EX-99.2
Published on March 5, 2026

March 2026 © Evommune, Inc. Corporate Presentation

Disclaimers © Evommune, Inc. This presentation has been prepared by Evommune, Inc. (“we”, “us” or “our”) and contains forward-looking statements, including: statements about our expectations regarding the potential benefits, clinical activity and tolerability of our product candidates; our expectations with regard to the results of our clinical trials, preclinical studies and research and development programs, including the the potential therapeutic benefit of EVO756 and EVO301, the design, objectives, initiation, timing, progress and results of current and future preclinical studies and clinical trials of our product candidates, including the ongoing Phase 2 clinical trials for EVO756 and EVO301; anticipated cash runway; and continued advancement of our portfolio. These statements involve substantial known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements we make. These and other risks are described more fully in our Annual Report on Form 10-K for the year ended December 31, 2025 and our other filings with the Securities and Exchange Commission (the “SEC”) and our other documents subsequently filed with or furnished to the SEC. All forward-looking statements represent our views as of the date of this presentation. All forward-looking statements contained in this presentation speak only as of the date on which they were made. Except to the extent required by law, we undertake no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made. This presentation also contains estimates made by independent parties relating to industry market size and other data. These estimates involve a number of assumptions and limitations, and you are cautioned not to give undue weight on such estimates. We have not independently verified the accuracy or completeness of such information and we do not take any responsibility with the accuracy or completeness of such information. The trademarks included in this presentation are the property of the owners thereof and are used for reference purposes only.

Evommune (EVMN) is Addressing Chronic Inflammation, a Global Healthcare Crisis 5 Substantial Burden on the Healthcare System Annual direct cost of at least $90B2 Existing Treatment Options Have Critical Limitations Current therapies fail to deliver efficacy and safety suitable for the majority of patients Chronic Inflammation Destroys Lives Contributes to 3 out of 5 deaths worldwide1 Experienced Team Distinct Mechanisms Portfolio Approach Evommune is Delivering Next Generation Therapies Footnotes: https://www.ncbi.nlm.nih.gov/books/NBK493173/ Wylezinski LS, et al. J Clin Med. 2019;8:493 © Evommune, Inc.
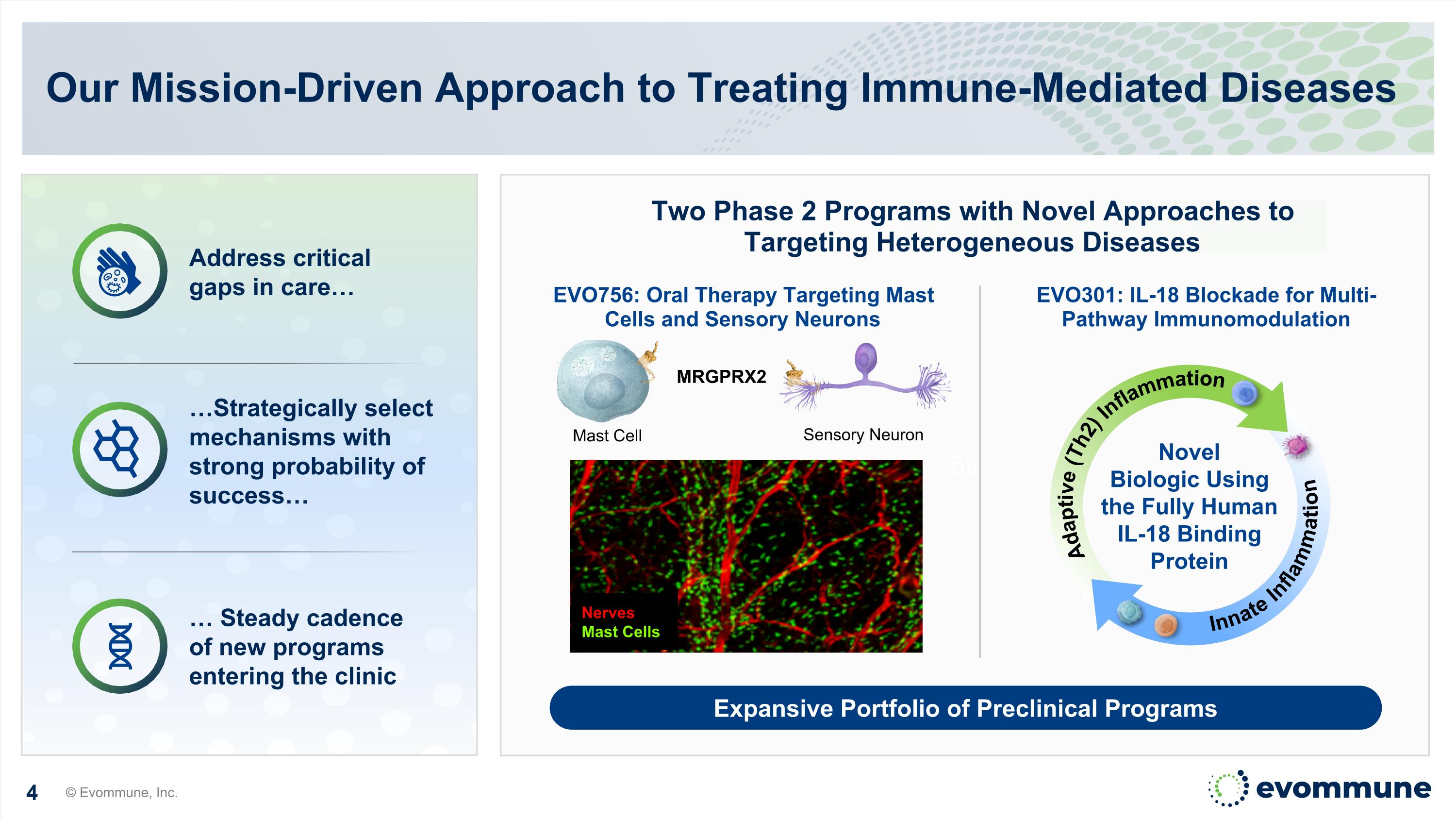
5v Our Mission-Driven Approach to Treating Immune-Mediated Diseases © Evommune, Inc. Two Phase 2 Programs with Novel Approaches to Targeting Heterogeneous Diseases EVO756: Oral Therapy Targeting Mast Cells and Sensory Neurons EVO301: IL-18 Blockade for Multi-Pathway Immunomodulation Address critical gaps in care… Expansive Portfolio of Preclinical Programs Sensory Neuron Mast Cell MRGPRX2 Nerves Mast Cells Novel Biologic Using the Fully Human IL-18 Binding Protein Adaptive (Th2) Inflammation Innate Inflammation …Strategically select mechanisms with strong probability of success… … Steady cadence of new programs entering the clinic
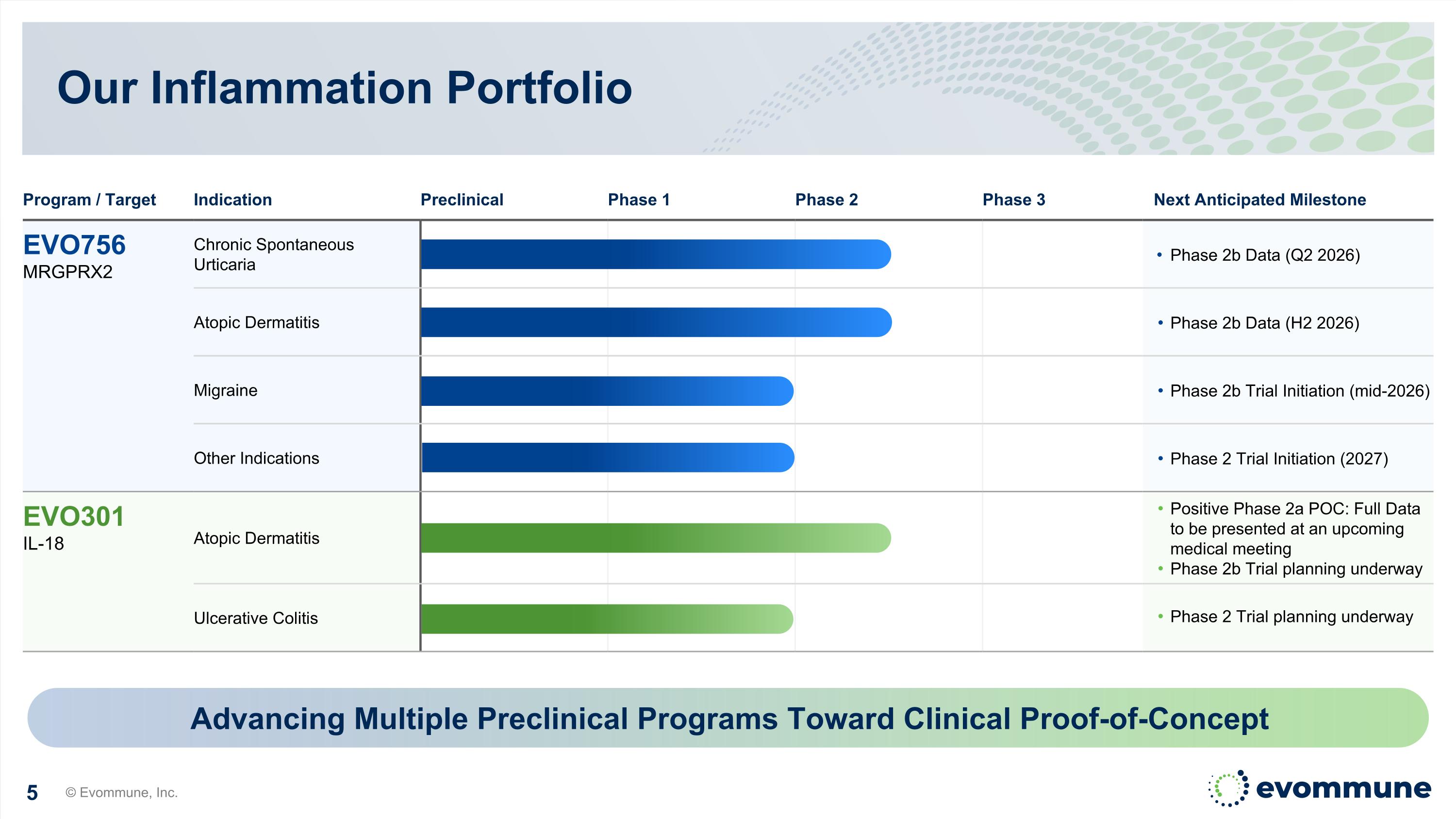
Our Inflammation Portfolio © Evommune, Inc. Program / Target Indication Preclinical Phase 1 Phase 2 Phase 3 Next Anticipated Milestone EVO756 MRGPRX2 Chronic Spontaneous Urticaria Phase 2b Data (Q2 2026) Atopic Dermatitis Phase 2b Data (H2 2026) Migraine Phase 2b Trial Initiation (mid-2026) Other Indications Phase 2 Trial Initiation (2027) EVO301 IL-18 Atopic Dermatitis Positive Phase 2a POC: Full Data to be presented at an upcoming medical meeting Phase 2b Trial planning underway Ulcerative Colitis Phase 2 Trial planning underway Advancing Multiple Preclinical Programs Toward Clinical Proof-of-Concept

EVO756: Oral MRGPRX2 Antagonist Targeted Approach to Controlling Mast Cell Mediated Diseases and Neuroinflammation © Evommune, Inc.
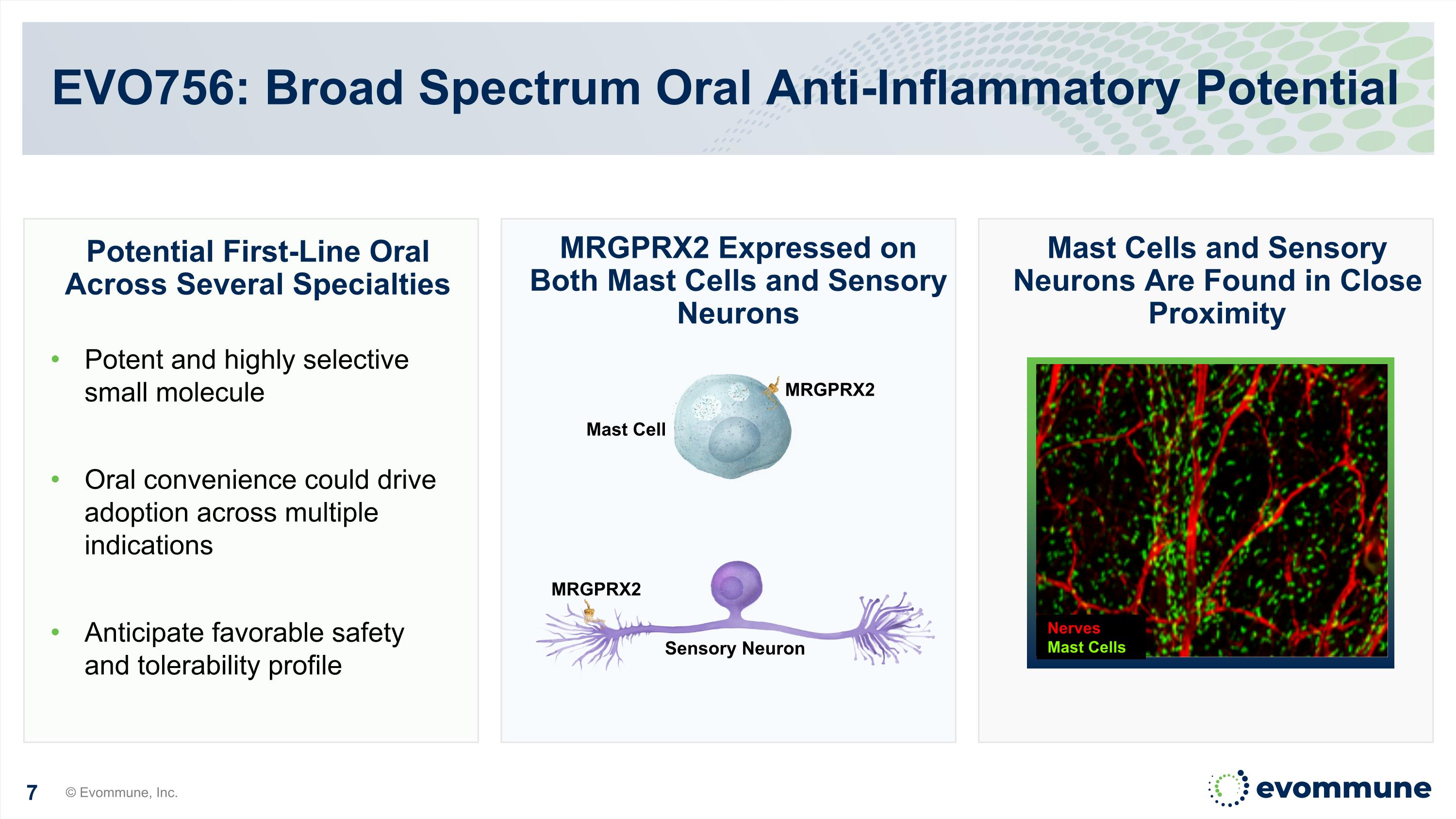
EVO756: Broad Spectrum Oral Anti-Inflammatory Potential MRGPRX2 Expressed on Both Mast Cells and Sensory Neurons Potent and highly selective small molecule Oral convenience could drive adoption across multiple indications Anticipate favorable safety and tolerability profile Potential First-Line Oral Across Several Specialties Sensory Neuron Mast Cell MRGPRX2 Nerves Mast Cells Mast Cells and Sensory Neurons Are Found in Close Proximity MRGPRX2 © Evommune, Inc.
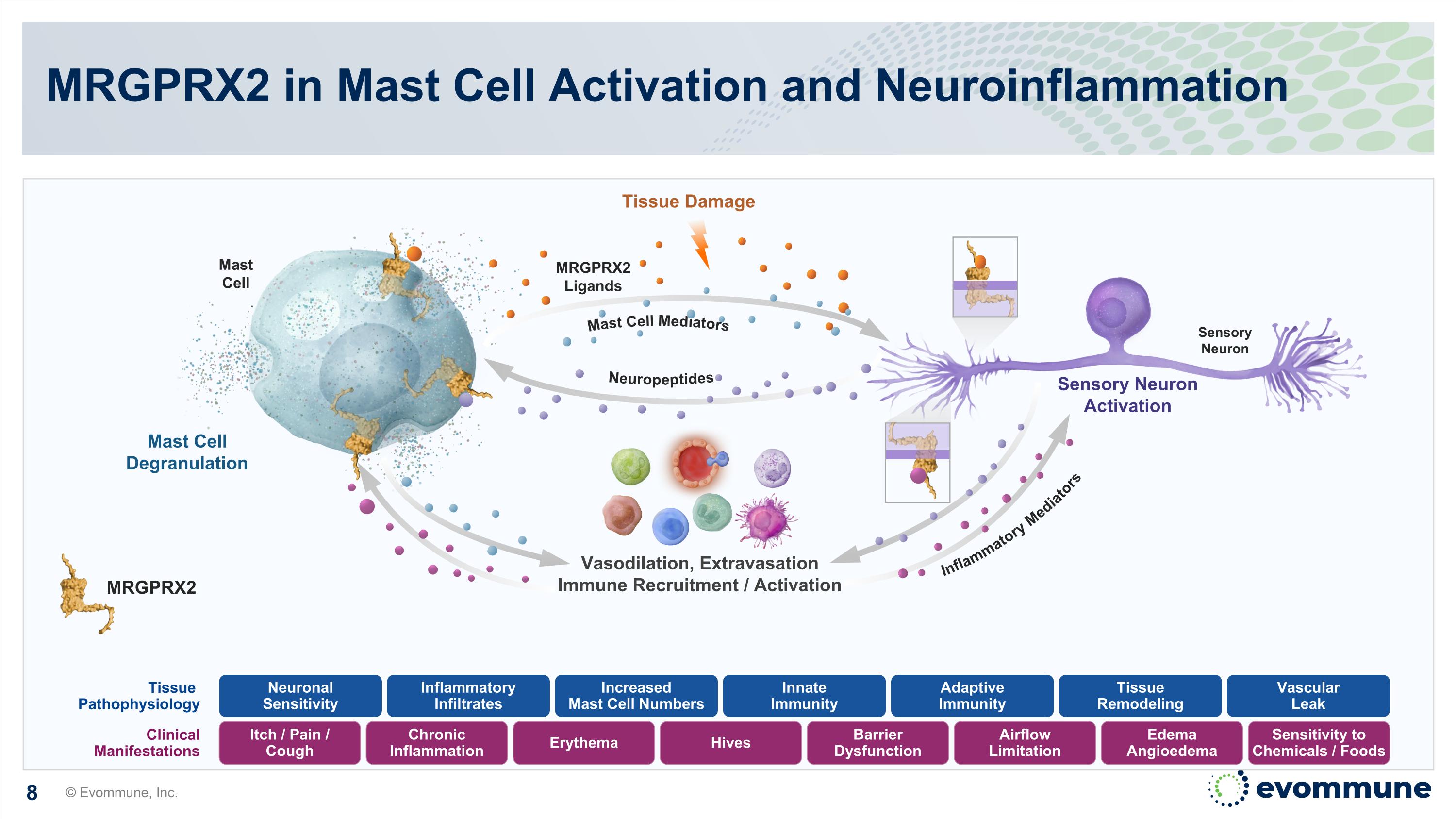
Mast Cell MRGPRX2 Ligands Tissue Damage Sensory Neuron Vasodilation, Extravasation Immune Recruitment / Activation Sensory Neuron Activation Mast Cell Degranulation Itch / Pain / Cough Tissue Pathophysiology Clinical Manifestations Inflammatory Infiltrates Increased Mast Cell Numbers Innate Immunity Adaptive Immunity Tissue Remodeling Vascular Leak Neuronal Sensitivity Chronic Inflammation Erythema Hives Barrier Dysfunction Airflow Limitation Edema Angioedema Sensitivity to Chemicals / Foods MRGPRX2 Neuropeptides Mast Cell Mediators Inflammatory Mediators MRGPRX2 in Mast Cell Activation and Neuroinflammation © Evommune, Inc.
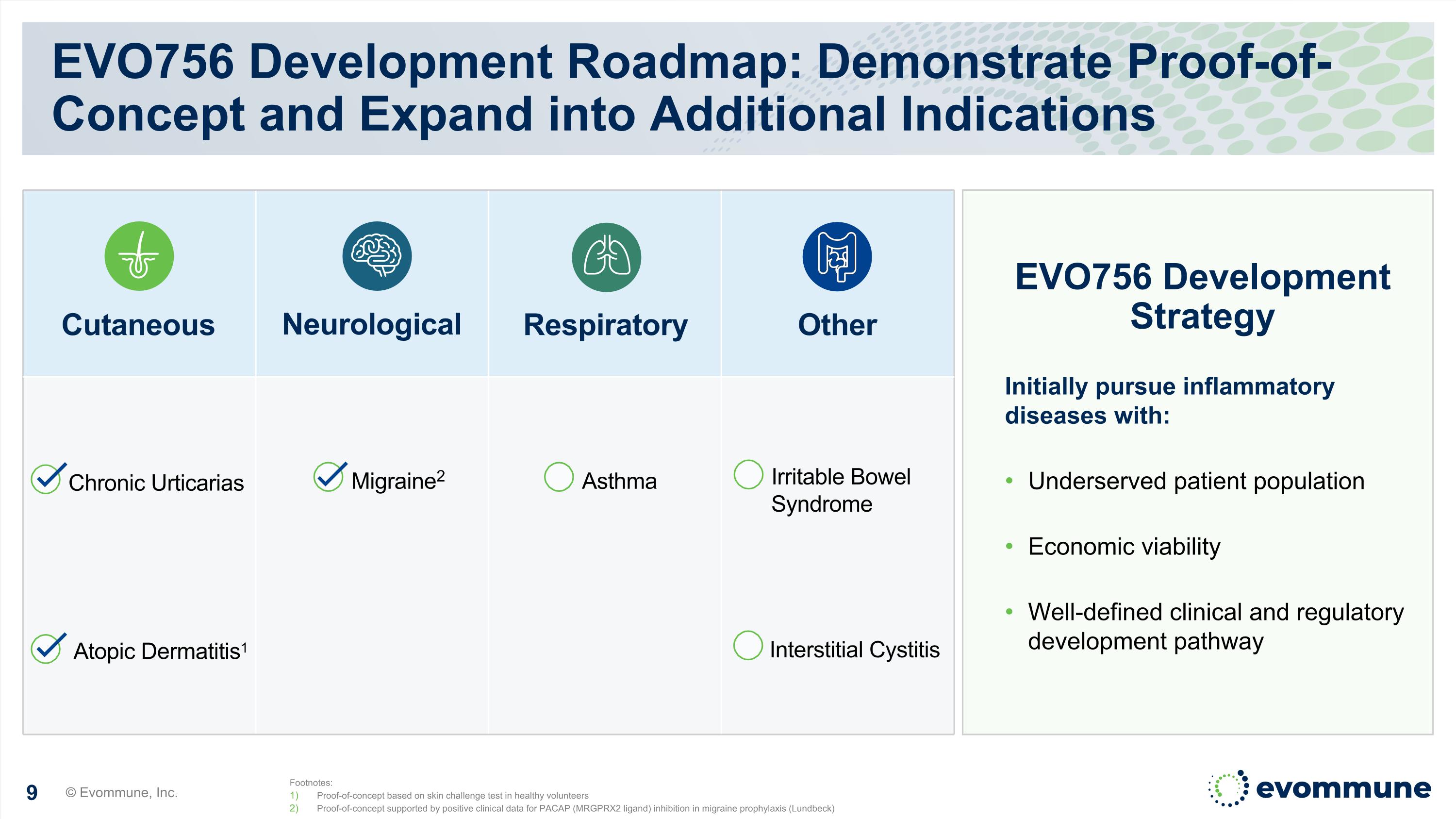
EVO756 Development Roadmap: Demonstrate Proof-of-Concept and Expand into Additional Indications Initially pursue inflammatory diseases with: Underserved patient population Economic viability Well-defined clinical and regulatory development pathway Cutaneous Neurological Respiratory Other EVO756 Development Strategy Footnotes: Proof-of-concept based on skin challenge test in healthy volunteers Proof-of-concept supported by positive clinical data for PACAP (MRGPRX2 ligand) inhibition in migraine prophylaxis (Lundbeck) Migraine2 Asthma Chronic Urticarias Atopic Dermatitis1 Irritable Bowel Syndrome Interstitial Cystitis © Evommune, Inc.

10 EVO756 Clinical Data © Evommune, Inc. Dual Mechanism Modulates Both Mast Cells and Peripheral Sensory Neurons

EVO756: Encouraging Results in Two Clinical Trials Support Initial Development in CSU and AD © Evommune, Inc. CIndU = Chronic inducible urticaria; CSU = Chronic spontaneous urticaria; AD = Atopic dermatitis EVO756 Clinical Development Summary Trial Phase 1 Proof-of-Concept Phase 2 Phase 2b Phase 2b N 132 30 ~160 ~120 Indication Healthy Volunteers CIndU CSU AD Key Takeaways Well-tolerated across all doses Clear target engagement in skin challenge Concentration dose proportional and linear Well-tolerated across all doses Complete responses as early as week 1 POC achieved after just 4 weeks of dosing Topline Data Expected Q2 2026 Topline Data Expected H2 2026
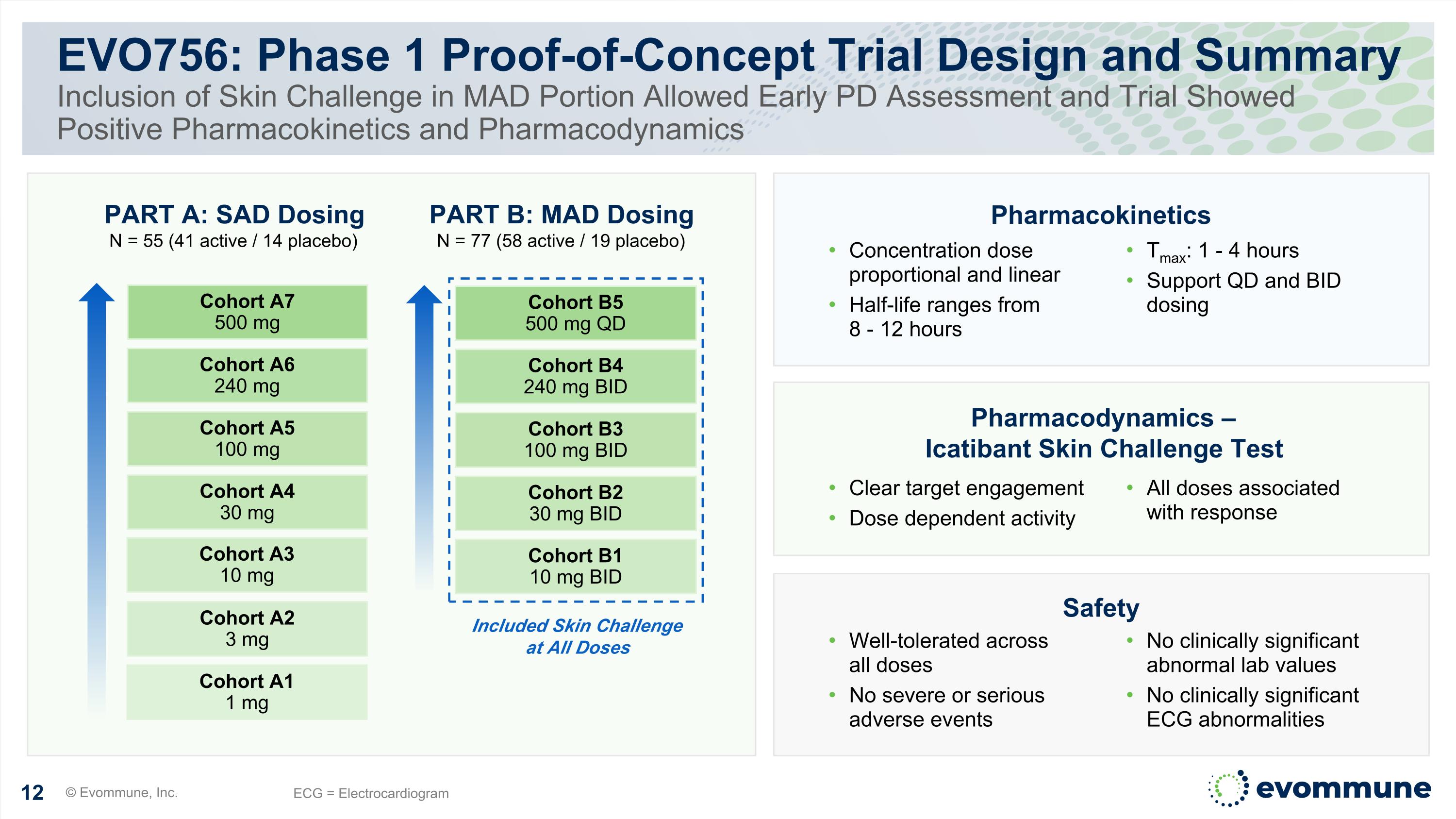
PART A: SAD Dosing N = 55 (41 active / 14 placebo) EVO756: Phase 1 Proof-of-Concept Trial Design and Summary Inclusion of Skin Challenge in MAD Portion Allowed Early PD Assessment and Trial Showed Positive Pharmacokinetics and Pharmacodynamics © Evommune, Inc. ECG = Electrocardiogram Cohort A1 1 mg Cohort A5 100 mg Cohort A4 30 mg Cohort A3 10 mg Cohort A2 3 mg Cohort A6 240 mg Cohort A7 500 mg Cohort B1 10 mg BID Cohort B3 100 mg BID Cohort B2 30 mg BID Cohort B4 240 mg BID Cohort B5 500 mg QD PART B: MAD Dosing N = 77 (58 active / 19 placebo) Included Skin Challenge at All Doses Pharmacokinetics Pharmacodynamics – Icatibant Skin Challenge Test Safety Concentration dose proportional and linear Half-life ranges from 8 - 12 hours Tmax: 1 - 4 hours Support QD and BID dosing Clear target engagement Dose dependent activity All doses associated with response Well-tolerated across all doses No severe or serious adverse events No clinically significant abnormal lab values No clinically significant ECG abnormalities

EVO756: Phase 2 Trial in Chronic Inducible Urticaria (CIndU) Data © Evommune, Inc.
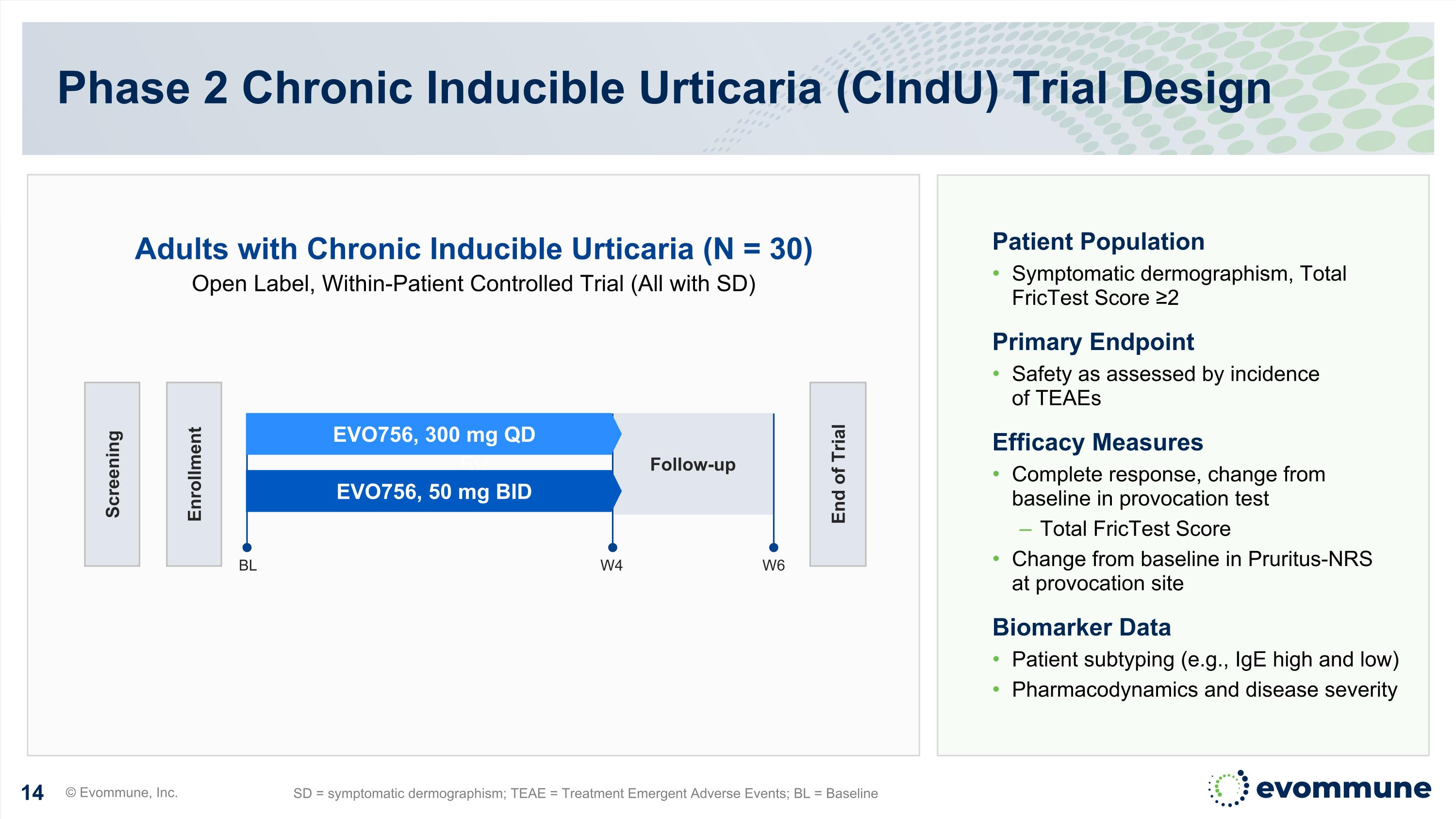
5v Phase 2 Chronic Inducible Urticaria (CIndU) Trial Design © Evommune, Inc. SD = symptomatic dermographism; TEAE = Treatment Emergent Adverse Events; BL = Baseline Follow-up Screening Enrollment End of Trial BL W6 EVO756, 300 mg QD EVO756, 50 mg BID W4 Patient Population Symptomatic dermographism, Total FricTest Score ≥2 Primary Endpoint Safety as assessed by incidence of TEAEs Efficacy Measures Complete response, change from baseline in provocation test Total FricTest Score Change from baseline in Pruritus-NRS at provocation site Biomarker Data Patient subtyping (e.g., IgE high and low) Pharmacodynamics and disease severity Adults with Chronic Inducible Urticaria (N = 30) Open Label, Within-Patient Controlled Trial (All with SD)
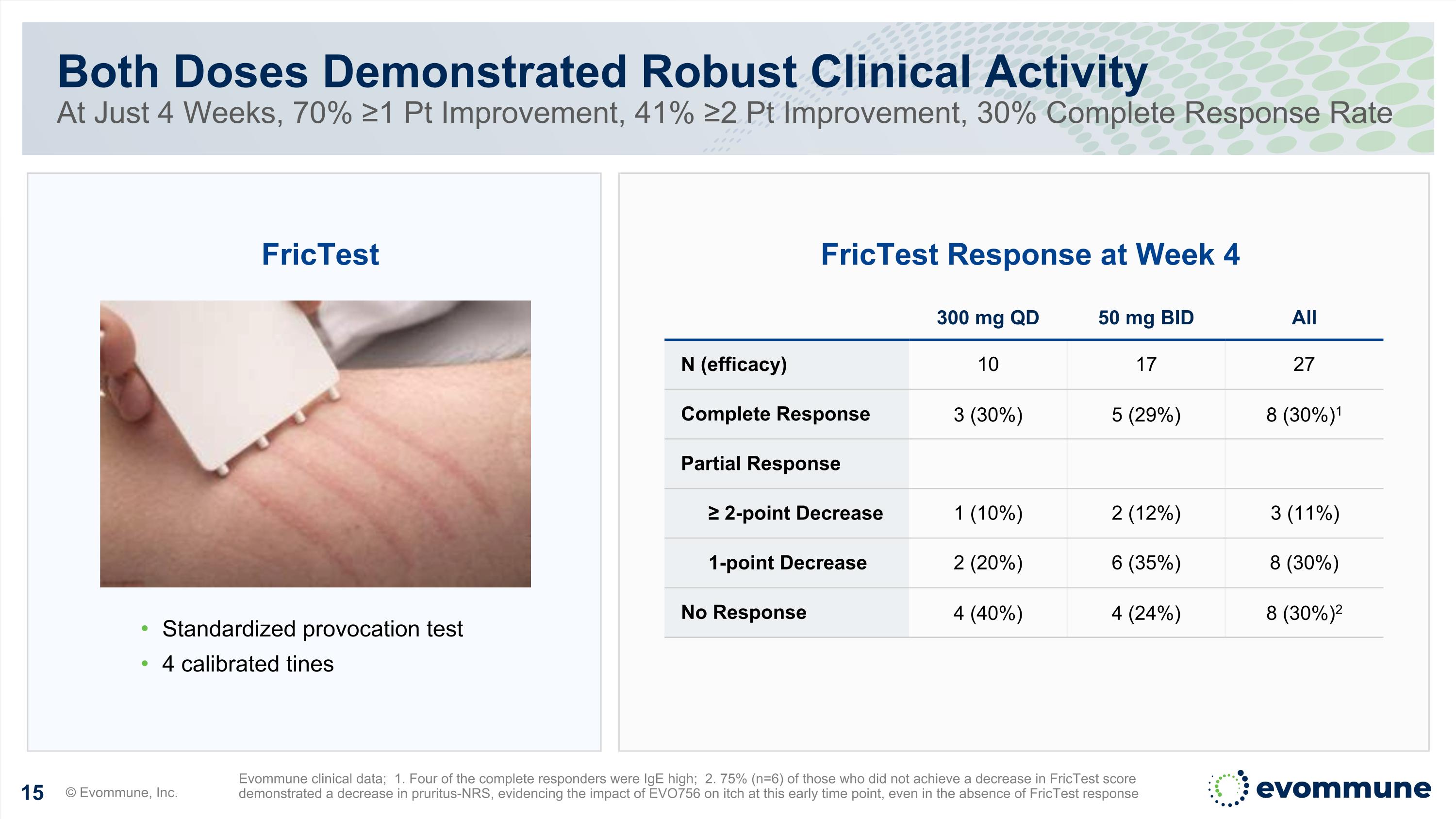
Both Doses Demonstrated Robust Clinical Activity At Just 4 Weeks, 70% ≥1 Pt Improvement, 41% ≥2 Pt Improvement, 30% Complete Response Rate © Evommune, Inc. Evommune clinical data; 1. Four of the complete responders were IgE high; 2. 75% (n=6) of those who did not achieve a decrease in FricTest score demonstrated a decrease in pruritus-NRS, evidencing the impact of EVO756 on itch at this early time point, even in the absence of FricTest response Standardized provocation test 4 calibrated tines FricTest FricTest Response at Week 4 300 mg QD 50 mg BID All N (efficacy) 10 17 27 Complete Response 3 (30%) 5 (29%) 8 (30%)1 Partial Response ≥ 2-point Decrease 1 (10%) 2 (12%) 3 (11%) 1-point Decrease 2 (20%) 6 (35%) 8 (30%) No Response 4 (40%) 4 (24%) 8 (30%)2
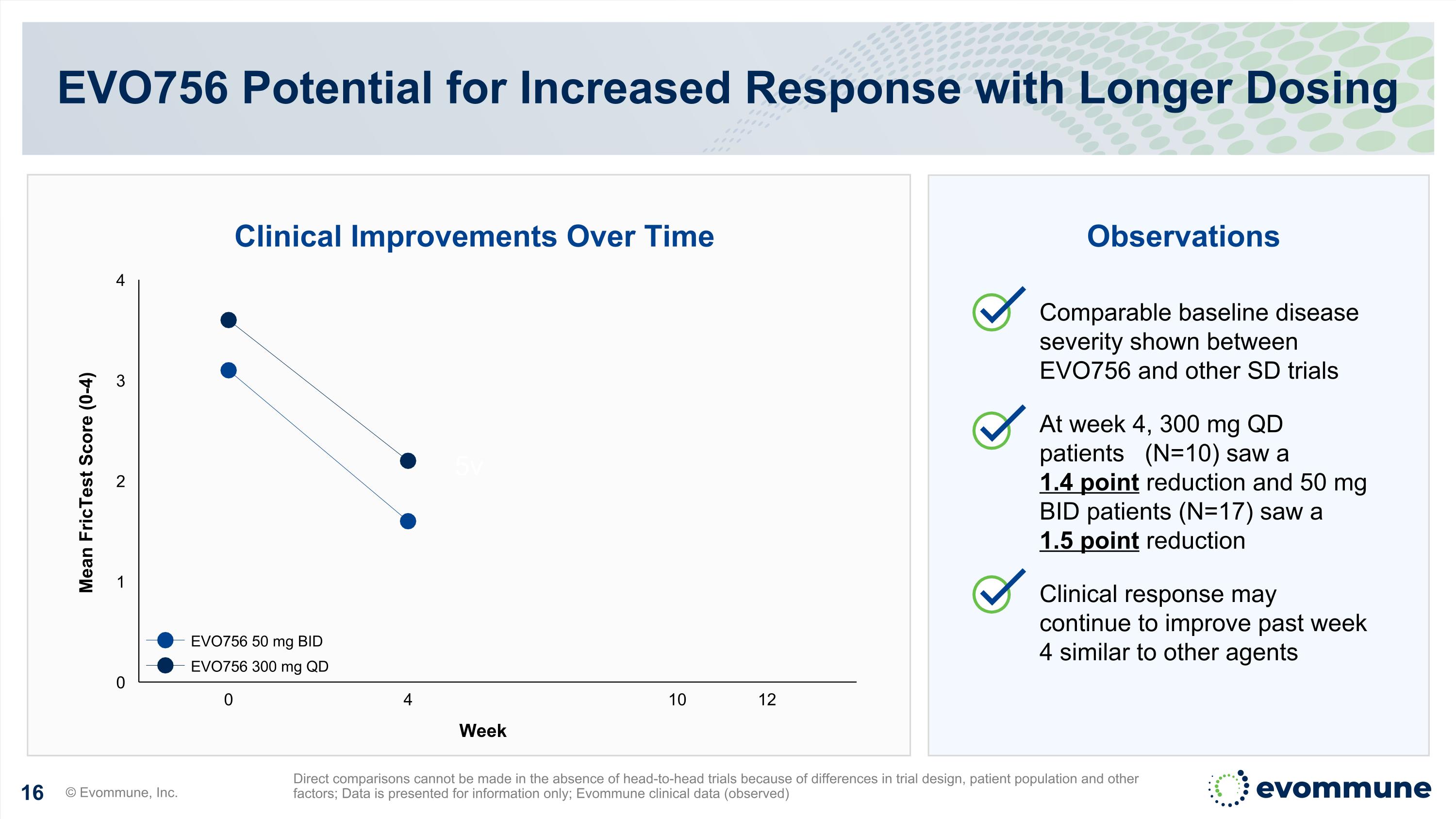
5v EVO756 Potential for Increased Response with Longer Dosing © Evommune, Inc. Direct comparisons cannot be made in the absence of head-to-head trials because of differences in trial design, patient population and other factors; Data is presented for information only; Evommune clinical data (observed) Week Mean FricTest Score (0-4) 0 4 10 12 EVO756 50 mg BID EVO756 300 mg QD Clinical Improvements Over Time Observations Comparable baseline disease severity shown between EVO756 and other SD trials At week 4, 300 mg QD patients (N=10) saw a 1.4 point reduction and 50 mg BID patients (N=17) saw a 1.5 point reduction Clinical response may continue to improve past week 4 similar to other agents
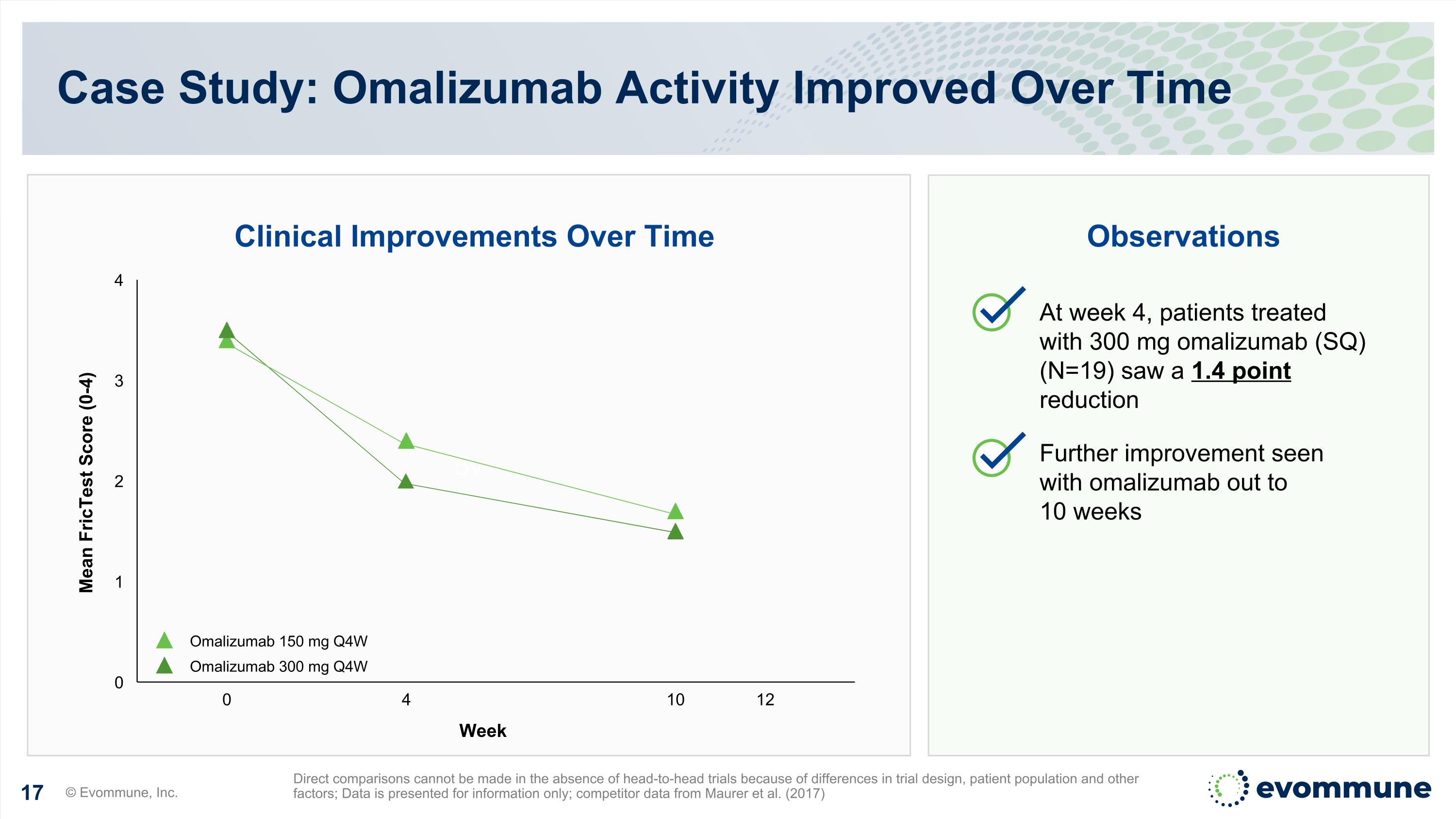
5v Clinical Improvements Over Time Observations Case Study: Omalizumab Activity Improved Over Time © Evommune, Inc. Direct comparisons cannot be made in the absence of head-to-head trials because of differences in trial design, patient population and other factors; Data is presented for information only; competitor data from Maurer et al. (2017) 0 4 10 12 Omalizumab 150 mg Q4W Omalizumab 300 mg Q4W At week 4, patients treated with 300 mg omalizumab (SQ) (N=19) saw a 1.4 point reduction Further improvement seen with omalizumab out to 10 weeks Week Mean FricTest Score (0-4)
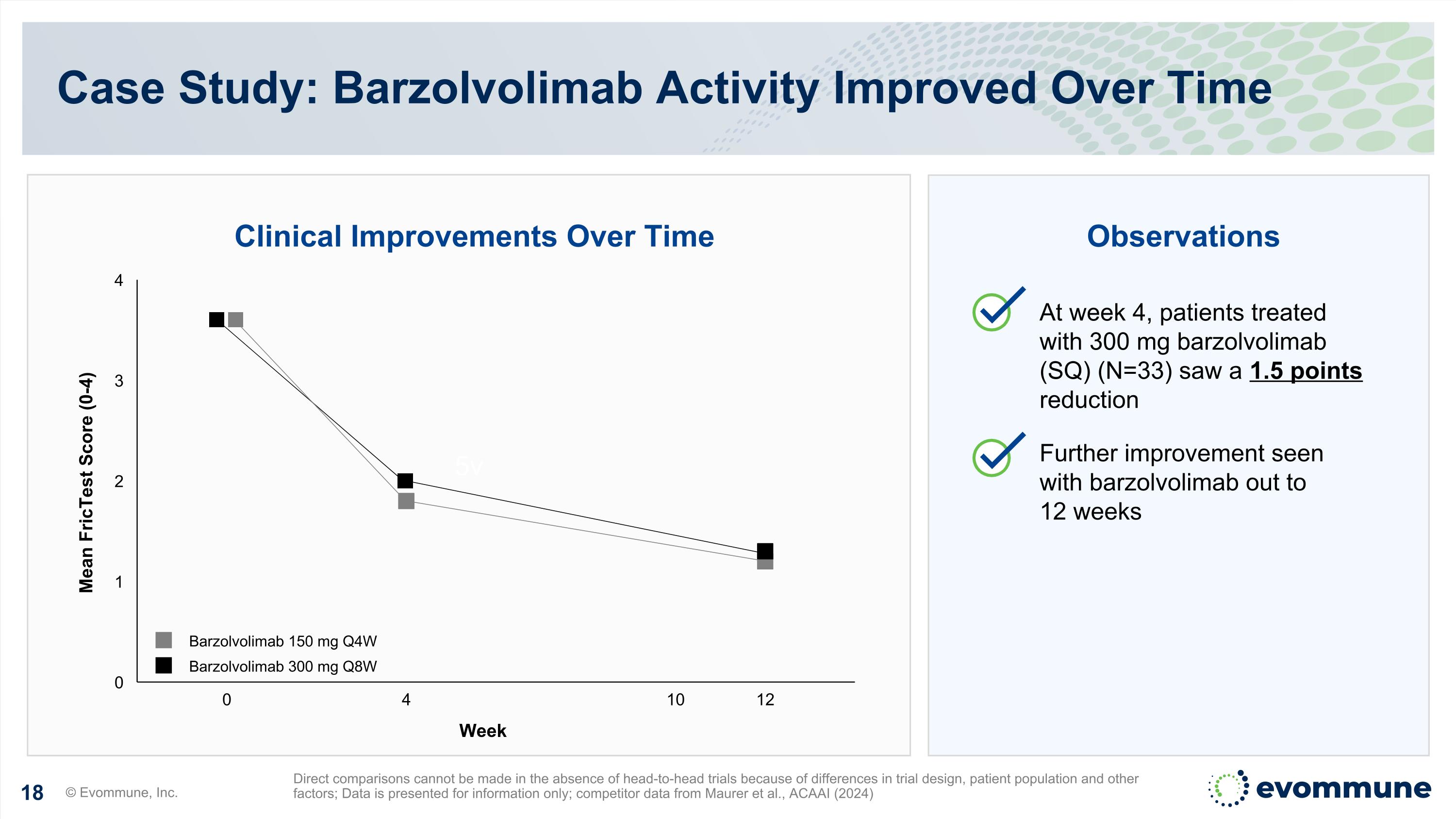
5v Clinical Improvements Over Time Observations At week 4, patients treated with 300 mg barzolvolimab (SQ) (N=33) saw a 1.5 points reduction Further improvement seen with barzolvolimab out to 12 weeks Case Study: Barzolvolimab Activity Improved Over Time © Evommune, Inc. Direct comparisons cannot be made in the absence of head-to-head trials because of differences in trial design, patient population and other factors; Data is presented for information only; competitor data from Maurer et al., ACAAI (2024) 0 4 10 12 Barzolvolimab 150 mg Q4W Barzolvolimab 300 mg Q8W Week Mean FricTest Score (0-4)
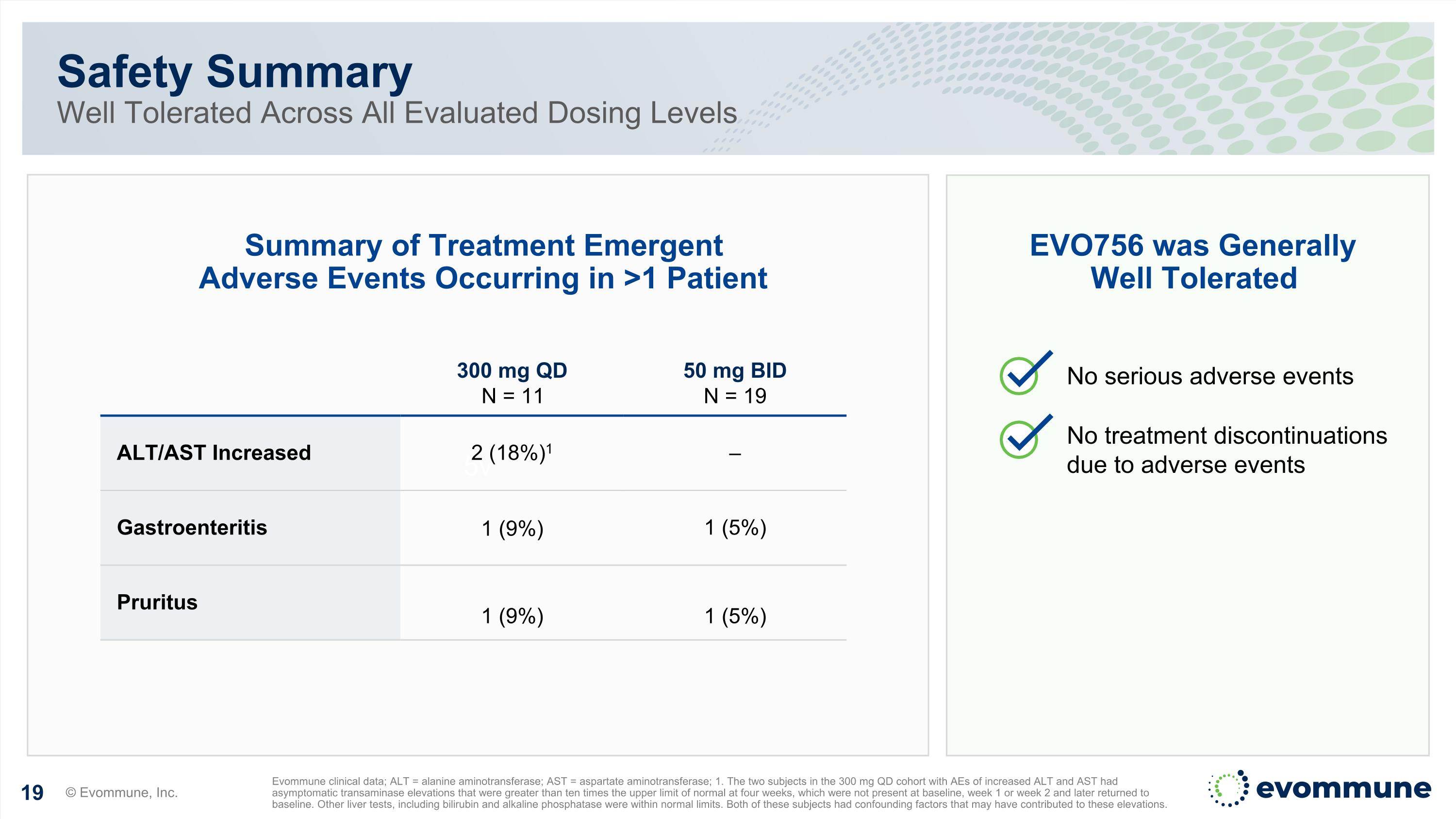
5v Safety Summary Well Tolerated Across All Evaluated Dosing Levels © Evommune, Inc. Evommune clinical data; ALT = alanine aminotransferase; AST = aspartate aminotransferase; 1. The two subjects in the 300 mg QD cohort with AEs of increased ALT and AST had asymptomatic transaminase elevations that were greater than ten times the upper limit of normal at four weeks, which were not present at baseline, week 1 or week 2 and later returned to baseline. Other liver tests, including bilirubin and alkaline phosphatase were within normal limits. Both of these subjects had confounding factors that may have contributed to these elevations. 300 mg QD N = 11 50 mg BID N = 19 ALT/AST Increased 2 (18%)1 – Gastroenteritis 1 (9%) 1 (5%) Pruritus 1 (9%) 1 (5%) Summary of Treatment Emergent Adverse Events Occurring in >1 Patient EVO756 was Generally Well Tolerated No serious adverse events No treatment discontinuations due to adverse events
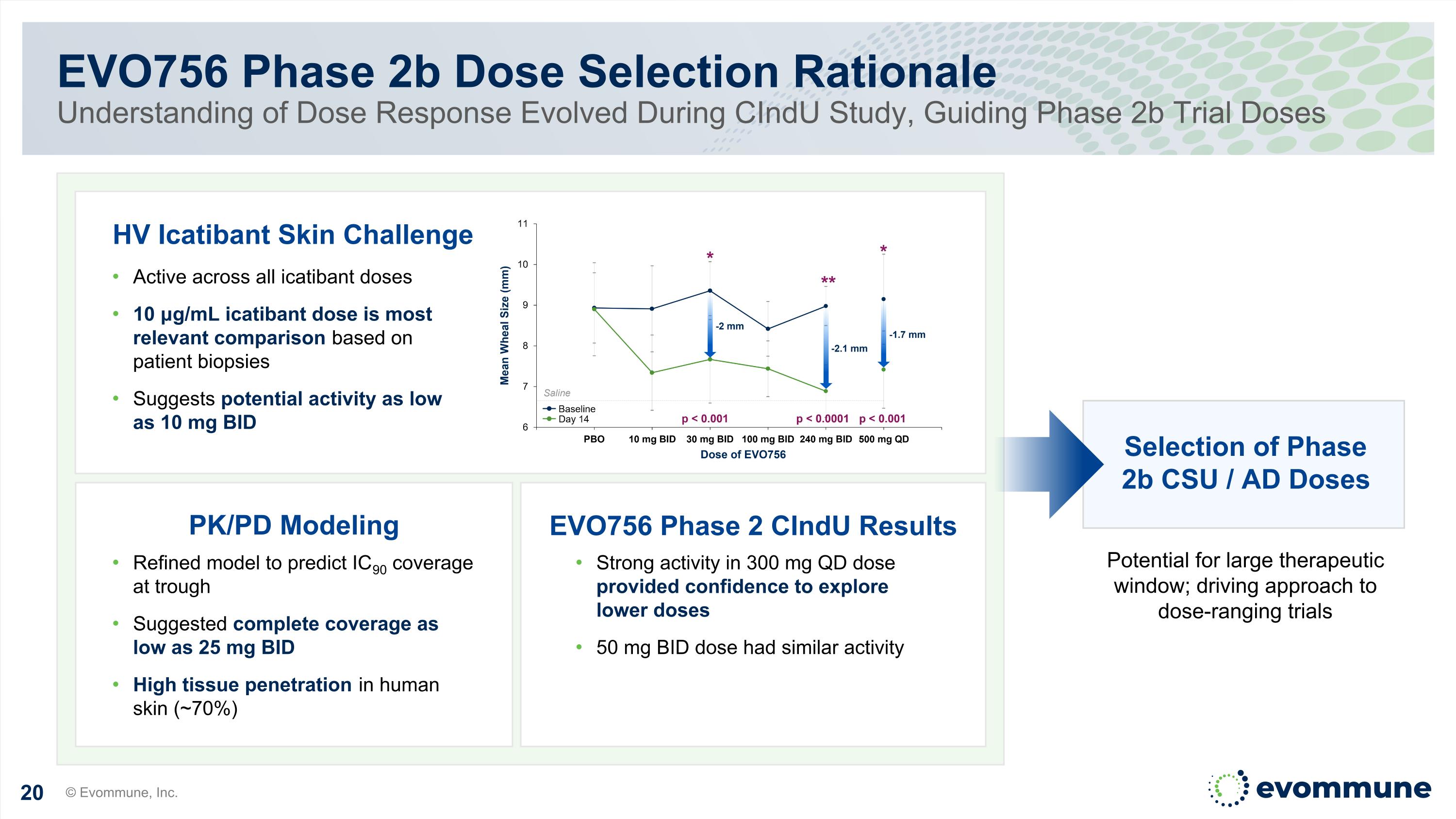
EVO756 Phase 2b Dose Selection Rationale Understanding of Dose Response Evolved During CIndU Study, Guiding Phase 2b Trial Doses © Evommune, Inc. Active across all icatibant doses 10 μg/mL icatibant dose is most relevant comparison based on patient biopsies Suggests potential activity as low as 10 mg BID PK/PD Modeling Refined model to predict IC90 coverage at trough Suggested complete coverage as low as 25 mg BID High tissue penetration in human skin (~70%) Strong activity in 300 mg QD dose provided confidence to explore lower doses 50 mg BID dose had similar activity EVO756 Phase 2 CIndU Results HV Icatibant Skin Challenge Selection of Phase 2b CSU / AD Doses Potential for large therapeutic window; driving approach to dose-ranging trials

EVO756: Phase 2b Trial in Chronic Spontaneous Urticaria (CSU) © Evommune, Inc.

Relationship Between CIndU Efficacy and Impact on CSU Phase 2 EVO756 Results in CIndU Provide Early Support for Potential CSU Clinical Profile © Evommune, Inc. Direct comparisons cannot be made in the absence of head-to-head trials because of differences in trial design, patient population and other factors. Data is presented for information only and does not account for difference in enrolment populations or other cross-trial variabilities MOA Cell Target Drug CIndU CSU IgE Mast Cells Basophils Eosinophils omalizumab KIT Mast Cells Hematopoietic Stem Cells Germ Cells Melanocytes barzolvolimab BTK Mast Cells B Cells Basophils Myeloid Cells remibrutinib IL-4 / IL-13 Th2 Cells Epithelial Cells Macrophages dupilumab Correlation Between CIndU Success and CSU Benefit
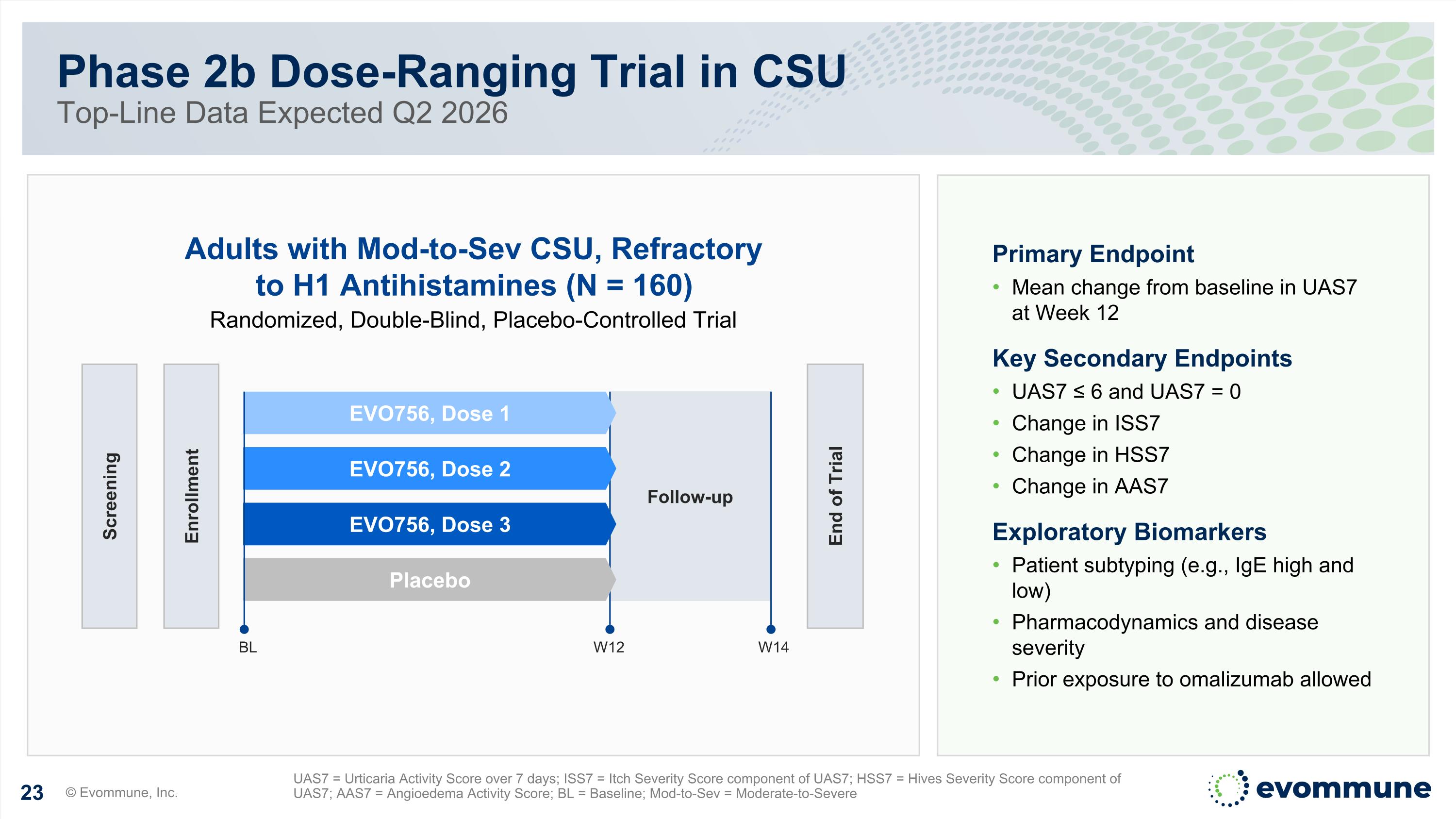
Phase 2b Dose-Ranging Trial in CSU Top-Line Data Expected Q2 2026 © Evommune, Inc. UAS7 = Urticaria Activity Score over 7 days; ISS7 = Itch Severity Score component of UAS7; HSS7 = Hives Severity Score component of UAS7; AAS7 = Angioedema Activity Score; BL = Baseline; Mod-to-Sev = Moderate-to-Severe Primary Endpoint Mean change from baseline in UAS7 at Week 12 Key Secondary Endpoints UAS7 ≤ 6 and UAS7 = 0 Change in ISS7 Change in HSS7 Change in AAS7 Exploratory Biomarkers Patient subtyping (e.g., IgE high and low) Pharmacodynamics and disease severity Prior exposure to omalizumab allowed 5v BL W14 Adults with Mod-to-Sev CSU, Refractory to H1 Antihistamines (N = 160) Randomized, Double-Blind, Placebo-Controlled Trial Follow-up Screening Enrollment End of Trial W12 EVO756, Dose 1 EVO756, Dose 2 EVO756, Dose 3 Placebo
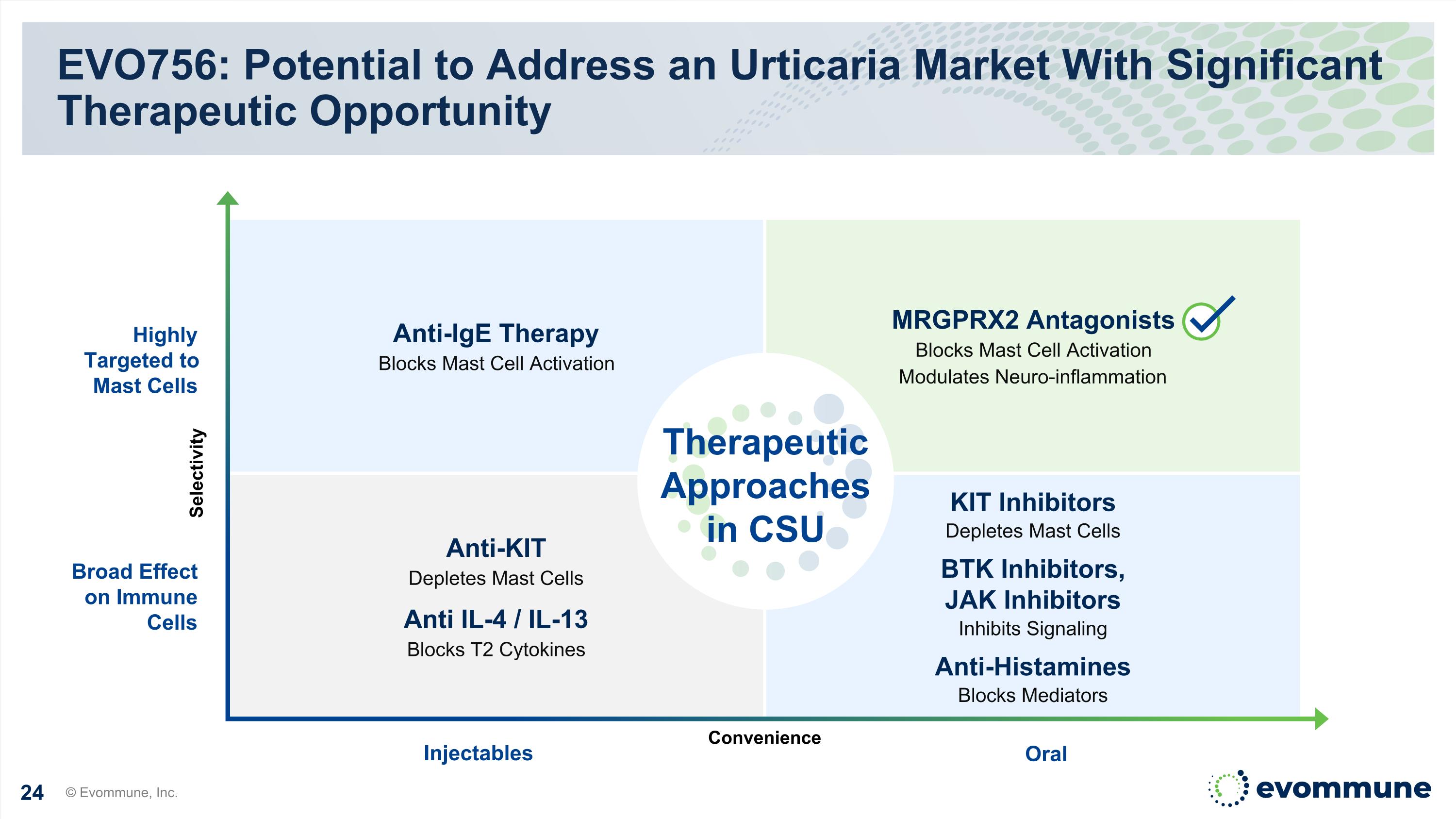
EVO756: Potential to Address an Urticaria Market With Significant Therapeutic Opportunity Anti-IgE Therapy Blocks Mast Cell Activation Anti-KIT Depletes Mast Cells Anti IL-4 / IL-13 Blocks T2 Cytokines MRGPRX2 Antagonists Blocks Mast Cell Activation Modulates Neuro-inflammation KIT Inhibitors Depletes Mast Cells BTK Inhibitors, JAK Inhibitors Inhibits Signaling Anti-Histamines Blocks Mediators Oral Injectables Highly Targeted to Mast Cells Broad Effect on Immune Cells © Evommune, Inc. Selectivity Convenience Therapeutic Approaches in CSU
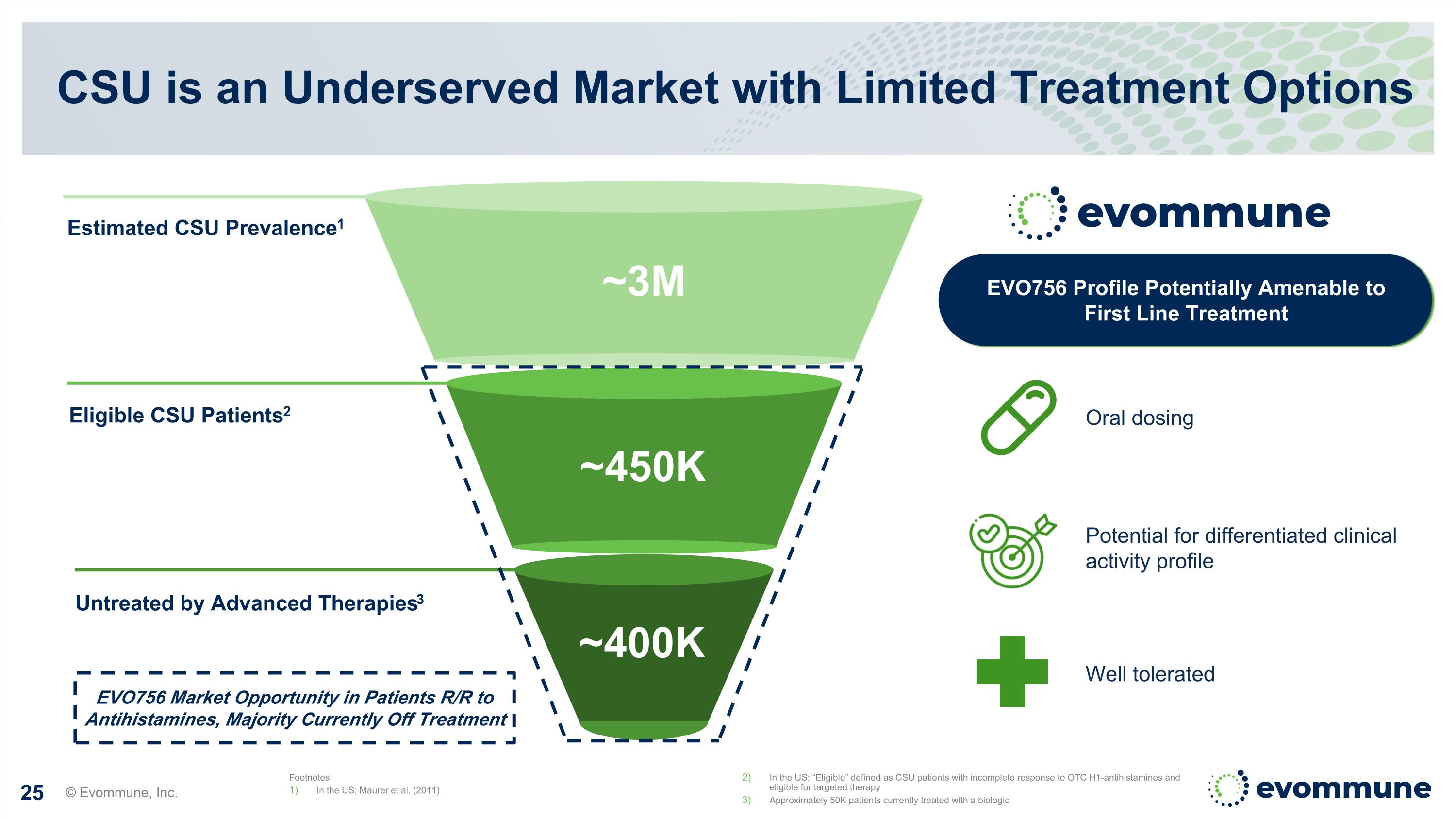
Estimated CSU Prevalence1 Eligible CSU Patients2 Untreated by Advanced Therapies3 CSU is an Underserved Market with Limited Treatment Options Footnotes: In the US; Maurer et al. (2011) In the US; “Eligible” defined as CSU patients with incomplete response to OTC H1-antihistamines and eligible for targeted therapy Approximately 50K patients currently treated with a biologic Potential for differentiated clinical activity profile Oral dosing Well tolerated EVO756 Profile Potentially Amenable to First Line Treatment ~450K ~3M ~400K EVO756 Market Opportunity in Patients R/R to Antihistamines, Majority Currently Off Treatment © Evommune, Inc.

EVO756 in Atopic Dermatitis (AD) © Evommune, Inc.

MRGPRX2 is Only Dual MOA: Targeting Inflammatory Lesions and Neuroinflammation Strong Scientific Rationale for EVO756 in AD Expect Benefit on Mast Cell and Neuroinflammation Aspects of AD © Evommune, Inc. Pathway activation in disease and preclinical evidence of Mrgprb2/X2 involvement Strong translational validation Broad therapeutic potential MOA likely effective across patient endotypes Direct effect on sensory neurons Rapid impact on itch Dual mechanism impacting key inflammatory pathways Mast cell and neuroinflammatory disease Sensory Neuron Mast Cell MRGPRX2 Nerves Mast Cells

Phase 2b Dose-Ranging Trial in AD Top-Line Data Expected H2 2026 © Evommune, Inc. EASI = Eczema Area and Severity Index; vIGA = Validated Investigator Global Assessment; Pruritus-NRS = Pruritus Numerical Rating Scale; BSA = Body Surface Area; BL = Baseline Primary Endpoint Percent change from EASI at Week 12 Key Secondary Endpoints EASI-50, EASI-75, and EASI-90 Change in vIGA Change in Pruritus-NRS Proportion of patients achieving ≥4 point reduction in Pruritus-NRS Change in BSA affected Exploratory Biomarkers Patient subtyping Pharmacodynamics & disease severity 5v BL W14 Adults with Moderate-to-Severe Atopic Dermatitis (N = 120) Randomized, Double-Blind, Placebo-Controlled Trial Follow-up Screening Enrollment End of Trial W12 EVO756, Dose 1 EVO756, Dose 2 EVO756, Dose 3 Placebo
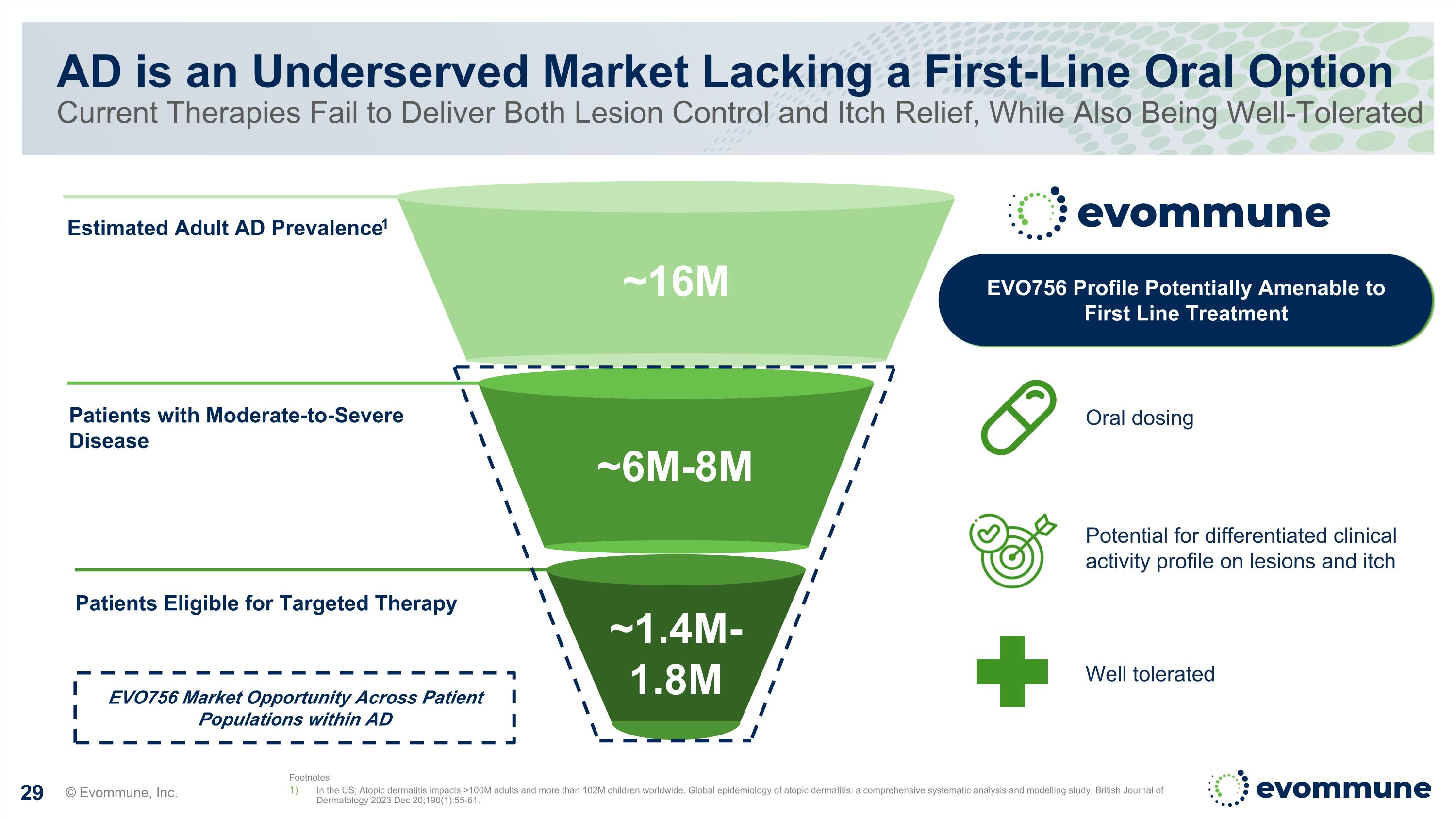
Estimated Adult AD Prevalence1 Patients with Moderate-to-Severe Disease Patients Eligible for Targeted Therapy AD is an Underserved Market Lacking a First-Line Oral Option Current Therapies Fail to Deliver Both Lesion Control and Itch Relief, While Also Being Well-Tolerated Footnotes: In the US; Atopic dermatitis impacts >100M adults and more than 102M children worldwide. Global epidemiology of atopic dermatitis: a comprehensive systematic analysis and modelling study. British Journal of Dermatology 2023 Dec 20;190(1):55-61. Potential for differentiated clinical activity profile on lesions and itch Oral dosing Well tolerated EVO756 Profile Potentially Amenable to First Line Treatment ~6M-8M ~16M EVO756 Market Opportunity Across Patient Populations within AD ~1.4M-1.8M © Evommune, Inc.
EVO301: IL-18BP Fusion Protein Long-Acting Serum Albumin-Binding Injectable Therapeutic Fusion Protein Designed to Neutralize IL-18 Signaling © Evommune, Inc.
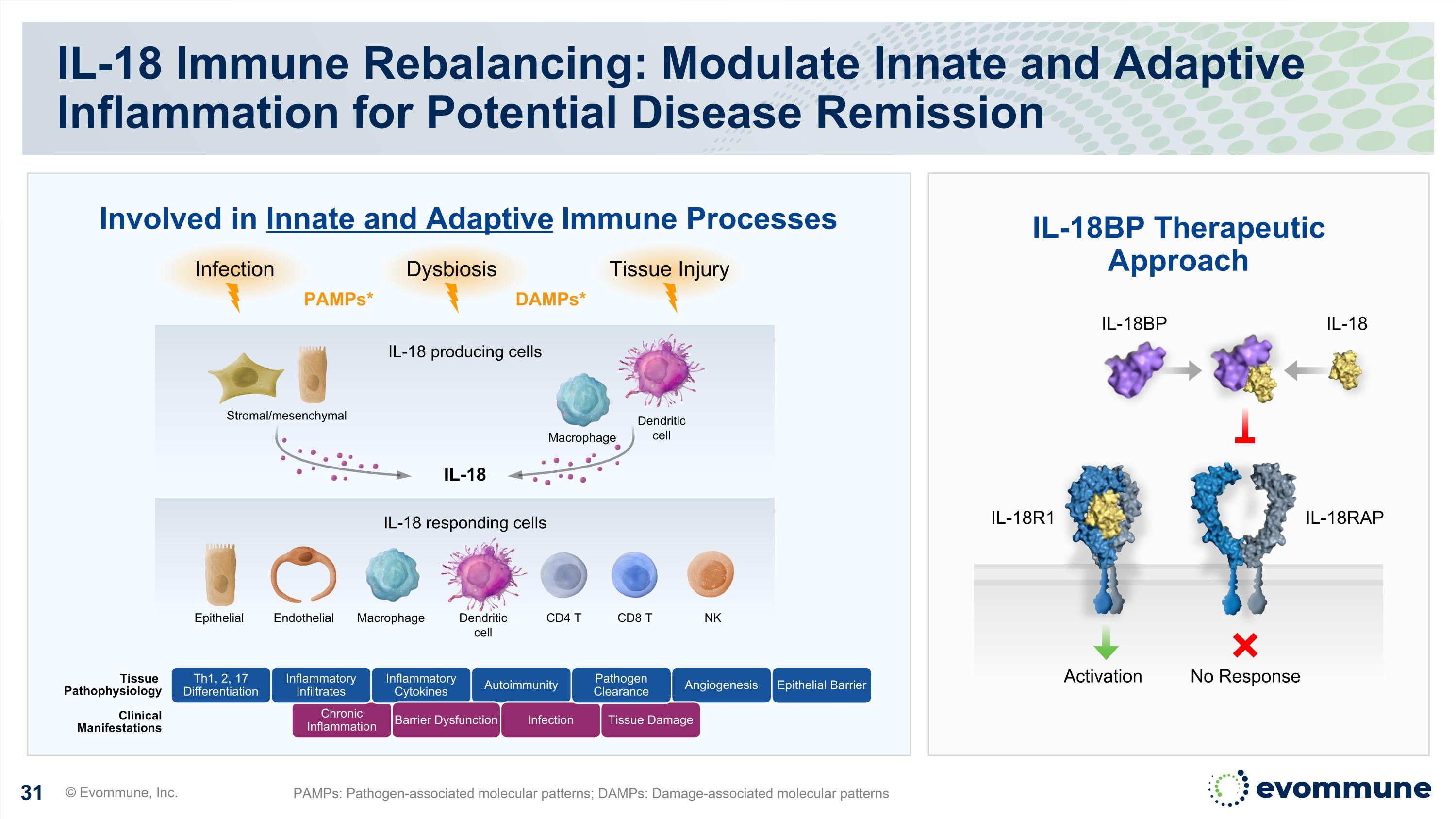
IL-18 Immune Rebalancing: Modulate Innate and Adaptive Inflammation for Potential Disease Remission © Evommune, Inc. PAMPs: Pathogen-associated molecular patterns; DAMPs: Damage-associated molecular patterns Chronic Inflammation Tissue Pathophysiology Clinical Manifestations Inflammatory Infiltrates Inflammatory Cytokines Autoimmunity Angiogenesis Th1, 2, 17 Differentiation Barrier Dysfunction Infection Tissue Damage Pathogen Clearance IL-18BP Therapeutic Approach Involved in Innate and Adaptive Immune Processes IL-18 producing cells IL-18 responding cells Stromal/mesenchymal IL-18 Epithelial Endothelial CD4 T NK CD8 T Macrophage Dendritic cell Dendritic cell Macrophage Dysbiosis Tissue Injury Infection DAMPs* PAMPs* IL-18R1 IL-18RAP No Response Activation IL-18 IL-18BP Epithelial Barrier
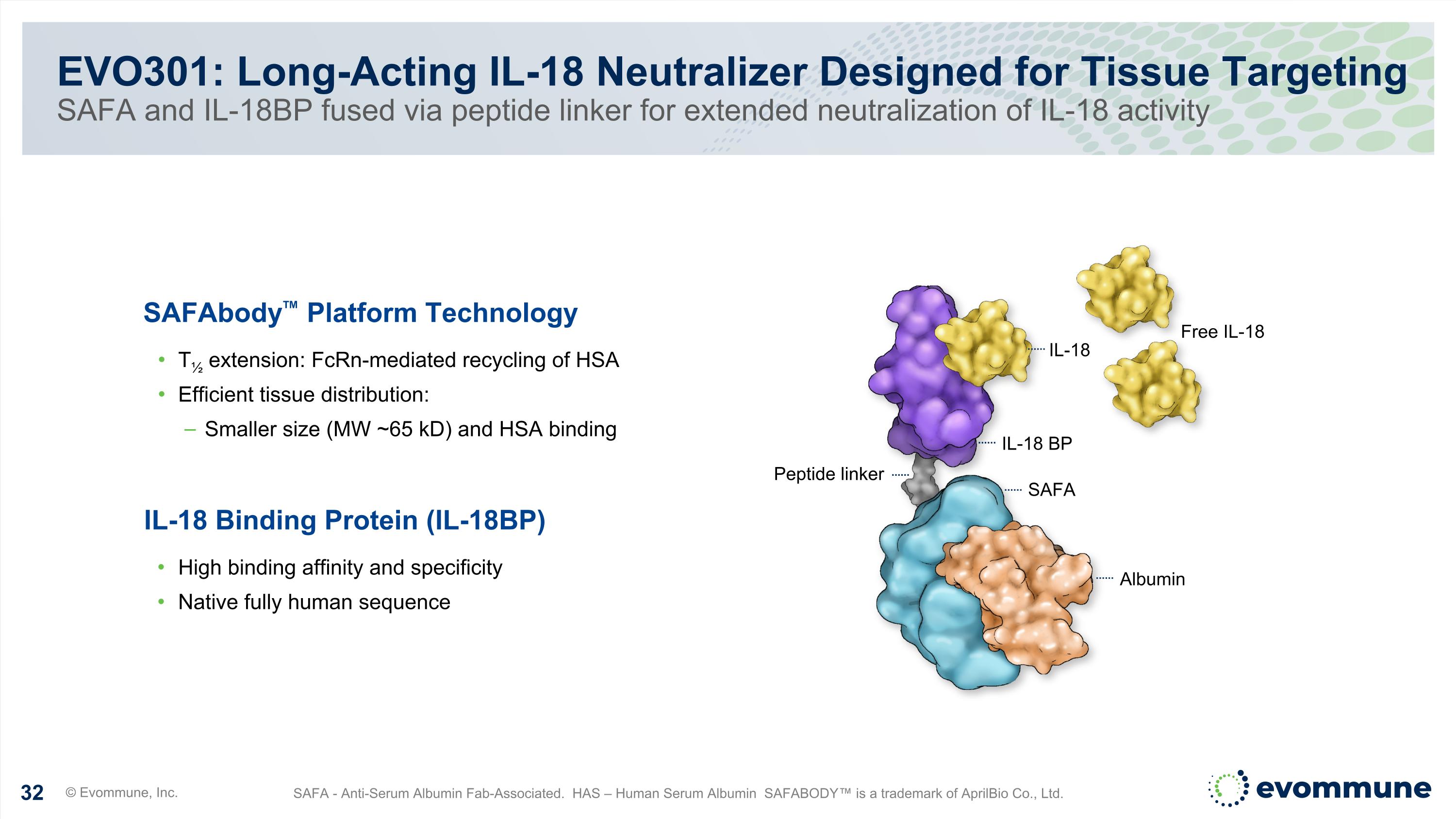
EVO301: Long-Acting IL-18 Neutralizer Designed for Tissue Targeting SAFA and IL-18BP fused via peptide linker for extended neutralization of IL-18 activity © Evommune, Inc. SAFA - Anti-Serum Albumin Fab-Associated. HAS – Human Serum Albumin SAFABODY™ is a trademark of AprilBio Co., Ltd. Free IL-18 IL-18 IL-18 BP SAFA Peptide linker Albumin SAFAbody™ Platform Technology T½ extension: FcRn-mediated recycling of HSA Efficient tissue distribution: Smaller size (MW ~65 kD) and HSA binding IL-18 Binding Protein (IL-18BP) High binding affinity and specificity Native fully human sequence

Biologic Pathway Adaptive Inflammation Innate Inflammation Skin Barrier (IL-22) TH2 TH1 TH17 IL-18 DUPIXENT® EBGLYSS® ADBRY® NEMLUVIO® EVO301 Addresses Limitations of Existing Biologics; Demonstrating Ability to Impact Multiple Drivers of AD, while being Well Tolerated © Evommune, Inc. Green = impacts biological pathway; Red = negative effect. Broader inflammatory signaling of IL-18 can address endotypes not fully captured by Th2-targeted therapies —enabling potential for broader patient coverage and efficacy

EVO301 Phase 2a Proof of Concept Trial Design © Evommune, Inc. EASI = Eczema Area and Severity Index; vIGA = Validated Investigator Global Assessment; BSA = Body Surface Area; BL = Baseline; PBO = Placebo Screening Randomization 2 Active : 1 PBO End of Trial BL W12 W4 Adults with Moderate-to-Severe Atopic Dermatitis (N = 70) Randomized, Double-Blind, Parallel Group, Placebo-Controlled Trial AD Population EASI ≥16 vIGA ≥3 BSA ≥10% Primary Endpoint Percent change from EASI at Week 12 (Bayesian) Pharmacokinetics Target Engagement W8 EVO301: 5 mg/kg IV Placebo Dosing day Dosing day W2

EVO301 Achieved the Primary Endpoint Phase 2a Proof-of-Concept Trial in Moderate-to-Severe Atopic Dermatitis Highly statistically significant EASI reductions at weeks 4, 8, and 12 versus placebo 34% and 33% placebo adjusted improvement in EASI at week 8 and 12, respectively 23% of patients achieved IGA 0/1 at week 12 versus 0% placebo Well-tolerated, with no treatment related serious or severe adverse events reported Corresponding reductions in secondary endpoints, as well as key Th2 and non Th2 cytokines Pharmacokinetics (PK) continues to support a Q4 week dosing regimen © Evommune, Inc. Clinical Data Supports Continued Development, with Phase 2b Planning Underway
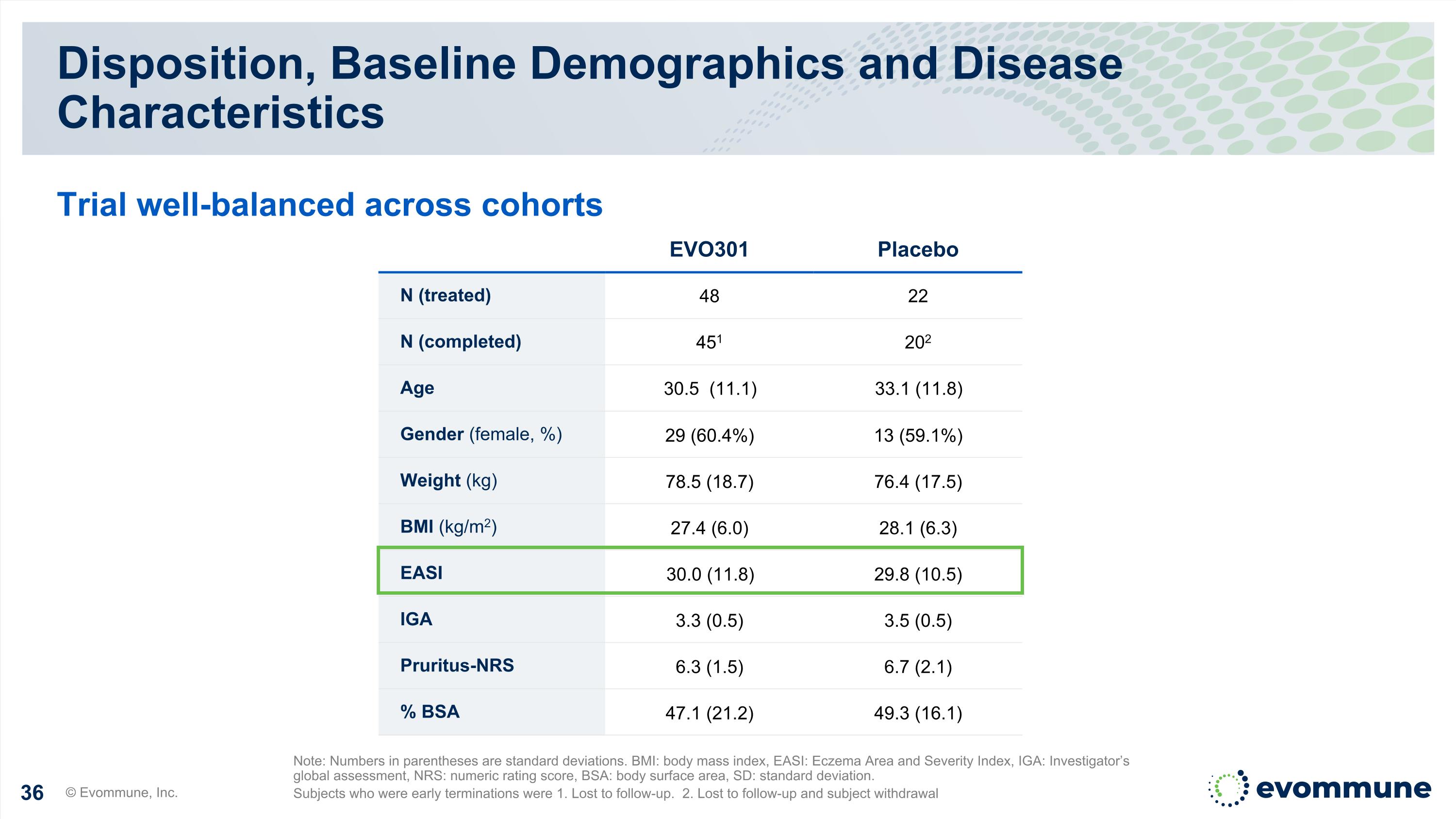
Disposition, Baseline Demographics and Disease Characteristics © Evommune, Inc. Note: Numbers in parentheses are standard deviations. BMI: body mass index, EASI: Eczema Area and Severity Index, IGA: Investigator’s global assessment, NRS: numeric rating score, BSA: body surface area, SD: standard deviation. Subjects who were early terminations were 1. Lost to follow-up. 2. Lost to follow-up and subject withdrawal Trial well-balanced across cohorts EVO301 Placebo N (treated) 48 22 N (completed) 451 202 Age 30.5 (11.1) 33.1 (11.8) Gender (female, %) 29 (60.4%) 13 (59.1%) Weight (kg) 78.5 (18.7) 76.4 (17.5) BMI (kg/m2) 27.4 (6.0) 28.1 (6.3) EASI 30.0 (11.8) 29.8 (10.5) IGA 3.3 (0.5) 3.5 (0.5) Pruritus-NRS 6.3 (1.5) 6.7 (2.1) % BSA 47.1 (21.2) 49.3 (16.1)
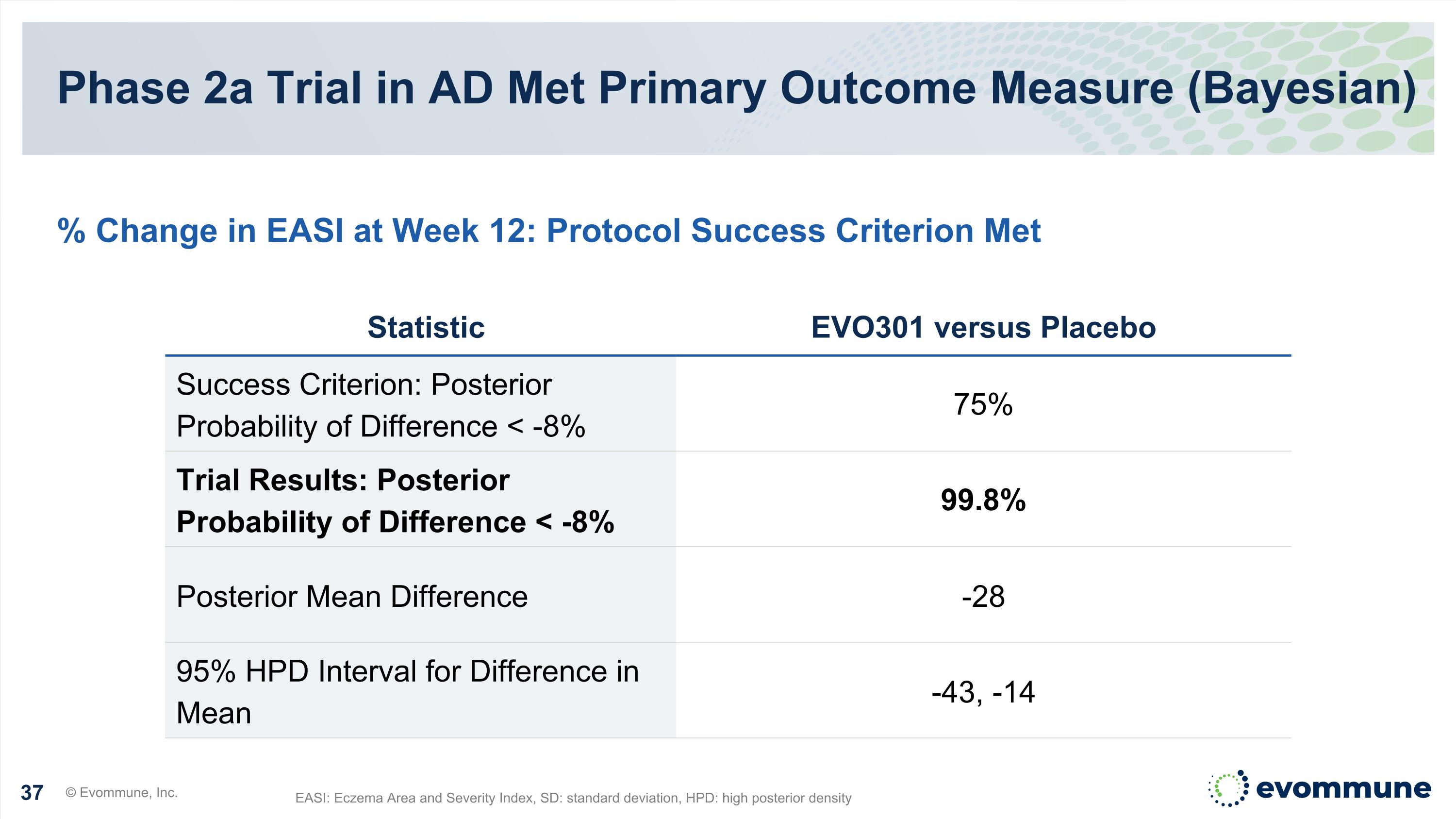
Phase 2a Trial in AD Met Primary Outcome Measure (Bayesian) EASI: Eczema Area and Severity Index, SD: standard deviation, HPD: high posterior density % Change in EASI at Week 12: Protocol Success Criterion Met Statistic EVO301 versus Placebo Success Criterion: Posterior Probability of Difference < -8% 75% Trial Results: Posterior Probability of Difference < -8% 99.8% Posterior Mean Difference -28 95% HPD Interval for Difference in Mean -43, -14 © Evommune, Inc.
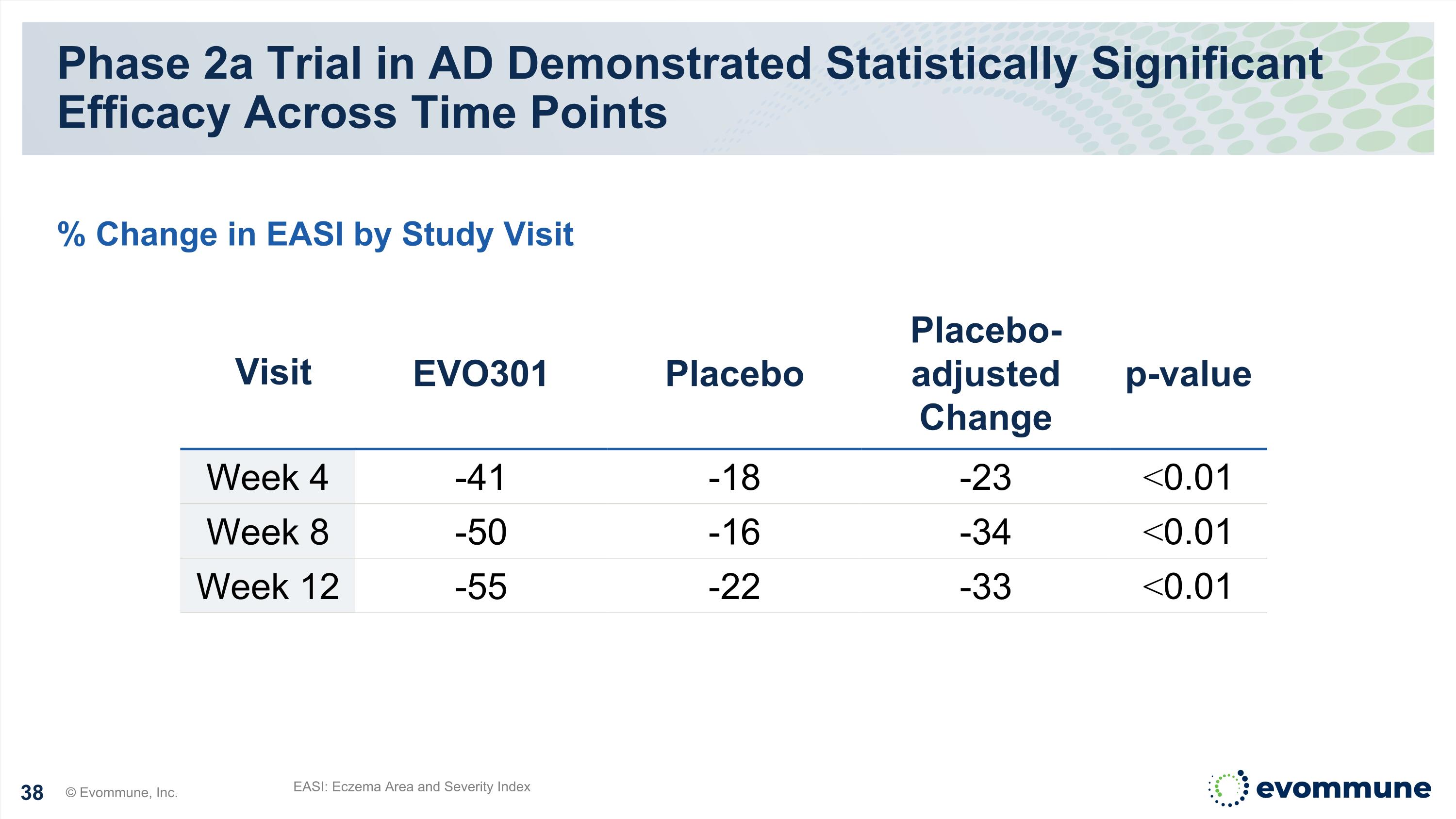
Phase 2a Trial in AD Demonstrated Statistically Significant Efficacy Across Time Points EASI: Eczema Area and Severity Index % Change in EASI by Study Visit Visit EVO301 Placebo Placebo-adjusted Change p-value Week 4 -41 -18 -23 <0.01 Week 8 -50 -16 -34 <0.01 Week 12 -55 -22 -33 <0.01 © Evommune, Inc.
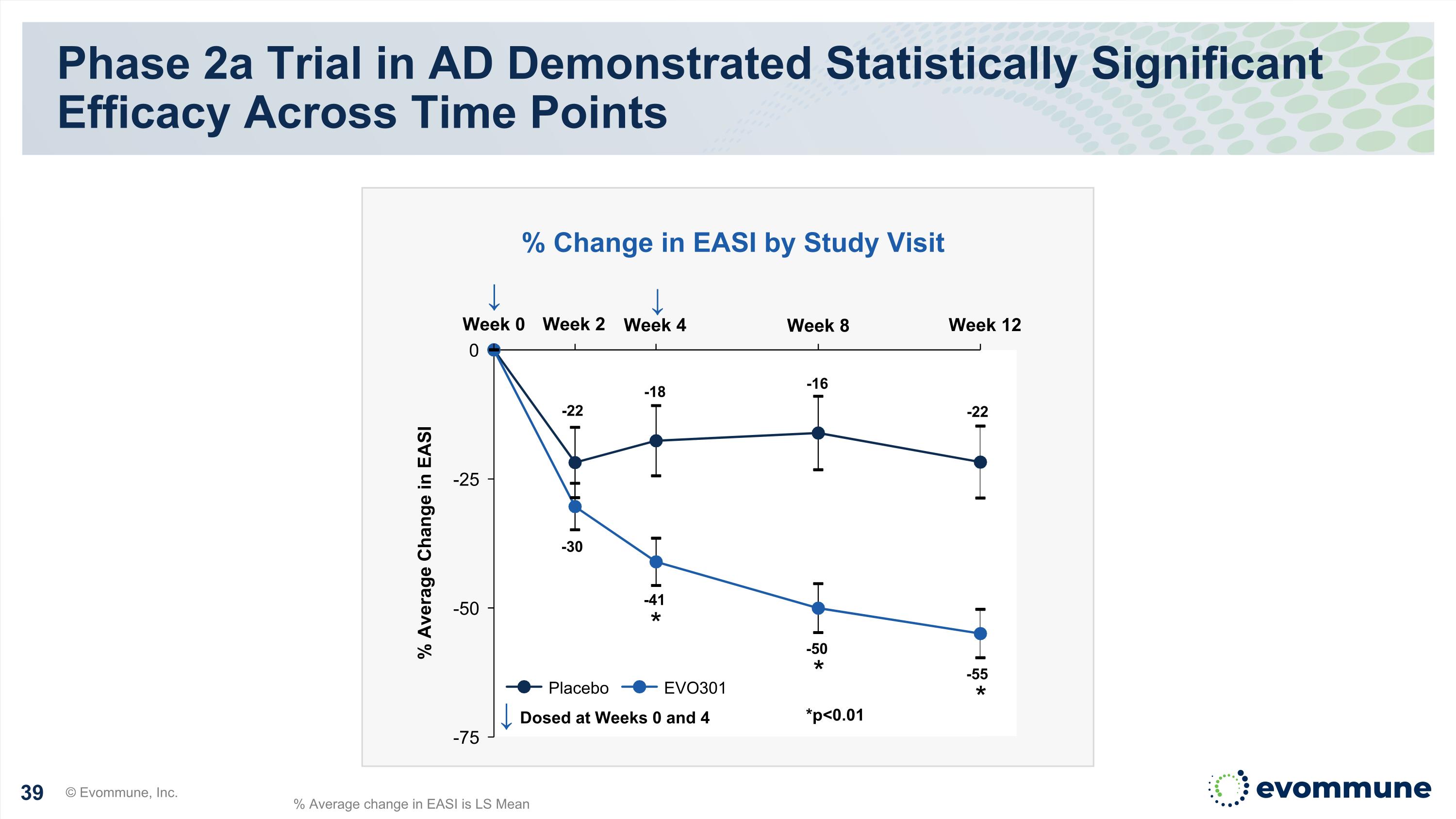
Week 2 Phase 2a Trial in AD Demonstrated Statistically Significant Efficacy Across Time Points © Evommune, Inc. Week 0 Week 4 Week 8 Week 12 % Average Change in EASI ↓ Dosed at Weeks 0 and 4 Placebo EVO301 * * % Change in EASI by Study Visit *p<0.01 ↓ % Average change in EASI is LS Mean -22 -18 -16 -22 -30 -41 -50 -55 ↓ *
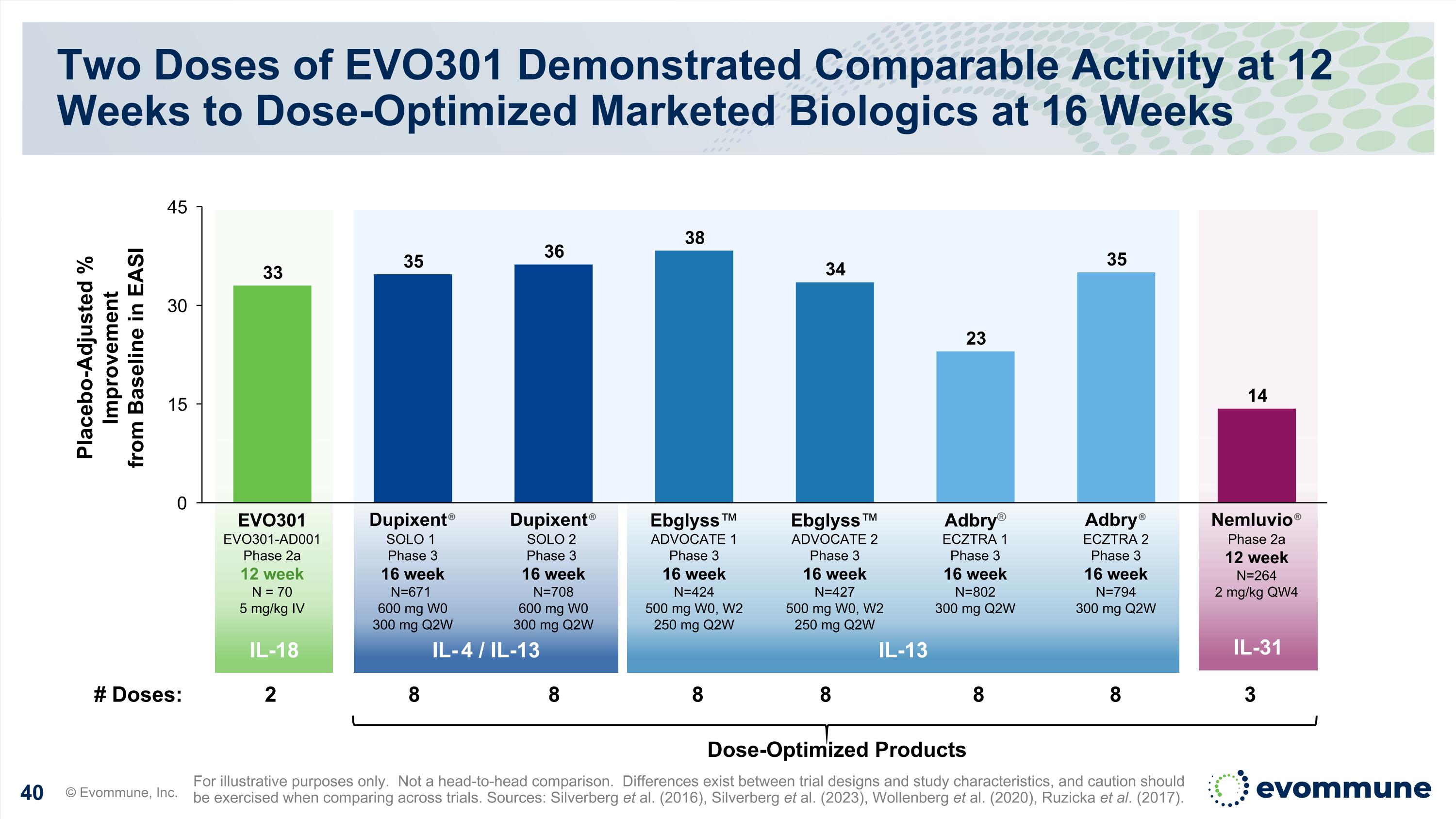
IL-18 IL-31 IL-13 IL-4 / IL-13 Two Doses of EVO301 Demonstrated Comparable Activity at 12 Weeks to Dose-Optimized Marketed Biologics at 16 Weeks © Evommune, Inc. For illustrative purposes only. Not a head-to-head comparison. Differences exist between trial designs and study characteristics, and caution should be exercised when comparing across trials. Sources: Silverberg et al. (2016), Silverberg et al. (2023), Wollenberg et al. (2020), Ruzicka et al. (2017). Placebo-Adjusted % Improvement from Baseline in EASI Dose-Optimized Products # Doses: 2 8 8 8 8 8 8 3 EVO301 EVO301-AD001 Phase 2a 12 week N = 70 5 mg/kg IV Dupixent® SOLO 1 Phase 3 16 week N=671 600 mg W0 300 mg Q2W Dupixent® SOLO 2 Phase 3 16 week N=708 600 mg W0 300 mg Q2W Ebglyss™ ADVOCATE 1 Phase 3 16 week N=424 500 mg W0, W2 250 mg Q2W Ebglyss™ ADVOCATE 2 Phase 3 16 week N=427 500 mg W0, W2 250 mg Q2W Adbry® ECZTRA 1 Phase 3 16 week N=802 300 mg Q2W Adbry® ECZTRA 2 Phase 3 16 week N=794 300 mg Q2W Nemluvio® Phase 2a 12 week N=264 2 mg/kg QW4
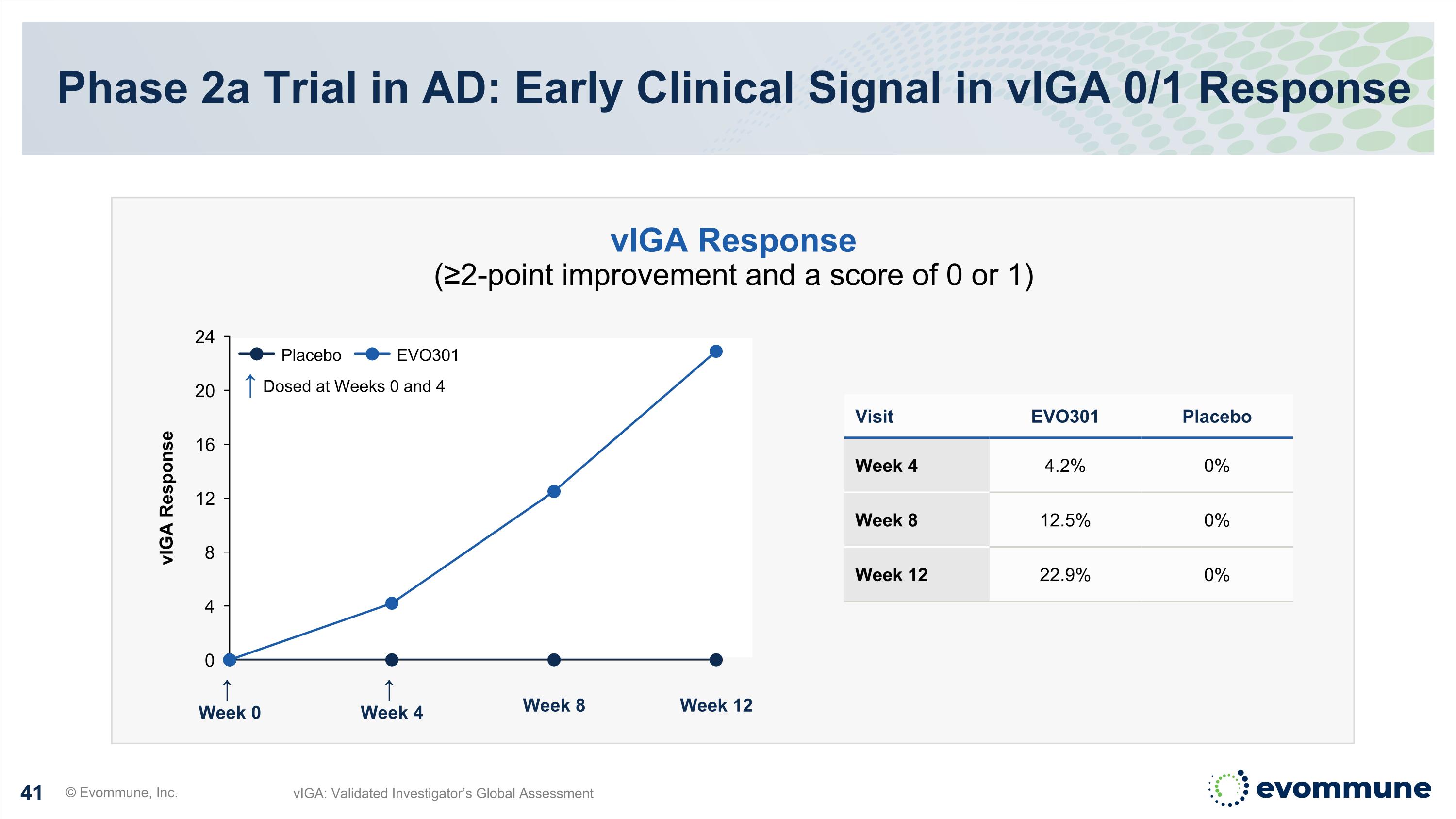
↑ Week 0 ↑ Week 4 Week 8 Week 12 vIGA Response Phase 2a Trial in AD: Early Clinical Signal in vIGA 0/1 Response © Evommune, Inc. vIGA: Validated Investigator’s Global Assessment Visit EVO301 Placebo Week 4 4.2% 0% Week 8 12.5% 0% Week 12 22.9% 0% vIGA Response (≥2-point improvement and a score of 0 or 1) ↑ Dosed at Weeks 0 and 4 Placebo EVO301
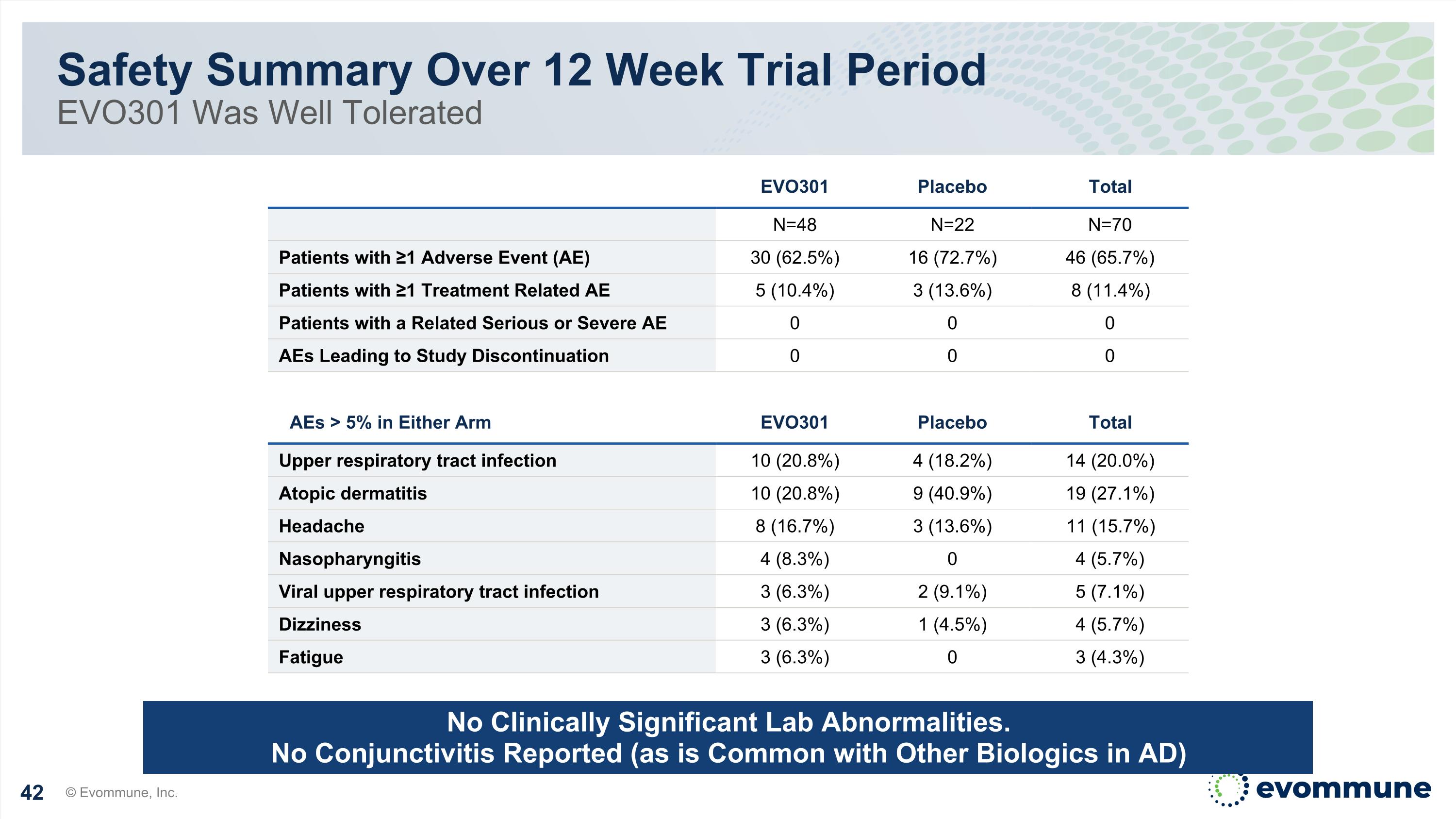
Safety Summary Over 12 Week Trial Period EVO301 Was Well Tolerated © Evommune, Inc. No Clinically Significant Lab Abnormalities. No Conjunctivitis Reported (as is Common with Other Biologics in AD) EVO301 Placebo Total N=48 N=22 N=70 Patients with ≥1 Adverse Event (AE) 30 (62.5%) 16 (72.7%) 46 (65.7%) Patients with ≥1 Treatment Related AE 5 (10.4%) 3 (13.6%) 8 (11.4%) Patients with a Related Serious or Severe AE 0 0 0 AEs Leading to Study Discontinuation 0 0 0 AEs > 5% in Either Arm EVO301 Placebo Total Upper respiratory tract infection 10 (20.8%) 4 (18.2%) 14 (20.0%) Atopic dermatitis 10 (20.8%) 9 (40.9%) 19 (27.1%) Headache 8 (16.7%) 3 (13.6%) 11 (15.7%) Nasopharyngitis 4 (8.3%) 0 4 (5.7%) Viral upper respiratory tract infection 3 (6.3%) 2 (9.1%) 5 (7.1%) Dizziness 3 (6.3%) 1 (4.5%) 4 (5.7%) Fatigue 3 (6.3%) 0 3 (4.3%)
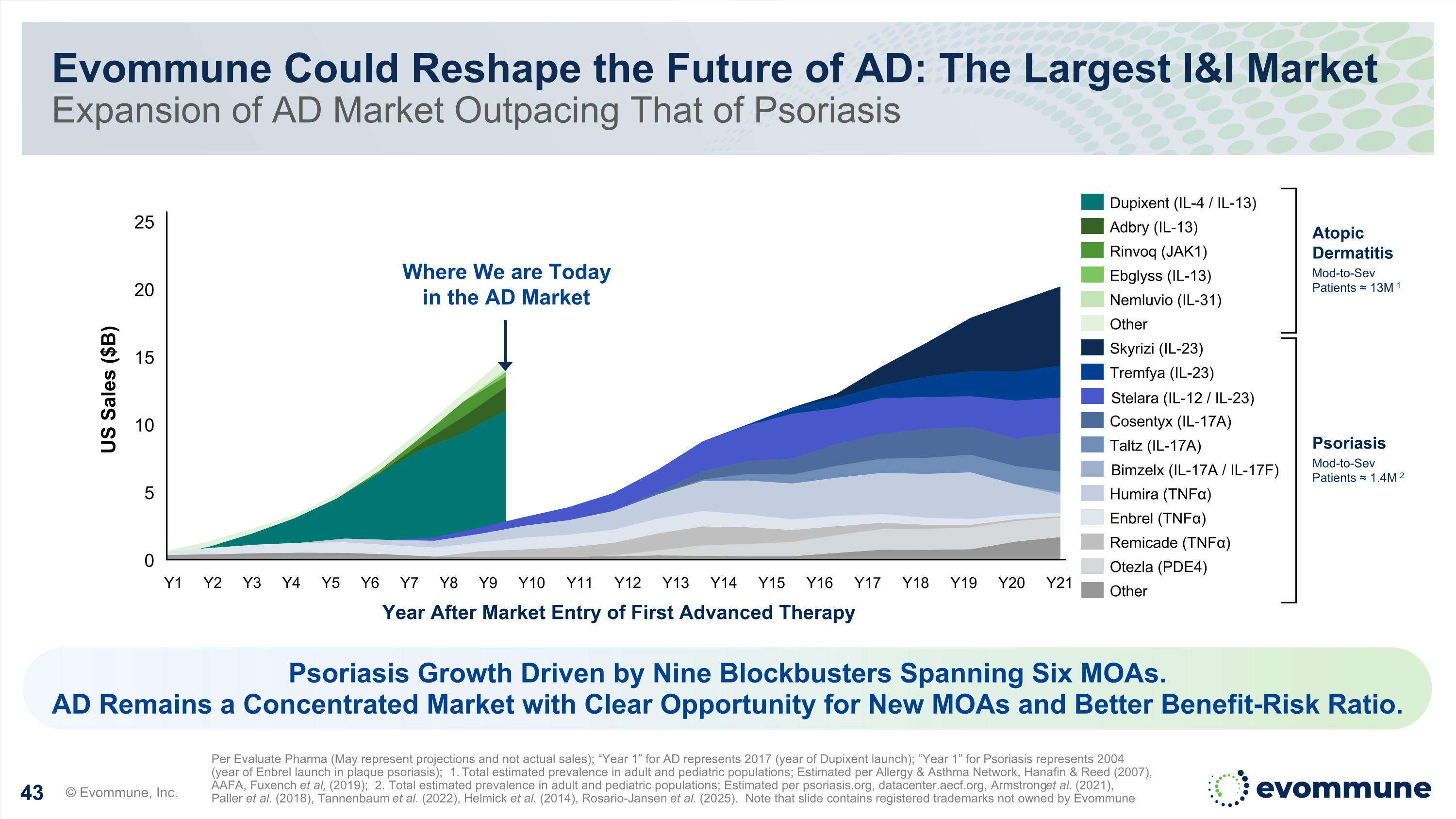
Evommune Could Reshape the Future of AD: The Largest I&I Market Expansion of AD Market Outpacing That of Psoriasis © Evommune, Inc. Per Evaluate Pharma (May represent projections and not actual sales); “Year 1” for AD represents 2017 (year of Dupixent launch); “Year 1” for Psoriasis represents 2004 (year of Enbrel launch in plaque psoriasis); 1. Total estimated prevalence in adult and pediatric populations; Estimated per Allergy & Asthma Network, Hanafin & Reed (2007), AAFA, Fuxench et al, (2019); 2. Total estimated prevalence in adult and pediatric populations; Estimated per psoriasis.org, datacenter.aecf.org, Armstrong et al. (2021), Paller et al. (2018), Tannenbaum et al. (2022), Helmick et al. (2014), Rosario-Jansen et al. (2025). Note that slide contains registered trademarks not owned by Evommune 0 5 10 15 20 25 Year After Market Entry of First Advanced Therapy US Sales ($B) Y1 Y2 Y3 Y4 Y5 Y6 Y7 Y8 Y9 Y10 Y11 Y12 Y13 Y14 Y15 Y16 Y17 Y18 Y19 Y20 Y21 Dupixent (IL-4 / IL-13) Adbry (IL-13) Rinvoq (JAK1) Ebglyss (IL-13) Other Skyrizi (IL-23) Tremfya (IL-23) Stelara (IL-12/IL-23) Cosentyx (IL-17A) Taltz (IL-17A) Bimzelx (IL-17A/IL-17F) Humira (TNFα) Enbrel (TNFα) Remicade (TNFα) Otezla (PDE4) Other Atopic Dermatitis Mod-to-Sev Patients ≈ 13M 1 Where We are Today in the AD Market Psoriasis Growth Driven by Nine Blockbusters Spanning Six MOAs. AD Remains a Concentrated Market with Clear Opportunity for New MOAs and Better Benefit-Risk Ratio. Stelara (IL-12 / IL-23) Bimzelx (IL-17A / IL-17F) Psoriasis Mod-to-Sev Patients ≈ 1.4M 2 Nemluvio (IL-31)
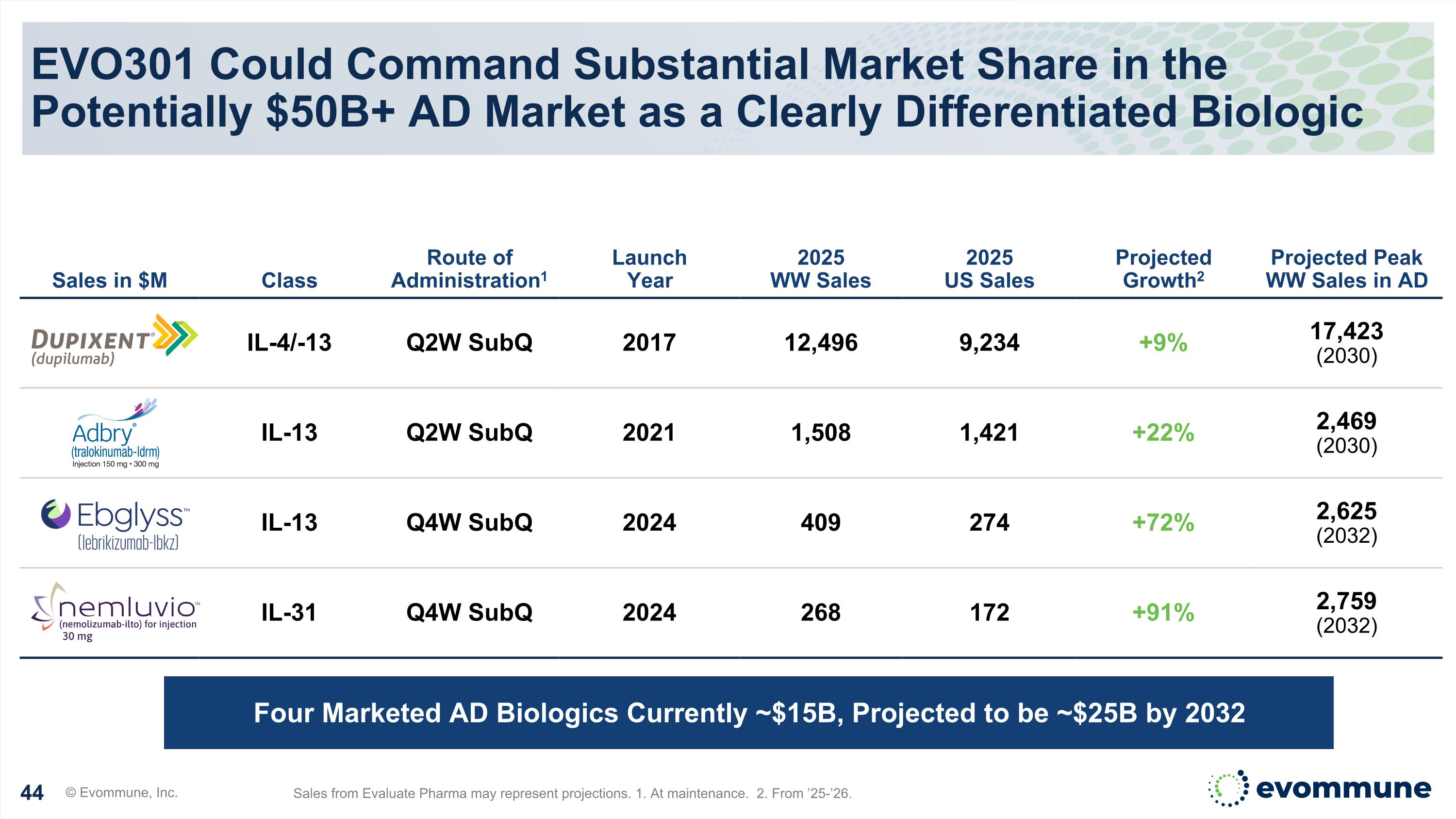
EVO301 Could Command Substantial Market Share in the Potentially $50B+ AD Market as a Clearly Differentiated Biologic © Evommune, Inc. Sales from Evaluate Pharma may represent projections. 1. At maintenance. 2. From ’25-’26. Sales in $M Class Route of Administration1 Launch Year 2025 WW Sales 2025 US Sales Projected Growth2 Projected Peak WW Sales in AD IL-4/-13 Q2W SubQ 2017 12,496 9,234 +9% 17,423 (2030) IL-13 Q2W SubQ 2021 1,508 1,421 +22% 2,469 (2030) IL-13 Q4W SubQ 2024 409 274 +72% 2,625 (2032) IL-31 Q4W SubQ 2024 268 172 +91% 2,759 (2032) Four Marketed AD Biologics Currently ~$15B, Projected to be ~$25B by 2032
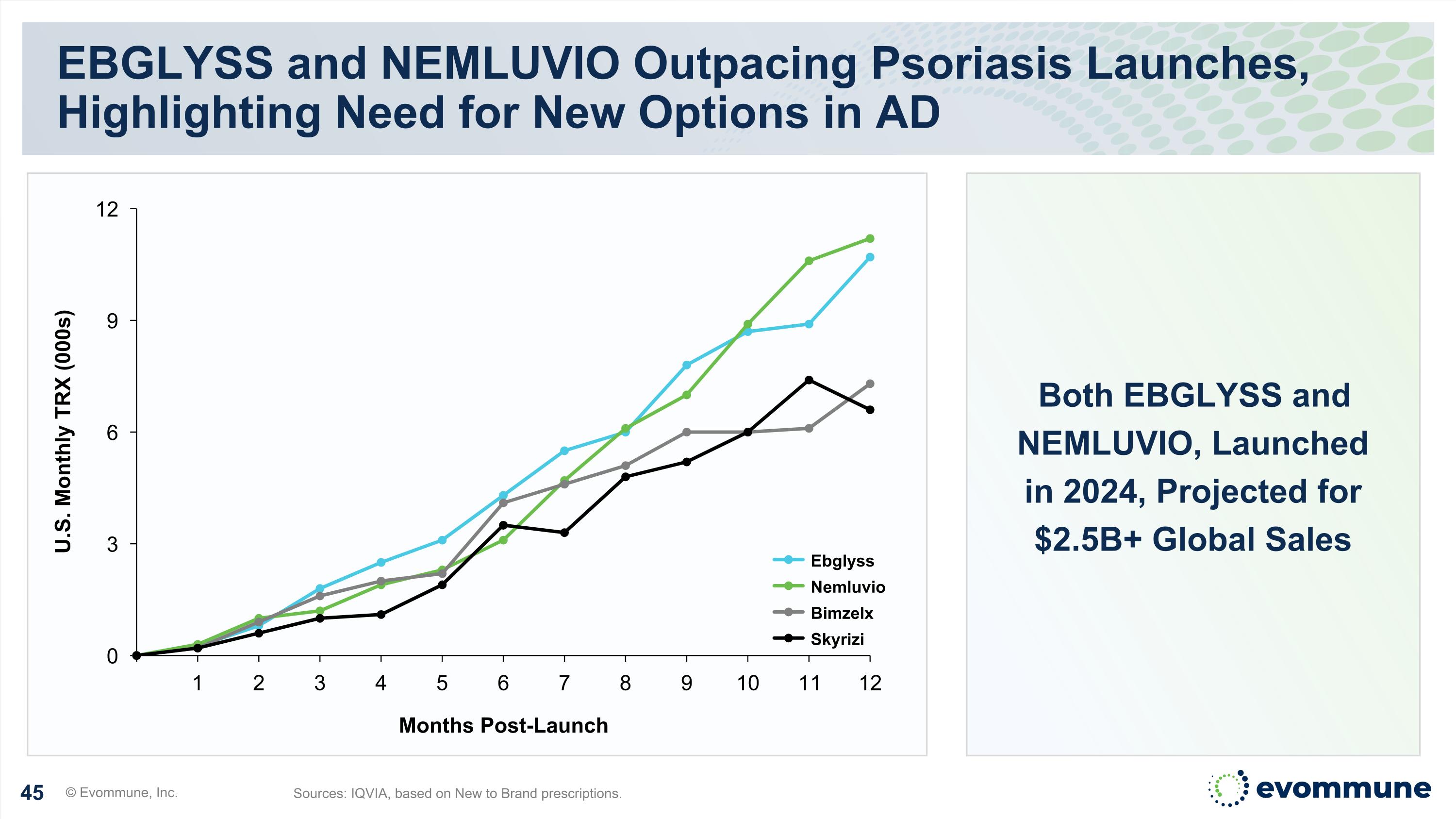
Both EBGLYSS and NEMLUVIO, Launched in 2024, Projected for $2.5B+ Global Sales EBGLYSS and NEMLUVIO Outpacing Psoriasis Launches, Highlighting Need for New Options in AD © Evommune, Inc. Sources: IQVIA, based on New to Brand prescriptions. 1 2 3 4 5 6 7 8 9 10 11 12 Months Post-Launch U.S. Monthly TRX (000s) Ebglyss Nemluvio Bimzelx Skyrizi

IL-18BP Evommune Could Reshape the Future of AD: The Largest I&I Market Novel MoAs Enable Treatment Across AD Patient Journey and Severity Spectrum Topical Therapies MRGPRX2 Inhibitors EVO756 and EVO301 Have Synergistic Potential to Address Different Segments of the AD Landscape An AD Patient’s Treatment Journey EVO756 as a first-line oral treatment post topical therapies EVO301 as a preferred biologic for moderate-to-severe patients EVO756 JAK Inhibitors Injection Oral Topical Approved Biologics (IL-4 / IL-13, IL-31) IL-2 / Other EVO301 © Evommune, Inc.

Company Overview © Evommune, Inc.

Luis Peña Founder, President & CEO Eugene Bauer, MD Founder, CMO Kyle Carver, MBA CFO Greg Moss, Esq CBO & CLO Jeegar Patel, PhD CSO Janice Drew, MPH Chief of Development Operations Lou Sehl, PhD SVP, Technical Operations Daniel Burge, MD SVP, Clinical Development Leadership in >25 Companies Key Roles in Almost 30 NDA / BLAs (Acquired by Eli Lilly for $1.1B) (Acquired by Sanofi for $1.9B) (Acquired by GlaxoSmithKline for $2.9B) (Acquired by LEO Pharma for $288M) (Acquired by Bristol Myers Squibb for $13.1B) (Acquired by Eli Lilly for $6.5B) (Acquired by Stiefel for $930M) (Acquired by Angiotech for ~$50M) Proven and Experienced Leadership Team Has Delivered Almost 30 NDAs and BLAs Footnotes: Acquisition prices from press releases Mark Jackson, MD SVP, Clinical Development © Evommune, Inc.

Mid-stage clinical company developing novel therapeutics for immune-mediated chronic inflammatory diseases Two programs in Phase 2: EVO756 (oral MRGPRX2 antagonist) in chronic spontaneous urticaria and atopic dermatitis, with migraine trial planned for mid-2026 EVO301 (long-acting IL-18bp fusion protein) in atopic dermatitis Three clinical data readouts in 2026: EVO301 reported positive data in a Phase 2a in AD (Feb 2026), moving to Phase 2b with subcutaneous formulation EVO756 Phase 2b in CSU expected in Q2 2026 EVO756 Phase 2b in AD expected in H2 2026 Proven and experienced leadership team has played key roles in almost 30 NDAs and BLAs Steady cadence of new programs entering the clinic in a broad range of inflammatory diseases © Evommune, Inc. $173 million IPO in Nov 2025 $125 million Private Placement in Feb 2026 ~$335 million of cash & investments as of December 31, 2025 (pro forma with PIPE net proceeds) Strong Cash Position with Multiple Clinical Milestones in 2026

Thank You! © Evommune, Inc.