, information rights, voting rights, and rights of first refusal, with certain holders of our capital stock, including entities affiliated with LSP 7 Coöperatief UA, Pivotal bioVenture, RA Capital and BioDiscovery 6 FPCI, each a holder of more than 5% of our capital stock. These agreements terminated upon the closing of the IPO, except for the registration rights granted under our investors’ rights agreement. See also the section titled “Principal Stockholders” for additional information regarding beneficial ownership of our capital stock.
26
Table of Contents
Equity Grants to Directors and Executive Officers
We have granted stock options and restricted stock awards to our executive officers and certain members of our Board. For more information regarding the options granted to our executive officers and directors, see the sections titled “Non-Employee Director Compensation” and “Executive Compensation.”
Indemnification Agreements
Our amended and restated certificate of incorporation contains provisions limiting the liability of directors and officers, and our amended and restated bylaws provide that we will indemnify each of our directors and officers to the fullest extent permitted under Delaware law. Our amended and restated certificate of incorporation and amended and restated bylaws also provide our Board with discretion to indemnify our employees when determined appropriate by the Board. In addition, we have entered into indemnification agreements with each of our directors and executive officers.
Related Person Transactions Policy
We have adopted a written related-person transactions policy that sets forth our policies and procedures regarding the identification, review, consideration, and approval or ratification of “related-person transactions.” For purposes of our policy only, a “related-person transaction” is a transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which we and any “related person” are, were or will be participants involving an amount that exceeds $120,000 or, if less, 1% of the average of our total assets for the last two competed fiscal years. Transactions involving compensation for services provided to us as an employee or director are not covered by this policy. A related person is any executive officer, director, nominee to become a director or a beneficial owner of more than 5% of any class of our voting securities, including any of their immediate family members and affiliates, including entities owned or controlled by such persons.
Under the policy, where a transaction has been identified as a related-person transaction, including any transaction that was not a related person transaction when originally consummated or any transaction that was not initially identified as a related person transaction prior to consummation, our management must present information regarding the proposed related person transaction to our Audit Committee (or, where review by our Audit Committee would be inappropriate, to another independent body of the Board) for review, consideration, and approval or ratification. The presentation must include a description of, among other things, all of the parties thereto, the material facts, the interests, direct and indirect, of the related persons, the benefits to us of the transaction, and whether the transaction is on terms that are comparable to the terms available to or from, as the case may be, an unrelated third party or to or from employees generally. Under the policy, we collect information that we deem reasonably necessary from each director, executive officer, and, to the extent feasible, significant stockholder to enable us to identify any existing or potential related-person transactions and to effectuate the terms of the policy. In addition, under our Code of Conduct, our employees and directors have an affirmative responsibility to disclose any transaction or relationship that reasonably could be expected to give rise to a conflict of interest.
In considering related person transactions, our Audit Committee, or other independent body of the Board, will take into account the relevant available facts and circumstances including:
| • | the risks, costs, and benefits to us; |
| • | the impact on a director’s independence in the event the related person is a director, immediate family member of a director or an entity with which a director is affiliated; |
| • | the terms of the transaction; |
| • | the availability of other sources for comparable services or products; and |
| • | the terms available to or from, as the case may be, unrelated third parties or to or from employees generally. |
27
Table of Contents
The policy requires that, in determining whether to approve, ratify or reject a related person transaction, our Audit Committee, or other independent body of our board of directors, must consider, in light of known circumstances, whether the transaction is in, or is not inconsistent with, our best interests and those of our stockholders, as our audit committee, or other independent body of our board of directors, determines in the good faith exercise of its discretion.
All of the transactions described above prior to our IPO were entered into prior to the adoption of this policy. Although we did not previously have a written policy for the review and approval of transactions with related persons, our board of directors has historically reviewed and approved any transaction where a director or officer had a financial interest, including the transactions described above. Prior to approving such a transaction, the material facts as to a director’s or officer’s relationship or interest in the agreement or transaction were disclosed to our board of directors. Our board of directors took this information into account when evaluating the transaction and in determining whether such transaction was fair to us and in the best interest of all our stockholders.
28
Table of Contents
The following table sets forth information with respect to the beneficial ownership of our shares of common stock as of March 31, 2026 by:
| • | each person, or group of affiliated persons, known by us to beneficially own more than 5% of our common stock; |
| • | each named executive officer; |
| • | each of our directors; and |
| • | all of our current directors and executive officers as a group. |
We have determined beneficial ownership in accordance with the rules of the SEC. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities. In addition, the rules include shares of common stock issuable pursuant to the exercise of stock options or warrants that are either immediately exercisable or exercisable on or before May 30, 2026, which is 60 days after March 31, 2026. These shares are deemed to be outstanding and beneficially owned by the person holding those options or warrants for the purpose of computing the percentage ownership of that person, but they are not treated as outstanding for the purpose of computing the percentage ownership of any other person. Unless otherwise indicated, we believe based on information provided to us, the persons or entities identified in this table have sole voting and investment power with respect to all shares shown as beneficially owned by them, subject to applicable community property laws.
The percentage ownership information shown in the table below is based on 36,018,372 shares of common stock outstanding as of March 31, 2026.
Unless otherwise indicated, the address for each beneficial owner listed in the table below is c/o Evommune, Inc., 1891 Page Mill Road, Palo Alto, California 94304.
| Beneficial Ownership | ||||||||
| Name and Address of Beneficial Owner |
Number of Shares Beneficially Owned |
Percentage of Shares Beneficially Owned |
||||||
| 5% or Greater Stockholders: |
||||||||
| LSP 7 Coöperatief UA(1) |
5,288,313 | 14.7 | % | |||||
| Entities Affiliated with RA Capital(2) |
3,494,638 | 9.7 | % | |||||
| Entities Affiliated with Pivotal bioVenture(3) |
3,367,975 | 9.4 | % | |||||
| BioDiscovery 6 FPCI(4) |
1,897,362 | 5.3 | % | |||||
| Named Executive Officers and Directors: |
||||||||
| Luis Peña(5) |
1,396,037 | 3.8 | % | |||||
| Eugene A. Bauer, M.D.(6) |
468,696 | 1.3 | % | |||||
| Kyle Carver, M.B.A., C.P.A.(7) |
293,740 | * | ||||||
| Gregory S. Moss, Esq.(8) |
352,387 | 1.0 | % | |||||
| Benjamin F. McGraw, III, Pharma. D.(9) |
89,907 | * | ||||||
| David E. Cohen, M.D., M.P.H.(10) |
97,002 | * | ||||||
| Derek DiRocco, Ph.D |
— | — | ||||||
| Rob Hopfner, R.Ph., Ph.D.(3)(11) |
3,372,001 | 9.4 | % | |||||
| Felice Verduyn-van Weegen(1) |
5,288,313 | 14.7 | % | |||||
| Arthur Kirsch |
— | * | ||||||
| All current directors and executive officers as a group (12 persons)(12) |
11,814,214 | 31.11 | % | |||||
| * | Represents beneficial ownership of less than 1%. |
29
Table of Contents
| (1) | As the sole director of LSP 7 Coöperatief UA, LSP 7 Management BV may be deemed to beneficially own these securities. As managing directors of LSP 7 Management BV, each of Martijn Kleijwegt, René Kuijten and Joachim Rothe may also be deemed to beneficially own these securities. The business address of LSP 7 Coöperatief UA is Johannes Vermeerplein 9 1071 DV Amsterdam, Netherlands. |
| (2) | Consists of (i) 2,710,293 shares of common stock held by RA Capital Healthcare Fund, LP, and (ii) 784,345 shares of common stock held by RA Capital Nexus Fund III, L.P (the “RA Funds”). RA Capital Management, L.P. is the investment manager for the RA Funds. The general partner of RA Capital Management, L.P. is RA Capital Management GP, LLC, of which Peter Kolchinsky and Rajeev Shah are the managing members. Each of RA Capital Management, L.P., RA Capital Management GP, LLC, Mr. Kolchinsky and Mr. Shah may be deemed to have voting and investment power over the shares held by the RA Funds. RA Capital Management, L.P., RA Capital Management GP, LLC, Mr. Kolchinsky and Mr. Shah disclaim beneficial ownership of such shares, except to the extent of any pecuniary interest therein. The principal business address of the persons and entities listed above is 200 Berkeley Street, 18th Floor, Boston, MA 02116. |
| (3) | Consists of (i)(a) 419,441 shares of common stock issuable upon the conversion of Series Seed preferred stock, (b) 908,821 shares of common stock issuable upon the conversion of Series A preferred stock, (c) 267,367 shares of common stock issuable upon the conversion of Series B preferred stock, and (d) 36,812 shares of common stock issuable upon the conversion of Series C preferred stock in each case held by Pivotal bioVenture Partners Fund I, L.P. (“Pivotal I”), (ii) 294,502 shares of common stock issuable upon the conversion of Series C preferred stock held by Pivotal bioVenture Partners Fund II, L.P. (“Pivotal II”), and (iii)(a) 838,886 shares of common stock issuable upon the conversion of Series Seed preferred stock, (b) 324,578 shares of common stock issuable upon the conversion of Series A preferred stock, (c) 240,756 shares of common stock issuable upon the conversion of Series B preferred stock preferred stock, and (d) 36,812 shares of common stock issuable upon the conversion of Series C preferred stock preferred stock in each case held by NFLS Delta III Limited (“NFLS Delta”). NFLS Delta, Pivotal bioVenture Partners Fund I G.P., L.P., the general partner of Pivotal I, and Pivotal bioVenture Partners Fund II G.P. Ltd, the general partner of Pivotal II, are each wholly-owned indirect subsidiaries of Nan Fung Group Holdings Limited. Investment and voting decisions with respect to the securities held by NFLS Delta are made by the members of Nan Fung Group Holdings Limited’s Life Sciences Investment Committee, who are Mr. Kam Chung Leung, Mr. Vincent Sai Sing Cheung, Mr. Stephen Pui Kuen Cheung, Ms. Vanessa Tih Lin Cheung, Mr. Meng Gao, Ms. Anna Xintong Sun, Mr. Peter Bisgaard, and Dr. Robert Hopfner. Investment and voting decisions with respect to the securities held by Pivotal I and Pivotal II are made by the members of the Investment Committees of Pivotal bioVenture Partners Fund I G.P., L.P., and Pivotal bioVenture Partners Fund II, G.P. Ltd., who are Mr. Vincent Sai Sing Cheung, Mr. Peter Bisgaard, and Dr. Robert Hopfner. The address of NFLS Delta and the individual members of Nan Fung Group Holdings Limited’s Life Sciences Investment Committee other than Mr. Peter Bisgaard and Dr. Robert Hopfner is 17th Floor, Airside, 2 Concorde Road, Kai Tak, Hong Kong SAR. The address of all other entities and individuals referenced in this footnote is 501 2nd Street, Suite 200, San Francisco, California 94107. |
| (4) | Voting and investment decisions by BioDiscovery 6 FPCI is exercised by its Management Company Andera Partners. The Managing Partners of Andera Partners are François-Xavier Mauron (Chairman) and Stéphane Bergez (Chief Executive Officer), neither of whom has individual voting or investment power with respect to the shares. |
| (5) | Consists of (i) 654,683 shares of common stock beneficially owned by Mr. Peña and (ii) 741,354 shares of common stock issuable upon the exercise of options held by Mr. Peña that are exercisable within 60 days of March 31, 2026. |
| (6) | Consists of (i) 270,818 shares of common stock beneficially owned by Dr. Bauer and (ii) 197,878 shares of common stock issuable upon the exercise of options held by Dr. Bauer that are exercisable within 60 days of March 31, 2026. |
| (7) | Consists of (i) 62,510 shares of common stock and (ii) 231,230 shares of common stock issuable upon the exercise of options held by Mr. Carver that are exercisable within 60 days of March 31, 2026. |
| (8) | Consists of (i) 64,010 shares of common stock beneficially held by Mr. Moss, and (ii) 288,377 shares of common stock issuable upon the exercise of options held by Mr. Moss that are exercisable within 60 days of March 31, 2026. |
30
Table of Contents
| (9) | Consists of 89,907 shares of common stock issuable upon the exercise of options held by Dr. McGraw that are exercisable within 60 days of March 31, 2026. |
| (10) | Consists of (i) 23,665 shares of common stock beneficially owned by Dr. Cohen and (ii) 73,337 shares of common stock issuable upon the exercise of options held by Dr. Cohen that are exercisable within 60 days of March 31, 2026. |
| (11) | Consists of (i) 4,026 shares of common stock beneficially owned by Dr. Hopfner. |
| (12) | Consists of (i) 9,853,372 shares of common stock beneficially owned by our current executive officers and directors, and (ii) 1,960,842 shares of common stock issuable upon the exercise of options held by our current executive officers and directors within 60 days of March 31, 2026. |
31
Table of Contents
The following description of our common stock and certain provisions of our amended and restated certificate of incorporation (the “restated certificate”) and amended and restated bylaws (the “restated bylaws”) are summaries. These summaries are qualified in their entirety by reference to the provisions of the Delaware General Corporation Law and the complete text of the restated certificate and restated bylaws, which are incorporated by reference as Exhibits 3.1 and 3.2, respectively, of the Company’s Annual Report on Form 10-K to which this description is also an exhibit.
General
The restated certificate authorizes the Company to issue up to 500,000,000 shares of common stock, par value $0.0001 per share, and 10,000,000 shares of preferred stock, par value $0.0001 per share, all of which shares of preferred stock are undesignated. The Company’s board of directors may establish the rights and preferences of the preferred stock from time to time.
Common Stock
Voting
Each holder of the Company’s common stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders, including the election of directors. The Company’s stockholders do not have cumulative voting rights in the election of directors. Accordingly, holders of a majority of the voting shares are able to elect all of the directors.
Dividends
Subject to preferences that may be applicable to any then-outstanding preferred stock, the holders of common stock are entitled to receive ratably dividends, if any, as may be declared from time to time by the Company’s board of directors out of legally available funds.
Liquidation
In the event of the Company’s liquidation, dissolution or winding-up, holders of its common stock will be entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all of the Company’s debts and other liabilities, subject to the satisfaction of any liquidation preference granted to the holders of any outstanding shares of preferred stock.
Rights, Preferences, and Privileges
Holders of the Company’s common stock have no preemptive, conversion, or subscription rights, and there are no redemption or sinking fund provisions applicable to its common stock. The rights, preferences, and privileges of the holders of the Company’s common stock are subject to, and may be adversely affected by, the rights of the holders of shares of any series of the Company’s preferred stock that may be designated and issued in the future.
Fully Paid and Nonassessable
All of the Company’s outstanding shares of common stock are fully paid and nonassessable.
Preferred Stock
The Company’s board of directors has the authority, without action by the stockholders, to designate and issue up to an aggregate of 10,000,000 shares of preferred stock in one or more series, to establish from time to
32
Table of Contents
time the number of shares to be included in each such series, to fix the rights, preferences and privileges of the shares of each wholly unissued series and any qualifications, limitations, or restrictions thereon and to increase or decrease the number of shares of any such series, but not below the number of shares of such series then outstanding.
The Company’s board of directors may authorize the issuance of preferred stock with voting or conversion rights that could adversely affect the voting power or other rights of the holders of common stock. The issuance of preferred stock, while providing flexibility in connection with possible future financings and acquisitions and other corporate purposes could, under certain circumstances, have the effect of restricting dividends on the Company’s common stock, diluting the voting power of the Company’s common stock, impairing the liquidation rights of the Company’s common stock or delaying, deferring or preventing a change in control of us, which might harm the market price of the Company’s common stock.
The Company’s board of directors will make any determination to issue such shares based on its judgment as to the Company’s best interests and the best interests of the Company’s stockholders. The Company has no shares of preferred stock outstanding and has no current plans to issue any shares of preferred stock.
Registration Rights
Certain holders of shares of the Company’s common stock are entitled to rights with respect to the registration of these securities under the Securities Act of 1933, as amended (the “Securities Act”). These rights are provided under the terms of an amended and restated investors’ rights agreement (the “Investors’ Rights Agreement”), by and among the Company and certain of its stockholders. These shares are collectively herein referred to as registrable securities.
The Investors’ Rights Agreement includes demand registration rights, short-form registration rights and piggyback registration rights. The demand, short form, and piggyback registration rights described below will generally expire on the fifth anniversary of the Company’s initial public offering, or with respect to any particular holder, at such time that such holder can sell its shares without limitation under Rule 144 of the Securities Act during any three-month period, or after the consummation of a liquidation event.
Demand registration rights
At any time after May 5, 2026, the holders of the Company’s registrable securities will be entitled to certain demand registration rights. Upon the request of holders of at least a majority of its outstanding registrable securities, the Company will be required to file a registration statement and effect the registration of these shares for public resale, unless our board of directors decides in good faith that such registration would be materially detrimental to the Company and its stockholders under the circumstances. The Company is required to effect up to two registrations pursuant to this provision of the Investors’ Rights Agreement.
Short form registration rights
The holders of the Company’s registrable securities are also entitled to short form registration rights. If the Company is eligible to file a registration statement on Form S-3, upon the request of holders of at least 20% of its outstanding registrable securities to sell registrable securities with an anticipated aggregate offering amount of at least $5.0 million net of certain expenses related to the offering, the Company will be required to effect a registration of such shares, unless our board of directors decides in good faith that such registration would be materially detrimental to the Company and its stockholders under the circumstances. The Company is required to effect up to two registrations in any twelve-month period pursuant to this provision of the Investors’ Rights Agreement.
33
Table of Contents
Piggyback registration rights
In the event that the Company proposes to, subject to limited exceptions, register any of its securities under the Securities Act, either for its own account or for the account of other security holders, the holders of the Company’s outstanding registrable securities are entitled to include their shares in the registration. Subject to certain exceptions contained in the Investors’ Rights Agreement, the Company and the underwriters may limit the number of shares included in the underwritten offering if the underwriters determine that marketing factors require a limitation of the number of shares to be underwritten.
Expenses of registration
The Company will pay the registration expenses, subject to certain limited exceptions contained in the Investors’ Rights Agreement, of the holders of the shares registered pursuant to the demand, short form and piggyback registration rights described above, including the expenses of one counsel for the selling holders.
Anti-Takeover Provisions
Delaware Takeover Statute
The Company is subject to the provisions of Section 203 of the DGCL. In general, Section 203 prohibits a publicly held Delaware corporation from engaging in a “business combination” with an “interested stockholder” for a three-year period following the time that this stockholder becomes an interested stockholder, unless the business combination is approved in a prescribed manner. Under Section 203, a business combination between a corporation and an interested stockholder is prohibited unless it satisfies one of the following conditions:
| • | before the stockholder became interested, the board of directors approved either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder; |
| • | upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, shares owned by persons who are directors and also officers and employee stock plans, in some instances, but not the outstanding voting stock owned by the interested stockholder; or |
| • | at or after the time the stockholder became interested, the business combination was approved by the board of directors and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least two-thirds of the outstanding voting stock which is not owned by the interested stockholder. |
Section 203 defines a business combination to include:
| • | any merger or consolidation involving the corporation and the interested stockholder; |
| • | any sale, transfer, lease, pledge, exchange, mortgage or other disposition involving the interested stockholder of 10% or more of the assets of the corporation; |
| • | subject to exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; |
| • | subject to exceptions, any transaction involving the corporation that has the effect of increasing the proportionate share of the stock of any class or series of the corporation beneficially owned by the interested stockholder; or |
| • | the receipt by the interested stockholder of the benefit of any loans, advances, guarantees, pledges or other financial benefits provided by or through the corporation. |
In general, Section 203 defines an interested stockholder as any entity or person beneficially owning 15% or more of the outstanding voting stock of the corporation and any entity or person affiliated with or controlling or controlled by the entity or person.
34
Table of Contents
Provisions of Amended and Restated Certificate of Incorporation and Amended and Restated Bylaws
Provisions of the Company’s restated certificate and restated bylaws may have the effect of delaying, deferring or discouraging another party from acquiring control of us and encouraging persons considering unsolicited tender offers or other unilateral takeover proposals to negotiate with the Company’s board of directors rather than pursue non-negotiated takeover attempts. These provisions include the items described below.
Board composition and filling vacancies. In accordance with the restated certificate, our board is divided into three classes serving staggered three-year terms, with one class being elected each year. The restated certificate also provides that directors may be removed only for cause and then only by the affirmative vote of the holders of or more of the shares then entitled to vote at an election of directors. Furthermore, any vacancy on the board of directors, however occurring, including a vacancy resulting from an increase in the size of the board, may only be filled by the affirmative vote of a majority of the directors then in office even if less than a quorum.
No written consent of stockholders. The restated certificate provides that all stockholder actions are required to be taken by a vote of the stockholders at an annual or special meeting and that stockholders may not take any action by written consent in lieu of a meeting. This limit may lengthen the amount of time required to take stockholder actions and would prevent the amendment of the restated bylaws or removal of directors by the Company’s stockholders without holding a meeting of stockholders.
Meetings of stockholders. The restated bylaws provide that only a majority of the members of the Company’s board of directors then in office may call special meetings of stockholders and only those matters set forth in the notice of the special meeting may be considered or acted upon at a special meeting of stockholders. The restated bylaws limit the business that may be conducted at an annual meeting of stockholders to those matters properly brought before the meeting.
Advance notice requirements. The restated bylaws establish advance notice procedures with regard to stockholder proposals relating to the nomination of candidates for election as directors or new business to be brought before meetings of the stockholders. These procedures provide that notice of stockholder proposals must be timely given in writing to the Company’s corporate secretary prior to the meeting at which the action is to be taken. Generally, to be timely, notice must be received at the Company’s principal executive offices not less than 90 days or more than 120 days prior to the first anniversary date of the annual meeting for the preceding year. The notice must contain certain information specified in the restated bylaws.
Amendment to certificate of incorporation and bylaws. As required by the DGCL, any amendment of the restated certificate of incorporation must first be approved by a majority of the board of directors and, if required by law or the restated certificate, must thereafter be approved by a majority of the outstanding shares entitled to vote on the amendment and a majority of the outstanding shares of each class entitled to vote thereon as a class, except that the amendment of the provisions relating to stockholder action, directors, limitation of liability and the amendment of the restated certificate must be approved by at least a majority of all of the outstanding shares of each class entitled to vote thereon voting together as a single class. The restated bylaws may be amended by the affirmative vote of a majority vote of the directors then in office, subject to any limitations set forth in the restated bylaws, and may also be amended by the affirmative vote of at least a majority of the outstanding shares entitled to vote on the amendment, voting together as a single class.
Undesignated preferred stock. The restated certificate provides for 10,000,000 authorized shares of preferred stock. The existence of authorized but unissued shares of preferred stock may enable the Company’s board of directors to render more difficult or to discourage an attempt to obtain control of the Company by means of a merger, tender offer, proxy contest or otherwise. The issuance of shares of preferred stock could decrease the amount of earnings and assets available for distribution to holders of shares of common stock. The issuance may also adversely affect the rights and powers, including voting rights, of these holders and may have the effect of delaying, deterring or preventing a change in control of us.
35
Table of Contents
Exclusive forum. The restated certificate of incorporation provides that the Court of Chancery of the State of Delaware shall be the sole and exclusive forum for any state law claims for: (i) any derivative action or proceeding brought on the Company’s behalf, (ii) any action asserting a claim of breach of a fiduciary duty owed by any of the directors or officers to the Company or its stockholders, (iii) any action asserting a claim against us arising pursuant to any provision of the DGCL or the certificate of incorporation or bylaws, (iv) any action to interpret, apply, enforce or determine the validity of the certificate of incorporation or bylaws or (v) any action asserting a claim against us governed by the internal affairs doctrine. This exclusive forum provision will not apply to any causes of action arising under the Securities Act or the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. Unless the Company consents in writing to the selection of an alternate forum, the United States District Court for the District of Delaware shall be the sole and exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act, as the Company is a Delaware corporation. Although the restated bylaws contain the choice of forum provision described above, it is possible that a court could rule that such provisions are inapplicable for a particular claim or action or that such provisions are unenforceable.
Transfer Agent and Registrar
The transfer agent and registrar for our common stock is Computershare Trust Company, N.A. The transfer agent and registrar’s address is 150 Royall Street, Canton, Massachusetts 02021.
Listing
Our common stock is listed on NYSE under the symbol “EVMN.”
36
Table of Contents
This prospectus covers the sale or other disposition by the Selling Stockholders of up to the total number of shares of our common stock that were issued to the Selling Stockholders in the Private Placement. For additional information regarding the issuance of the Shares in connection with the Private Placement, see the section “Prospectus Summary—Private Placement” above. The table below sets forth, to our knowledge, information concerning the beneficial ownership of shares of our common stock by the Selling Stockholders as of March 31, 2026. The information in the table below with respect to each Selling Stockholder has been obtained from the respective Selling Stockholder and the Selling Stockholders may have sold, transferred or otherwise disposed of all or a portion of their securities after the date on which they provided us with information regarding their securities. Throughout this prospectus, when we refer to the Shares being registered on behalf of the Selling Stockholders, we are referring to the shares of our common stock pursuant to the Purchase Agreement. The Selling Stockholders may sell all, some or none of the shares of common stock subject to this prospectus. See “Plan of Distribution” below as it may be supplemented and amended from time to time.
The number of shares of common stock beneficially owned prior to the offering for each Selling Stockholder includes all shares of our common stock beneficially held by such Selling Stockholder as of March 31, 2026, which includes all shares of our common stock purchased by such Selling Stockholder in the Private Placement.
Other than as stated above, beneficial ownership is determined in accordance with the rules of the SEC and includes voting or investment power with respect to our common stock. Generally, a person “beneficially owns” shares of our common stock if the person has or shares with others the right to vote those shares or to dispose of them, or if the person has the right to acquire voting or disposition rights within 60 days. The percentages of shares owned before and after the offering are based on 36,018,372 shares of common stock outstanding as of March 31, 2026, which includes the outstanding shares of common stock offered by this prospectus. The inclusion of any shares in this table does not constitute an admission of beneficial ownership for any Selling Stockholder named below.
| Shares of Common Stock Beneficially Owned Prior to Offering |
Number of Shares of Common Stock Being Offered(1) |
Shares of Common Stock to be Beneficially Owned After Offering(2) | ||||||||
| Name of Selling Stockholder |
Number | Percentage | Number | Percentage | ||||||
| LSP 7 Coöperatief UA(3) |
5,288,313 | 14.68 | 358,680 | 4,929,633 | 13.69 | |||||
| RA Capital Healthcare Fund, L.P.(4) |
3,494,638 | 9.70 | 35,868 | 3,458,770 | 9.60 | |||||
| Entities affiliated with RTW Investments, LP(5) |
1,463,011 | 4.06 | 179,340 | 1,283,671 | 3.56 | |||||
| Entities affiliated with MPM BioImpact LLC(6) |
1,437,562 | 3.99 | 358,680 | 1,078,882 | 3.00 | |||||
| Entities advised or subadvised by T. Rowe Price Associates, Inc.(7) |
1,219,207 | 3.38 | 369,459 | 849,748 | 2.36 | |||||
| Entities affiliated with Janus Henderson Investors US LLC(8) |
896,700 | 2.49 | 896,700 | — | — | |||||
| Entities affiliated with Marshall Wace(9) |
879,728 | 2.44 | 143,472 | 736,256 | 2.04 | |||||
| Kynam Global Healthcare Master Fund, LP(10) |
641,026 | 1.78 | 538,020 | 103,006 | * | |||||
| Perceptive Life Sciences Master Fund, Ltd.(11) |
448,350 | 1.24 | 448,350 | — | — | |||||
| Eventide Healthcare & Life Sciences Fund(12) |
269,010 | * | 269,010 | — | — | |||||
| StemPoint Capital Master Fund LP(13) |
216,687 | * | 89,670 | 127,017 | * | |||||
| Entities affiliated with Monashee Investment Management LLC(14) |
214,031 | * | 179,340 | 34,691 | * | |||||
| Entities affiliated with Aberdeen Investments(15) |
179,340 | * | 179,340 | — | * | |||||
| Defilade Capital Master Fund, L.P.(16) |
143,472 | 143,472 | — | * | ||||||
| Octagon Investments Master Fund LP(17) |
143,472 | * | 143,472 | — | — | |||||
| Entities affiliated with Sphera Management(18) |
117,234 | * | 35,868 | 81,366 | * | |||||
| Entities affiliated with Seven Grand Managers, LLC(19) |
89,670 | * | 89,670 | — | — | |||||
| Atlas Private Holdings (Cayman) Ltd.(20) |
35,868 | * | 35,868 | — | — | |||||
| * | Represents beneficial ownership of less than 1%. |
37
Table of Contents
| (1) | The number of shares of our common stock in the column “Number of Shares of Common Stock Being Offered” represents all of the shares of our common stock that a Selling Stockholder may offer and sell from time to time under this prospectus. |
| (2) | We do not know when or in what amounts a Selling Stockholder may offer shares for sale. The Selling Stockholders might not sell any or might sell all of the shares offered by this prospectus. Because the Selling Stockholders may offer all or some of the shares pursuant to this offering, and because there are currently no agreements, arrangements or understandings with respect to the sale of any of the shares, we cannot estimate the number of the shares that will be held by the Selling Stockholders after completion of the offering. However, for purposes of this table, we have assumed no further acquisitions of shares of common stock (or securities exercisable or convertible into common stock) by the Selling Stockholders and that, after completion of the offering, none of the shares covered by this prospectus will be held by the Selling Stockholders. |
| (3) | Consists of (i) 4,929,633 shares of common stock previously held and (ii) 358,680 shares of common stock purchased in the Private Placement. As the sole director of LSP 7 Coöperatief UA, LSP 7 Management BV may be deemed to beneficially own these securities. As managing directors of LSP 7 Management BV, each of Martijn Kleijwegt, René Kuijten and Joachim Rothe may also be deemed to beneficially own these securities. The business address of LSP 7 Coöperatief UA is Johannes Vermeerplein 9 1071 DV Amsterdam, Netherlands. |
| (4) | Consists of (i)(a) 2,674,425 shares of common stock previously held by RA Capital Healthcare Fund, L.P. (“RACHF”) and (b) 35,868 shares of common stock purchased in the Private Placement by RACHF, and (ii) 784,345 share of common stock previously held by RA Capital Nexus Fund III, L.P. (“Nexus” and together with RACHF, the “RA Funds”). RA Capital Management, L.P. (“RACM”) is the investment manager for the RA Funds. The general partner of RACHF is RA Capital Healthcare Fund GP, LLC and the general partner of Nexus is RA Capital Nexus Fund III GP, LLC. Peter Kolchinsky and Rajeev Shah are the managing members of such general partners. Each of RACM, RA Capital Healthcare Fund GP, LLC, RA Capital Nexus Fund III GP, LLC, Mr. Kolchinsky and Mr. Shah may be deemed to have voting and investment power over the shares held by the RA Funds. RACMRA Capital Healthcare Fund GP, LLC, RA Capital Nexus Fund III GP, LLC, Mr. Kolchinsky and Mr. Shah disclaim beneficial ownership of such shares, except to the extent of any pecuniary interest therein. The principal business address of the persons and entities listed above is 200 Berkeley Street, 18th Floor, Boston, MA 02116. |
| (5) | Consists of (i) (a) 600,237 shares of common stock held by RTW Biotech Opportunities Operating Ltd. and (b) 7,903 shares of common stock purchased in the Private Placement by RTW Biotech Opportunities Operating Ltd., (ii) (a) 311,829 shares of common stock held by RTW Innovation Master Fund, Ltd. and (b) 78,381 shares of our common stock purchased in the Private Placement by RTW Biotech Opportunities Operating Ltd., and (iii) (a) 371,605 shares of common stock held by RTW Master Fund, Ltd. and (b) 93,056 shares of our common stock purchased in the Private Placement by RTW Master Fund, Ltd. (collectively, the “RTW Funds”). RTW Investments, LP (“RTW”) in its capacity as the investment manager of the RTW Funds, has the power to vote and the power to direct the disposition of the shares held by the RTW Funds. Accordingly, RTW may be deemed to be the beneficial owner of such securities. Roderick Wong, M.D., as the Managing Partner of RTW, has the power to direct the vote and disposition of the securities held by RTW. Dr. Wong disclaims beneficial ownership of the shares held by the RTW Funds, except to the extent of his pecuniary interest therein. The address and principal office of RTW Investments, LP is 40 10th Avenue, Floor 7, New York, NY 10014, and the address of Dr. Wong and each of the RTW Funds is c/o RTW Investments, LP, 40 10th Avenue, Floor 7, New York, NY 10014. |
| (6) | Consists of (i) (a) 424,366 shares of common stock held by BioImpact Equities Fund, LP, and (b) 140,313 shares of common stock purchased in the Private Placement by BioImpact Equities Fund, LP, (ii) (a) 169,569 shares of common stock held by GTM Capital Healthcare Fund, L.P. and (b) 56,310 shares of our common stock purchased in the Private Placement by GTM Capital Healthcare Fund, L.P., and (iii) (a) 358,294 shares of common stock held by MPM BioImpact Innovations Equities Master, LP and (b) 119,536 shares of our common stock purchased in the Private Placement by MPM BioImpact Innovations Equities Master, LP, (iv) (a) 126,653 shares of common stock held by BioImpact Equities Master Fund, LP, and (b) 42,521 shares of our common stock purchased in the Private Placement by BioImpact Equities Master Fund, LP (collectively, the “MPM Funds”). MPM BioImpact LLC serves as the investment adviser or sub-adviser to the MPM Funds. Christiana Bardon is a Managing Partner of MPM BioImpact LLC, has sole or shared voting or dispositive power with respect to the securities held by the MPM Funds and as such may be deemed to beneficially own such securities. Ms. Bardon disclaims beneficial ownership of all ordinary shares held by the MPM Funds, other than to the extent of her pecuniary interest therein. The principal address of the MPM Funds, MPM BioImpact LLC and Ms. Bardon is 399 Boylston Street, Suite 1100, Boston, MA 02116. |
| (7) | Consists of (i)(a) 260,101 shares of common stock held by T. Rowe Price Health Sciences Fund, Inc. and (b) 125,397 shares of common stock purchased in the Private Placement by T. Rowe Price Health Sciences Fund, Inc., (ii)(a) 22,317 shares of common stock held by TD Mutual Funds – TD Health Sciences Fund and (b) 10,226 shares of common stock |
38
Table of Contents
| purchased in the Private Placement by TD Mutual Funds - TD Health Sciences Fund, (iii)(a) 16,349 shares of common stock held by T. Rowe Price Health Sciences Portfolio and (b) 7,566 shares of common stock purchased in the Private Placement by T. Rowe Price Health Sciences Portfolio, (iv)(a) 365,871 shares of common stock held by T. Rowe Price New Horizons Fund, Inc. and (b) 152,155 shares of common stock purchased in the Private Placement by T. Rowe Price New Horizons Fund, Inc., (v)(a) 173,393 shares of common stock held by T. Rowe Price New Horizons Trust and (b) 69,247 shares of common stock purchased in the Private Placement by T. Rowe Price New Horizons Trust, and (vi)(a) 11,717 shares of common stock held by T. Rowe Price U.S. Equities Trust and (b) 4,868 shares of common stock purchased in the Private Placement by T. Rowe Price U.S. Equities Trust. T. Rowe Price Health Associates, Inc., or TRPA, serves as investment adviser or subadviser, as applicable, with power to direct investments and/or sole power to vote the securities owned by these funds and accounts. For purposes of reporting requirements under the Exchange Act, TRPA may be deemed to be the beneficial owner of such securities; however, TRPA expressly disclaims that it is, in fact, the beneficial owner of such securities. TRPA is a wholly owned subsidiary of T. Rowe Price Group, Inc., which is a publicly traded financial services holding company. The principal business address of TRPA is 1307 Point Street, Baltimore, MD 21231. |
| (8) | Consists of (i) 155,843 shares of common stock purchased in the Private Placement by Janus Henderson Biotech Innovation Master Fund II Limited (“Master Fund”) and (ii) 740,857 shares of common stock purchased in the Private Placement by Janus Henderson Biotech Innovation Master Fund Limited (together with Master Fund, the “Janus Funds”). Janus Henderson Investors US LLC (“Janus”) is an investment adviser registered under the Investment Advisers Act of 1940, as amended, that acts as investment adviser for each of the Janus Funds. Janus has the ability to make decisions with respect to the voting and disposition of the securities directly held by the Janus Funds subject to the oversight of the board of directors of each of the Janus Funds. Under the terms of its management contract with each of the Janus Funds, Janus has overall responsibility for directing the investments of each of the Janus Funds in accordance with the investment objective, policies, and limitations of each of the Janus Funds. Each of the Janus Funds has one or more portfolio managers appointed by and serving at the pleasure of Janus, and the portfolio managers may be deemed to exercise voting and investment discretion with respect to securities directly held by the Janus Funds. The portfolio managers for each of the Janus Funds are Andrew Acker, Daniel S. Lyons and Agustin Mohedas. The business address of each of the aforementioned parties is 151 Detroit Street, Denver, CO 80206. |
| (9) | Consists of (i)(a) 736,256 shares of common stock held by MW XO Health Innovations Fund II (“MW Health Innovations Fund”) and (b) 23,912 shares of common stock purchased in the Private Placement by MW Health Innovations Fund, and (ii) 119,560 shares of common stock purchased in the Private Placement by Marshall Wace Investment Strategies - Eureka Fund (“MW Eureka Fund”). Marshall Wace LLP is the investment manager of MW Eureka Fund, and Marshall Wace North America L.P. is the investment manager of MW Health Innovations Fund. No individual has ultimate beneficial ownership of the shares owned by such stockholders. The address of Marshall Wace LLP is George House, 131 Sloane Street, London SW1X 9AT, United Kingdom and the address of Marshall Wace North America L.P. is 66 Hudson Blvd E., New York, NY 10001. |
| (10) | Consists of (i) 103,006 shares of common stock previously held and (ii) 538,020 shares of common stock purchased in the Private Placement. Yue Tang is the managing member of Kynam Global Healthcare Master Fund, LP and has the power to vote and dispose of the securities held. The principal business address of the entities and persons is 221 Elm Rd., Princeton NJ 08540. |
| (11) | Perceptive Advisors LLC (the “Advisor”) is the investment advisor of the Perceptive Life Sciences Master Fund, Ltd. (the “Master Fund”) and may be deemed to have beneficial ownership of the shares beneficially owned thereby. Joseph Edelman is the controlling person of the Advisor and accordingly, may be deemed to have beneficial ownership of the shares beneficially owned by the Master Fund and the Advisor. The Master Fund, Mr. Edelman and the Advisor disclaims beneficial ownership of all such securities except to the extent of his/its pecuniary interest therein. The address for Perceptive Life Sciences Master Fund, Ltd. and Perceptive Advisors LLC is 51 Astor Place, 10th Floor, New York, NY 10003. |
| (12) | Eventide Asset Management, LLC (“Eventide”) is the investment adviser to Eventide Healthcare & Life Sciences Fund (“Eventide Fund”) with respect to the shares directly held by the Eventide Fund. Eventide has sole voting and dispositive power over the shares, and Robin C. John and Finny Kuruvilla, M.D., Ph.D. each have shared voting and dispositive power over the shares. The business address of Eventide, Mr. John and Dr. Kuruvilla is One International Place, Suite 4210, Boston, Massachusetts 02110. |
| (13) | Consists of (i) 127,017 shares of common stock previously held and (ii) 89,670 shares of common stock purchased in the Private Statement. Michelle Ross may be deemed to beneficially own the shares held by StemPoint Capital Master Fund LP. The address of this entity is 520 Madison Avenue, 19th Floor, New York, NY 10022. |
| (14) | Consists of (i) (a) 14,125 shares of common stock held by HIF Solitude Ltd (“HIF”) and (b) 54,972 shares of common stock purchased in the Private Placement by HIF, (ii) (a) 6,400 shares of common stock held by Mission Pure Alpha |
39
Table of Contents
| Master LP (“MPAM”) and (b) 69,745 shares of common stock purchased in the Private Placement by MPAM, and (iii) (a) 14,166 shares of common stock held by Monashee Pure Alpha SPV I LP (“MPAS”) and (b) 54,623 shares of common stock purchased in the Private Placement by MPAS. HIF, MPAM, and MPAS are managed by Monashee Investment Management, LLC (“Monashee Management”). Jeff Muller is CCO of Monashee Management and has voting and investment control over Monashee Management and, accordingly, may be deemed to have beneficial ownership of the shares held by HIF, MPAM, and MPAS. Jeff Muller, however, disclaims any beneficial ownership of the shares held by these entities. The business address of HIF, MPAM, MPAS, and Mr. Muller is c/o Monashee Investment Management, LLC, 75 Park Plaza, 4th Floor, Boston, MA 02116. |
| (15) | Consists of (i) 121,035 shares of common stock purchased in the Private Placement by Abrdn Healthcare Investors and (ii) 58,305 shares of common stock purchased in the Private Placement by Abrdn Life Sciences Investors. The securities held by the foregoing stockholders may be deemed to be beneficially owned by Aberdeen Investments, which acts as each stockholders’ appointed advisor, and/or Jason Akus, the portfolio manager of such fund. Notwithstanding the foregoing, Aberdeen Investments, as advisor, and Mr. Akus disclaim beneficial ownership of the shares except to the extent of their respective pecuniary interest therein. The address of the selling stockholders is 28 State Street, 17th Floor, Boston MA 02110. |
| (16) | The address for Defilade Capital Master Fund, L.P. is 6001 Shellmound Street, Suite 625, Emeryville, CA 94608. |
| (17) | Octagon Capital Advisors LP is the investment advisor of Octagon Investments Master Fund LP. Ting Jia, as the managing member of Octagon Capital Advisors LP, may be deemed to beneficially own the shares of common stock held by Octagon Investments Master Fund LP. The address of this entity is 654 Madison Avenue, 21st Floor, New York, NY 10065. |
| (18) | Consists of (i) (a) 66,255 shares of common stock held by Sphera Biotech Master Fund LP and (b) 32,281 shares of common stock purchased in the Private Placement by Sphera Biotech Master Fund LP, and (ii) (a)15,111 shares of common stock held by Sphera Global Healthcare Master Fund and (b) 3,587 shares of common stock purchased in the Private Placement by Sphera Global Healthcare Master Fund. The securities held by the foregoing stockholders may be deemed to be beneficially owned by Sphera Global Healthcare Management LP (“Sphera Management”). Sphera Management disclaims beneficial ownership of such shares, except to the extent of its pecuniary interest therein. The address of Sphera Biotech Master Fund LP, Sphera Global Healthcare Master Fund, and Sphera Management is 4 Yitzhak Sadeh, Entrance A, 29th Floor, Tel Aviv 6777520, Israel. |
| (19) | Consists of (a) 27,907 shares of common stock purchased in the Private Placement by CM Sub-Advised Fund 2 Ltd, (b) 37,210 shares of common stock purchased in the Private Placement by Riverview Omni Master Fund LP, (c) 475 shares of common stock purchased in the Private Placement by Seven Grand Partners LLC, (d) 6,555 shares of common stock purchased in the Private Placement by SummitTX Master, SPC – SummitTX Alpha SP, (e) 6,820 Shares shares of common stock purchased in the Private Placement by SummitTX Master, SPC – SummitTX Apex SP, and (f) 10,703 shares of common stock purchased in the Private Placement by SummitTX Pinnacle Master, L.P (collectively, the “Seven Grand Entities”). The Seven Grand Entities are managed by Seven Grand Managers, LLC (“Seven Grand Managers”). The business address of the Seven Grand Entities and Seven Grand Managers is c/o Seven grand Managers, LLC, 81 Pondfield Road, Suite C302, Bronxville, NY 10708. |
| (20) | Balyasny Asset Management L.P. is the investment adviser of Atlas Private Holdings (Cayman) Ltd. Dmitry Balyasny, via intermediate entities, manages Balyasny Asset Management L.P. and has voting and investment control over the reported securities. The address for Atlas Private Holdings (Cayman) Ltd. and Balyasny Asset Management L.P. is 444 West Lake Street, 50th Floor, Chicago, IL 60606. |
Relationships with the Selling Stockholders
As discussed in greater detail above under the section titled “Prospectus Summary—Private Placement,” in February 2026, we entered into the Purchase Agreement with the Selling Stockholders, pursuant to which we sold and issued shares of our common stock and agreed to file a registration statement with the SEC to cover the resale by the Selling Stockholders of the shares of our common stock issued pursuant to the Purchase Agreement.
None of the Selling Stockholders has had a material relationship with us or any of our predecessors or affiliates within the past three years except as hereinafter described. Prior to the Private Placement, among the Selling Stockholders, LSP 7 Coöperatief UA and RA Capital Healthcare Fund, L.P. have been beneficial owners of more than 5% of our common stock. LSP 7 Coöperatief UA is an affiliate of Felice Verduyn-van Weegen, a member of our board of directors since September 2021. RA Capital Healthcare Fund, L.P. is an affiliate of Derek DiRocco, a member of our board of directors since October 2024.
40
Table of Contents
MATERIAL U.S. FEDERAL INCOME TAX CONSEQUENCES FOR NON-U.S. HOLDERS
The following is a summary of the material U.S. federal income tax consequences to non-U.S. holders (as defined below) of the ownership and disposition of the shares of our common stock to which this prospectus relates. This discussion is not a complete analysis of all potential U.S. federal income tax consequences relating thereto, does not address any tax consequences arising under any state, local or non-U.S. tax laws, any estate, gift or other U.S. federal tax laws other than income tax laws, and does not address the potential application of the Medicare contribution tax on net investment income, any alternative minimum tax, or the special tax accounting rules under Section 451(b) of the Internal Revenue Code of 1986, as amended (the “Code”). This discussion is based on the Code and applicable Treasury Regulations promulgated thereunder, judicial decisions, and published rulings and administrative pronouncements of the Internal Revenue Service (the “IRS”) all as in effect as of the date hereof. These authorities are subject to differing interpretations and may change, possibly retroactively, resulting in U.S. federal income tax consequences different from those discussed below. We have not requested nor do we intend to request a ruling from the IRS, or an opinion of counsel, with respect to the statements made and the conclusions reached in the following summary, and there can be no assurance that the IRS or a court will agree with such statements and conclusions.
This discussion is limited to non-U.S. holders that purchase the shares of our common stock to which this prospectus relates and that hold such shares of our common stock as a “capital asset” within the meaning of Section 1221 of the Code (generally, property held for investment). This discussion does not address all of the U.S. federal income tax consequences that may be relevant to a particular holder in light of such holder’s circumstances. This discussion also does not consider any specific facts or circumstances that may be relevant to holders subject to special rules under the U.S. federal income tax laws, including, without limitation:
| • | certain former citizens or long-term residents of the United States; |
| • | partnerships or other pass-through entities or arrangements (and investors therein); |
| • | “controlled foreign corporations” or “passive foreign investment companies;” |
| • | persons who have elected to mark securities to market; |
| • | corporations that accumulate earnings to avoid U.S. federal income tax; |
| • | banks, financial institutions, investment funds, insurance companies, or brokers or dealers in securities or foreign currencies; |
| • | tax-exempt organizations, governmental organizations, or tax-qualified retirement plans; |
| • | “qualified foreign pension funds” as defined in Section 897(l)(2) of the Code and entities all of the interests of which are held by qualified foreign pension funds; |
| • | persons that own, or have owned, actually or constructively, more than 5% of our common stock at any time (other than as expressly provided below); |
| • | persons that acquired our common stock pursuant to the exercise of warrants or conversion rights under convertible instruments; |
| • | persons who acquire our common stock through the exercise of options or otherwise as compensation for services; and |
| • | persons holding our common stock as part of a hedging or conversion transaction, straddle, synthetic security, constructive sale, or other risk reduction strategy or integrated investment. |
If a partnership (or an entity or arrangement that is treated as a partnership for U.S. federal income tax purposes) holds our common stock, the U.S. federal income tax treatment of a partner in the partnership will generally depend on the status of the partner, the activities of the partnership and certain determinations made at the partner level. Partnerships holding our common stock and the partners in such partnerships are urged to
41
Table of Contents
consult their tax advisors about the particular U.S. federal income tax consequences to them of holding and disposing of our common stock.
THIS DISCUSSION IS FOR INFORMATIONAL PURPOSES ONLY AND IS NOT TAX ADVICE. PROSPECTIVE INVESTORS SHOULD CONSULT THEIR TAX ADVISORS REGARDING THE PARTICULAR U.S. FEDERAL INCOME TAX CONSEQUENCES TO THEM OF ACQUIRING, OWNING AND DISPOSING OF OUR COMMON STOCK, AS WELL AS ANY TAX CONSEQUENCES ARISING UNDER ANY STATE, LOCAL OR NON-U.S. TAX LAWS AND ANY U.S. FEDERAL NON-INCOME TAX LAWS, OR UNDER ANY APPLICABLE INCOME TAX TREATY.
Definition of Non-U.S. Holder
For purposes of this discussion, a non-U.S. holder is any beneficial owner of our common stock that is neither a “U.S. holder” nor a partnership (including any entity or arrangement treated as a partnership or that is disregarded from its owner) for U.S. federal income tax purposes. A U.S. holder is any beneficial owner of our common stock that, for U.S. federal income tax purposes, is or is treated as any of the following:
| • | an individual who is a citizen or resident of the United States; |
| • | a corporation (or any entity treated as a corporation for U.S. federal income tax purposes) created or organized in or under the laws of the United States, any state thereof or the District of Columbia; |
| • | an estate, the income of which is subject to U.S. federal income tax regardless of its source; or |
| • | a trust (i) whose administration is subject to the primary supervision of a U.S. court, and which has one or more “United States persons” (within the meaning of Section 7701(a)(30) of the Code) that have the authority to control all substantial decisions of the trust, or (ii) that has a valid election in effect under applicable Treasury Regulations to be treated as a United States person. |
Distributions on Our Common Stock
As discussed under the “Dividend Policy” section of this prospectus, we have never declared or paid any cash dividends on our common stock, and we do not intend to pay cash dividends on our common stock for the foreseeable future. However, if we make cash or other property distributions on our common stock (other than certain pro rata distributions of our stock), such distributions will constitute dividends for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Any portion of a distribution that exceeds our current and accumulated earnings and profits will constitute a return of capital and will first be applied against and reduce the non-U.S. holder’s tax basis in our common stock, but not below zero. Any amount distributed in excess of tax basis will be treated as gain realized on the sale or other disposition of our common stock and will be treated as described in the section titled “Gain on Sale or Other Taxable Disposition of Our Common Stock” below.
Subject to the discussions below regarding effectively connected income, backup withholding and Sections 1471 through 1474 of the Code, the Treasury Regulations thereunder, and other official guidance relating thereto (commonly referred to as “FATCA”), dividends paid to a non-U.S. holder of our common stock generally will be subject to U.S. federal withholding tax at a rate of 30% of the gross amount of the dividends (or such lower rate specified by an applicable income tax treaty). To receive the benefit of a reduced treaty rate, a non-U.S. holder must furnish us or our paying agent with a valid IRS Form W-8BEN or IRS Form W-8BEN-E (or applicable successor form) and satisfy applicable certification and other requirements. This certification must be provided to us or our paying agent before the payment of dividends and must be updated periodically. If the non-U.S. holder holds our common stock through a financial institution or other agent acting on the non-U.S. holder’s behalf, the non-U.S. holder will be required to provide appropriate documentation to the applicable withholding agent, which then will be required to provide certification to us or our paying agent, either directly or through other intermediaries.
42
Table of Contents
Non-U.S. holders that do not provide the required certification on a timely basis, but that qualify for a reduced treaty rate, may obtain a refund of any excess amounts withheld by timely filing an appropriate claim for refund with the IRS. Non-U.S. holders should consult their tax advisors regarding their entitlement to benefits under any applicable income tax treaty.
If a non-U.S. holder holds our common stock in connection with the conduct of a trade or business in the United States, and dividends paid on our common stock are effectively connected with such non-U.S. holder’s U.S. trade or business (and are attributable to such non-U.S. holder’s permanent establishment in the United States if required by an applicable tax treaty), the non-U.S. holder generally will be exempt from U.S. federal withholding tax. To claim the exemption, the non-U.S. holder must generally furnish a valid IRS Form W-8ECI (or applicable successor form) to the applicable withholding agent.
However, any such effectively connected dividends paid on our common stock generally will be subject to U.S. federal income tax on a net income basis at the regular U.S. federal income tax rates in the same manner as if the non-U.S. holder were a resident of the United States. A non-U.S. holder that is a non-U.S. corporation also may be subject to an additional branch profits tax equal to 30% (or such lower rate specified by an applicable income tax treaty) of its effectively connected earnings and profits for the taxable year, as adjusted for certain items. Non-U.S. holders should consult their tax advisors regarding any applicable income tax treaties that may provide for different rules.
Gain on Sale or Other Taxable Disposition of Our Common Stock
Subject to the discussion below regarding backup withholding, a non-U.S. holder generally will not be subject to U.S. federal income tax on any gain realized on the sale or other taxable disposition of our common stock, unless:
| • | the gain is effectively connected with the non-U.S. holder’s conduct of a trade or business in the United States, and if required by an applicable income tax treaty, is attributable to a permanent establishment maintained by the non-U.S. holder in the United States; |
| • | the non-U.S. holder is a nonresident alien individual who is present in the United States for 183 days or more during the taxable year of the sale or other taxable disposition (as calculated pursuant to Section 7701(b) of the Code), and certain other requirements are met; or |
| • | our common stock constitutes a “United States real property interest” by reason of our status as a United States real property holding corporation (“USRPHC”) for U.S. federal income tax purposes at any time within the shorter of the five-year period preceding the sale or other taxable disposition or the non-U.S. holder’s holding period for our common stock. |
Determining whether we are a USRPHC depends on the fair market value of our U.S. real property interests relative to the fair market value of our worldwide real property interests and our other assets used or held for use in a trade or business. We believe that we are not currently, and do not anticipate becoming, a USRPHC for U.S. federal income tax purposes, although there can be no assurance in this regard. Even if we are or were to become a USRPHC, gain arising from the sale or other taxable disposition by a non-U.S. holder of our common stock will not be subject to U.S. federal income tax so long as (a) the non-U.S. holder owned, directly, indirectly, and constructively, no more than 5% of our common stock at all times within the shorter of (i) the five-year period preceding the disposition or (ii) the holder’s holding period and (b) our common stock is “regularly traded,” as defined by applicable Treasury Regulations, on an established securities market. There can be no assurance that our common stock will qualify as regularly traded on an established securities market. Prospective investors are encouraged to consult their own tax advisors regarding the possible consequences to them if we are, or were to become, a USRPHC.
43
Table of Contents
Gain described in the first bullet point above generally will be subject to U.S. federal income tax on a net income basis at the regular U.S. federal income tax rates in the same manner as if the non-U.S. holder were a resident of the United States. A non-U.S. holder that is a foreign corporation also may be subject to an additional branch profits tax equal to 30% (or such lower rate specified by an applicable income tax treaty) of its effectively connected earnings and profits for the taxable year, as adjusted for certain items.
A non-U.S. holder described in the second bullet point above will be subject to U.S. federal income tax at a flat 30% rate on gain realized upon the sale or other taxable disposition of our common stock which may be offset by certain U.S.-source capital losses (even though the individual is not considered a resident of the United States), provided that the non-U.S. holder has timely filed U.S. federal income tax returns with respect to such losses. Non-U.S. holders should consult their tax advisors regarding any applicable income tax treaties that may provide for different rules.
Information Reporting and Backup Withholding
Annual reports are required to be filed with the IRS and provided to each non-U.S. holder indicating the amount of distributions on our common stock paid to such non-U.S. holder and the amount of any tax withheld with respect to those distributions. These information reporting requirements apply even if no withholding was required (because the distributions were effectively connected with the non-U.S. holder’s conduct of a U.S. trade or business, or withholding was reduced or eliminated by an applicable income tax treaty) and regardless of whether such distributions constitute dividends. This information also may be made available under a specific treaty or agreement with the tax authorities in the country in which the non-U.S. holder resides or is established. Backup withholding, currently at a 24% rate, generally will not apply to payments to a non-U.S. holder of distributions on or the gross proceeds of a disposition of our common stock provided that the non-U.S. holder furnishes the required certification for its non-U.S. status, such as by providing a valid IRS Form W-8BEN, IRS Form W-8BEN-E or IRS Form W-8ECI, or certain other requirements are met. Backup withholding may apply if the payor has actual knowledge, or reason to know, that the non-U.S. holder is a United States person that is not an exempt recipient.
Backup withholding is not an additional tax. If any amount is withheld under the backup withholding rules, non-U.S. holders are urged to consult their U.S. tax advisors regarding the possibility of and procedure for obtaining a refund or a credit against the non-U.S. holder’s U.S. federal income tax liability, if any.
Withholding on Foreign Entities
FATCA imposes a U.S. federal withholding tax of 30% on certain payments made to a “foreign financial institution” (as specially defined under these rules) unless such institution enters into an agreement with the U.S. government to withhold on certain payments and to collect and provide to the U.S. tax authorities certain information regarding certain U.S. account holders of such institution (which includes certain equity and debt holders of such institution, as well as certain account holders that are foreign entities with U.S. owners) or an exemption applies. FATCA also generally imposes a U.S. federal withholding tax of 30% on certain payments made to a non-financial foreign entity unless such entity provides the withholding agent a certification identifying certain direct and indirect U.S. owners of the entity, or an exemption applies. An intergovernmental agreement between the United States and an applicable non-U.S. country may modify these requirements. Under certain circumstances, a non-U.S. holder might be eligible for refunds or credits of such taxes. FATCA currently applies to dividends paid on our common stock. The U.S. Treasury released proposed Treasury Regulations which, if finalized in their present form, would eliminate the U.S. federal withholding tax of 30% applicable to the gross proceeds of a sale or other disposition of our common stock. In its preamble to such proposed Treasury Regulations, the U.S. Treasury stated that taxpayers may generally rely on the proposed Treasury Regulations until final Treasury Regulations are issued.
Prospective investors are urged to consult with their tax advisors regarding the potential implications of FATCA on their investment in our common stock.
44
Table of Contents
EACH PROSPECTIVE INVESTOR SHOULD CONSULT ITS TAX ADVISOR REGARDING THE TAX CONSEQUENCES OF PURCHASING, HOLDING, AND DISPOSING OF OUR COMMON STOCK, INCLUDING THE CONSEQUENCES OF ANY RECENT AND PROPOSED CHANGE IN APPLICABLE LAW, AS WELL AS TAX CONSEQUENCES ARISING UNDER ANY STATE, LOCAL, NON-U.S. OR U.S. FEDERAL NON-INCOME TAX LAWS.
45
Table of Contents
The Selling Stockholders, which, as used herein, includes donees, pledgees, transferees or other successors-in-interest selling shares of common stock or interests in shares of common stock received after the date of this prospectus from a Selling Stockholder as a gift, pledge, partnership distribution or other transfer, may, from time to time, sell, transfer or otherwise dispose of any or all of their shares of common stock or interests in shares of common stock on any stock exchange, market or trading facility on which the shares are traded or in private transactions. These dispositions may be at fixed prices, at prevailing market prices at the time of sale, at prices related to the prevailing market price, at varying prices determined at the time of sale, or at negotiated prices.
The Selling Stockholders may use any one or more of the following methods when disposing of shares or interests therein:
| • | distributions to members, partners, stockholders or other equityholders of the Selling Stockholders; |
| • | ordinary brokerage transactions and transactions in which a broker-dealer solicits purchasers; |
| • | block trades in which a broker-dealer will attempt to sell the shares as agent, but may position and resell a portion of the block as principal to facilitate the transaction; |
| • | purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
| • | an exchange distribution in accordance with the rules of the applicable exchange; |
| • | privately negotiated transactions; |
| • | settlement of short sales effected after the date of the registration statement of which this prospectus is a part is declared effective by the SEC; |
| • | in transactions through broker-dealers that agree with the selling stockholders to sell a specified number of such securities at a stipulated price per security; |
| • | through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
| • | a combination of any such methods of sale; and |
| • | any other method permitted by applicable law. |
The Selling Stockholders may, from time to time, pledge or grant a security interest in some or all of the shares of common stock owned by them and, if they default in the performance of their secured obligations, the pledgees or secured parties may offer and sell the shares of common stock, from time to time, under this prospectus, or under an amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act, amending the list of Selling Stockholders to include the pledgee, transferee or other successors in interest as Selling Stockholders under this prospectus. The Selling Stockholders also may transfer the shares of common stock in other circumstances, in which case the transferees, pledgees, donees or other successors in interest will be the selling beneficial owners for purposes of this prospectus.
In connection with the sale of our common stock or interests therein, the Selling Stockholders may enter into hedging transactions with broker-dealers or other financial institutions, which may in turn engage in short sales of the common stock in the course of hedging the positions they assume. The Selling Stockholders may also sell shares of our common stock short and deliver these securities to close out their short positions, or loan or pledge the common stock to broker-dealers that in turn may sell these securities. The Selling Stockholders may also enter into option or other transactions with broker-dealers or other financial institutions or the creation of one or more derivative securities which require the delivery to such broker-dealer or other financial institution of shares offered by this prospectus, which shares such broker-dealer or other financial institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
46
Table of Contents
The aggregate proceeds to the Selling Stockholders from the sale of the common stock offered by them will be the purchase price of the common stock less discounts or commissions, if any. Each of the Selling Stockholders reserves the right to accept and, together with their respective agents from time to time, to reject, in whole or in part, any proposed purchase of common stock to be made directly or through agents. We will not receive any of the proceeds from an offering of securities under the prospectus.
The Selling Stockholders also may resell all or a portion of the shares in open market transactions in reliance upon Rule 144 under the Securities Act, provided that they meet the criteria and conform to the requirements of that rule, or another available exemption from the registration requirements under the Securities Act.
The Selling Stockholders and any underwriters, broker-dealers or agents that participate in the sale of the common stock or interests therein may be “underwriters” within the meaning of Section 2(a)(11) of the Securities Act (it being understood that the Selling Stockholders shall not be deemed to be underwriters solely as a result of their participation in this offering). Any discounts, commissions, concessions or profit they earn on any resale of the shares may be underwriting discounts and commissions under the Securities Act. Selling Stockholders who are “underwriters” within the meaning of Section 2(a)(11) of the Securities Act will be subject to the prospectus delivery requirements of the Securities Act.
To the extent required, the shares of our common stock to be sold, the names of the Selling Stockholders, the respective purchase prices and public offering prices, the names of any agent, dealer or underwriter, and any applicable commissions or discounts with respect to a particular offer will be set forth in an accompanying prospectus supplement or, if appropriate, a post-effective amendment to the registration statement that includes this prospectus.
In order to comply with the securities laws of some states, if applicable, the common stock may be sold in these jurisdictions only through registered or licensed brokers or dealers. In addition, in some states the common stock may not be sold unless it has been registered or qualified for sale or an exemption from registration or qualification requirements is available and is complied with.
We have advised the Selling Stockholders that the anti-manipulation rules of Regulation M under the Exchange Act may apply to sales of shares in the market and to the activities of the Selling Stockholders and their affiliates. In addition, to the extent applicable, we will make copies of this prospectus (as it may be supplemented or amended from time to time) available to the Selling Stockholders for the purpose of satisfying the prospectus delivery requirements of the Securities Act. The Selling Stockholders may indemnify any broker-dealer that participates in transactions involving the sale of the shares against certain liabilities, including liabilities arising under the Securities Act.
We have agreed to indemnify the Selling Stockholders against liabilities, including liabilities under the Securities Act and state securities laws, relating to the registration of the shares offered by this prospectus.
We have agreed with the Selling Stockholders to use commercially reasonable efforts to cause the registration statement of which this prospectus constitutes a part to become effective and to remain continuously effective until the earlier of: (i) the date on which the Selling Stockholders shall have resold or otherwise disposed of all the shares covered by this prospectus pursuant to Rule 144 or pursuant to this prospectus and (ii) the date on which the shares covered by this prospectus no longer constitute “Registrable Securities” as such term is defined in the Registration Rights Agreement, such that they may be resold by the Selling Stockholders without registration and without regard to any volume or manner-of-sale limitations and without current public information pursuant to Rule 144 under the Securities Act or any other rule of similar effect.
47
Table of Contents
The validity of the common stock offered hereby will be passed upon for us by Cooley LLP, Boston, Massachusetts.
The consolidated financial statements of Evommune, Inc. as of December 31, 2025 and 2024 and for the years then ended incorporated by reference in this Prospectus and in the Registration Statement have been so incorporated in reliance on the report of BDO USA, P.C., an independent registered public accounting firm, given on the authority of said firm as experts in auditing and accounting.
WHERE YOU CAN FIND ADDITIONAL INFORMATION
We have filed with the SEC a registration statement on Form S-1 under the Securities Act with respect to the shares of common stock being offered by this prospectus. This prospectus does not contain all of the information in the registration statement and its exhibits. For further information with respect to us and the common stock offered by this prospectus, we refer you to the registration statement and its exhibits. Statements contained in this prospectus as to the contents of any contract or any other document referred to are not necessarily complete, and, in each instance, we refer you to the copy of the contract or other document filed as an exhibit to the registration statement. Each of these statements is qualified in all respects by this reference.
You can read our SEC filings, including the registration statement, over the Internet at the SEC’s website at www.sec.gov. We are subject to the information reporting requirements of the Exchange Act, and we file reports, proxy statements and other information with the SEC. These reports, proxy statements and other information will be available via the SEC’s website at www.sec.gov. We also maintain a website at www.evommune.com, at which you may access these materials free of charge as soon as reasonably practicable after they are electronically filed with, or furnished to, the SEC. However, the information contained in or accessible through our website is not part of this prospectus or the registration statement of which this prospectus forms a part.
48
Table of Contents
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
The SEC allows us to “incorporate by reference” information that we file with it, which means that we can disclose important information to you by referring you to those documents. The information incorporated by reference is an important part of this prospectus. Information in this prospectus supersedes information incorporated by reference that we filed with the SEC prior to the date of this prospectus. We incorporate by reference into this prospectus and the registration statement of which this prospectus is a part the information or documents listed below that we have filed with or furnished to the SEC:
| • | Our Annual Report on Form 10-K for the fiscal year ended December 31, 2025, filed with the SEC on March 5, 2026; |
| • | Our Current Reports on Form 8-K filed with the SEC on February 10, 2026 and February 13, 2026 (each to the extent the information in such reports is filed and not furnished); and |
| • | The description of our common stock in our registration statement on Form 8-A12B filed with the SEC on November 4, 2025, including any amendments or reports filed for the purpose of updating such description, including Exhibit 4.3 to our Annual Report on Form 10-K for the fiscal year ended December 31, 2025. |
In addition, all documents subsequently filed by us (excluding any information furnished rather than filed) pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act, prior to the termination of the offering shall be deemed to be incorporated by reference into this prospectus.
We will provide to each person, including any beneficial owner, to whom a prospectus is delivered, without charge upon written or oral request, a copy of any or all of the documents that are incorporated by reference into this prospectus but not delivered with the prospectus, including exhibits which are specifically incorporated by reference into such documents. You should direct any requests for documents by writing us at 1891 Page Mill Road, Palo Alto, CA 94304, Attention: Corporate Secretary, or by telephoning us at (925) 247-4481. In addition, copies of the documents incorporated herein by reference may be accessed at our website at https://ir.evommune.com. The reference to our website address does not constitute incorporation by reference of the information contained on or accessible through our website, and you should not consider the contents of our website in making an investment decision with respect to our common stock.
In accordance with Rule 412 of the Securities Act, any statement contained in a document incorporated by reference herein shall be deemed modified or superseded to the extent that a statement contained herein or in any other subsequently filed document which also is or is deemed to be incorporated by reference herein modifies or supersedes such statement.
49
Table of Contents

Up to 4,494,279 Shares of Common Stock
PROSPECTUS
, 2026
Table of Contents
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
The following table sets forth all costs and expenses, other than underwriting discounts and commissions, payable by us in connection with the sale of the securities being registered. All amounts shown are estimates except for the registration fee of the Securities and Exchange Commission (“SEC”).
| Amount to Be Paid | ||||
| SEC registration fee |
$ | 16,230.26 | ||
| Legal fees and expenses |
150,000.00 | |||
| Accounting fees and expenses |
20,000.00 | |||
| Miscellaneous expenses |
50,000.00 | |||
|
|
|
|||
| Total |
$ | 236,230.26 | ||
|
|
|
|||
Discounts, concessions, commissions and similar selling expenses attributable to the sale of shares of our common stock covered by this prospectus will be borne by the selling stockholders. We will pay all expenses (other than discounts, concessions, commissions and similar selling expenses) relating to the registration of the shares with the SEC, as estimated in the table above.
Item 14. Indemnification of Directors and Officers.
Section 145 of the Delaware General Corporation Law (the “DGCL”) authorizes a corporation to indemnify its directors and officers against liabilities arising out of actions, suits and proceedings to which they are made or threatened to be made a party by reason of the fact that they have served or are currently serving as a director or officer to a corporation. The indemnity may cover expenses (including attorneys’ fees) judgments, fines and amounts paid in settlement actually and reasonably incurred by the director or officer in connection with any such action, suit or proceeding. Section 145 permits corporations to pay expenses (including attorneys’ fees) incurred by directors and officers in advance of the final disposition of such action, suit or proceeding. In addition, Section 145 provides that a corporation has the power to purchase and maintain insurance on behalf of its directors and officers against any liability asserted against them and incurred by them in their capacity as a director or officer or arising out of their status as such, whether or not the corporation would have the power to indemnify the director or officer against such liability under Section 145.
We have adopted provisions in our amended and restated certificate of incorporation and amended and restated bylaws (our “bylaws”) that limit or eliminate the personal liability of our directors to the fullest extent permitted by the DGCL, as it now exists or may in the future be amended. Consequently, a director will not be personally liable to us or our stockholders for monetary damages or breach of fiduciary duty as a director, except for liability for any breach of the director’s duty of loyalty to us or our stockholders, any act or omission not in good faith or that involves intentional misconduct or a knowing violation of law, any unlawful payments related to dividends or unlawful stock purchases, redemptions or other distributions or any transaction from which the director derived an improper personal benefit.
These limitations of liability do not alter director liability under the federal securities laws and do not affect the availability of equitable remedies such as an injunction or rescission.
In addition, our bylaws provide that:
| • | we will indemnify our directors, officers and, in the discretion of our board of directors, certain employees to the fullest extent permitted by the DGCL, as it now exists or may in the future be amended; and |
II-1
Table of Contents
| • | we will advance reasonable expenses, including attorneys’ fees, to our directors and, in the discretion of our board of directors, to our officers and certain employees, in connection with legal proceedings relating to their service for or on behalf of us, subject to limited exceptions. |
We have entered into indemnification agreements with each of our directors (and their affiliated funds), executive officers and other officers as determined from time to time by our board of directors or our Compensation Committee. These agreements provide that we will indemnify each of our directors (and their affiliated funds) and officers with whom we have entered into indemnification agreements to the fullest extent permitted by Delaware law. We will advance expenses, including attorneys’ fees (but excluding judgments, fines and settlement amounts), to each indemnified director (and their affiliated funds) or officer in connection with any proceeding in which indemnification is available and we will indemnify our directors and officers for certain actions or proceedings arising out of that person’s services as a director or officer brought on behalf of us or in furtherance of our rights. Additionally, certain of our directors or officers may have certain rights to indemnification, advancement of expenses or insurance provided by their affiliates or other third parties, which indemnification relates to and might apply to the same proceedings arising out of such director’s or officer’s services as a director referenced herein. Nonetheless, we will agree in the indemnification agreements that our obligations to those same directors or officers are primary and any obligation of such affiliates or other third parties to advance expenses or to provide indemnification for the expenses or liabilities incurred by those directors are secondary.
We maintain general liability insurance which covers certain liabilities of our directors and officers arising out of claims based on acts or omissions in their capacities as directors or officers, including liabilities under the Securities Act of 1933, as amended (the “Securities Act”).
Item 15. Recent Sales of Unregistered Securities.
In the three years preceding the filing of this registration statement, we have issued the following securities that were not registered under the Securities Act. No underwriters were involved in the sales and the certificates representing the securities sold and issued contain legends restricting transfer of the securities without registration under the Securities Act or an applicable exemption from registration.
(a) Issuances of Capital Stock
From January 2022 to February 2023, we issued an aggregate of 14,279,809 shares of our Series A Preferred Stock, par value $0.0001 per share (“Series A Preferred Stock”), to certain accredited investors for gross proceeds of approximately $27.6 million at a purchase price of $1.9363 per share. Of those, 180,756 shares of Series A Preferred Stock were subsequently converted into the shares of our common stock.
From March to June 2023, we issued an aggregate of 28,750,000 shares of our Series B Preferred Stock, par value $0.0001 per share, to certain accredited investors for gross proceeds of approximately $57.5 million at a purchase price of $2.00 per share.
In October 2024 and June 2025, we issued an aggregate of 72,435,110 shares of our Series C Preferred Stock, par value $0.0001 per share, to certain accredited investors for gross proceeds of approximately $115.5 million at a purchase price of $1.59453 per share.
(b) Grants and Exercises of Stock Options
Since January 1, 2023, we have granted options to purchase an aggregate of 5,348,894 shares of our common stock with a weighted-average exercise price of $10.50 per share, to certain employees, directors and consultants in connection with services provided to us by such persons pursuant to the 2020 Plan. Through the effective date of this registration statement, we have issued an aggregate of 1,833 shares of our common stock upon exercise of such stock options for aggregate consideration of $3,134.
II-2
Table of Contents
(c) Private Placement
On February 12, 2026, we entered into a securities purchase agreement (the “Purchase Agreement”), with certain selling stockholders named in this prospectus, pursuant to which we sold and issued to them in a private placement an aggregate of 4,494,279 shares of our common stock, par value $0.0001 per share, at a purchase price of $27.88 per share.
None of the foregoing transactions involved any underwriters, underwriting discounts or commissions, or any public offering. Unless otherwise specified above, we believe these transactions were exempt from registration under the Securities Act in reliance on Section 4(a)(2) of the Securities Act (and Regulation D or Regulation S promulgated thereunder) or Rule 701 promulgated under Section 3(b) of the Securities Act as transactions by an issuer not involving any public offering or under benefit plans and contracts relating to compensation as provided under Rule 701. The recipients of the securities in each of these transactions represented their intentions to acquire the securities for investment only and not with a view to or for sale in connection with any distribution thereof, and appropriate legends were placed on the share certificates issued in these transactions. All recipients had adequate access, through their relationships with us, to information about us. The sales of these securities were made without any general solicitation or advertising.
Item 16. Exhibits and Financial Statement Schedules.
| (a) | Exhibits |
The exhibits listed below are filed as part of this registration statement.
| Incorporated by Reference | ||||||||||||
| Exhibit Number |
Description |
Form | File No. | Exhibit | Filing Date | Filed Herewith | ||||||
| 3.1 | Amended and Restated Certificate of Incorporation of Registrant. | 8-K | 001-42938 | 3.1 | 11/7/2025 | |||||||
| 3.2 | Amended and Restated Bylaws of the Registrant. | S-1 | 333-290793 | 3.4 | 10/9/2025 | |||||||
| 4.1 | Form of Common Stock Certificate of the Registrant. | S-1 | 333-290793 | 4.1 | 10/9/2025 | |||||||
| 4.2† | Third Amended and Restated Investors’ Rights Agreement, dated October 30, 2024, by and among the Registrant and the investors party thereto. | S-1 | 333-290793 | 4.2 | 10/9/2025 | |||||||
| 5.1 | Opinion of Cooley LLP. | X | ||||||||||
| 10.1+ | Form of Indemnification Agreements. | S-1 | 333-290793 | 10.1 | 10/9/2025 | |||||||
| 10.2+ | Evommune, Inc. 2020 Stock Plan. | S-1 | 333-290793 | 10.2(a) | 10/9/2025 | |||||||
| 10.3+ | Forms of Stock Option Grant Notice, Option Agreement, Notice of Exercise, Restricted Stock Unit Agreement and Restricted Stock Unit Grant Notice under the 2020 Plan. | S-1 | 333-290793 | 10.2(b) | 10/9/2025 | |||||||
| 10.4+ | Evommune, Inc. 2025 Equity Incentive Plan. | S-8 | 333-291386 | 99.3 | 11/7/2025 | |||||||
II-3
Table of Contents
II-4
Table of Contents
| Incorporated by Reference | ||||||||||||
| Exhibit Number |
Description |
Form | File No. | Exhibit | Filing Date | Filed Herewith | ||||||
| 10.19† | Lease Agreement, dated June 29, 2025, by and between the Registrant and Hudson Page Mill Hill, LLC. | S-1 | 333-290793 | 10.17 | 10/9/2025 | |||||||
| 10.20# | Form of Securities Purchase Agreement relating to the Private Placement. | 8-K | 001-42938 | 10.1 | 2/12/2026 | |||||||
| 10.21# | Form of Registration Rights Agreement, dated February 12, 2026, by and among the Registrant and the investors thereunder. | 8-K | 001-42938 | 10.2 | 2/12/2026 | |||||||
| 21.1 | List of Subsidiaries of the Registrant. | 10-K | 001-42938 | 21.1 | 3/5/2026 | |||||||
| 23.1 | Consent of BDO USA, P.C., independent registered public accounting firm. | X | ||||||||||
| 23.2 | Consent of Cooley LLP (included in Exhibit 5.1). | X | ||||||||||
| 24.1 | Power of Attorney (included on signature page). | X | ||||||||||
| 107 | Filing Fee Table. | X | ||||||||||
| + | Indicates a management contract or any compensatory plan, contract or arrangement. |
| † | Certain schedules and exhibits have been omitted pursuant to Item 601(a)(5) of Regulation S-K. A copy of any omitted schedule and/or exhibit will be furnished to the SEC upon request. |
| # | Portions of this exhibit have been omitted in accordance with the rules of the SEC. |
(b) Financial Statements Schedules
None.
(c) Filing Fee Table
The information required to be furnished by paragraph (c) of this Item is incorporated herein by reference to Exhibit 107.
Item 17. Undertakings.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
II-5
Table of Contents
The undersigned hereby undertakes that:
| (1) | For purposes of determining any liability under the Securities Act, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. |
| (2) | For the purpose of determining any liability under the Securities Act, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
II-6
Table of Contents
Pursuant to the requirements of the Securities Act of 1933, as amended, the Registrant has duly caused this Registration Statement on Form S-1 to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Palo Alto, California, on the 17th day of April, 2026.
| EVOMMUNE, INC. | ||
| By: | /s/ Luis Peña | |
| Luis Peña Chief Executive Officer and President | ||
POWER OF ATTORNEY
KNOW ALL BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Luis Peña and Kyle Carver and each of them, as his or her true and lawful attorney-in-fact and agents, each with the full power of substitution, for him or her and in his or her name, place or stead, in any and all capacities, to sign any and all amendments to this registration statement (including post-effective amendments), and to sign any registration statement for the same offering covered by this registration statement that is to be effective upon filing pursuant to Rule 462(b) promulgated under the Securities Act, and all post-effective amendments thereto, and to file the same, with exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the premises, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorney-in-fact and agents, or their or his substitute or substitutes, may lawfully do or cause to be done by virtue hereof
Pursuant to the requirements of the Securities Act of 1933, as amended, this Registration Statement has been signed by the following persons in the capacities and on the 17th day of April, 2026.
| Signature |
Title |
Date | ||
| /s/ Luis Peña Luis Peña |
President, Chief Executive Officer and Director (Principal Executive Officer) |
April 17, 2026 | ||
| /s/ Kyle Carver Kyle Carver, M.B.A., C.P.A. |
Chief Financial Officer (Principal Financial and Accounting Officer) |
April 17, 2026 | ||
| /s/ Benjamin F. McGraw Benjamin F. McGraw, III, Pharm.D. |
Chairman | April 17, 2026 | ||
| /s/ Eugene A. Bauer Eugene A. Bauer, M.D. |
Director | April 17, 2026 | ||
| /s/ David E. Cohen David E. Cohen, M.D., M.P.H. |
Director | April 17, 2026 | ||
| /s/ Derek DiRocco Derek DiRocco, Ph.D. |
Director | April 17, 2026 | ||
| /s/ Arthur Kirsch Arthur Kirsch |
Director | April 17, 2026 | ||
II-7
Table of Contents
| Signature |
Title |
Date | ||
| /s/ Rob Hopfner Rob Hopfner, R.Ph., Ph.D. |
Director | April 17, 2026 | ||
| /s/ Felice Verduyn-van Weegen Felice Verduyn-van Weegen |
Director | April 17, 2026 | ||
II-8